Abstract
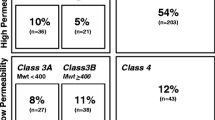
Here, we compile the Biopharmaceutics Drug Disposition Classification System (BDDCS) classification for 927 drugs, which include 30 active metabolites. Of the 897 parent drugs, 78.8% (707) are administered orally. Where the lowest measured solubility is found, this value is reported for 72.7% (513) of these orally administered drugs and a dose number is recorded. The measured values are reported for percent excreted unchanged in urine, LogP, and LogD 7.4 when available. For all 927 compounds, the in silico parameters for predicted Log solubility in water, calculated LogP, polar surface area, and the number of hydrogen bond acceptors and hydrogen bond donors for the active moiety are also provided, thereby allowing comparison analyses for both in silico and experimentally measured values. We discuss the potential use of BDDCS to estimate the disposition characteristics of novel chemicals (new molecular entities) in the early stages of drug discovery and development. Transporter effects in the intestine and the liver are not clinically relevant for BDDCS class 1 drugs, but potentially can have a high impact for class 2 (efflux in the gut, and efflux and uptake in the liver) and class 3 (uptake and efflux in both gut and liver) drugs. A combination of high dose and low solubility is likely to cause BDDCS class 4 to be underpopulated in terms of approved drugs (N = 53 compared with over 200 each in classes 1–3). The influence of several measured and in silico parameters in the process of BDDCS category assignment is discussed in detail.









Similar content being viewed by others
References
Wu C-Y, Benet LZ. Predicting drug disposition via application of BCS: transport/absorption/elimination interplay and development of a biopharmaceutics drug disposition classification system. Pharm Res. 2005;22:11–23.
Benet LZ, Amidon GL, Barends DM, Lennernäs H, Polli JE, Shah VP, et al. The use of BDDCS in classifying the permeability of marketed drugs. Pharm Res. 2008;25:483–8.
Takagi T, Ramachandran C, Bermejo M, Yamashita S, Yu LX, Amidon GL. A provisional biopharmaceutical classification of the top 200 oral drug products in the United States, Great Britain, Spain and Japan. Mol Pharmaceut. 2006;3:631–43.
Food and Drug Administration. Guidance for industry: waiver of in vivo bioavailability and bioequivalence studies for immediate-release solid oral dosage forms based on a biopharmaceutics classification system. Food and Drug Administration, Rockville, MD. 2000. www.fda.gov/cder/guidance/index.htm.
Amidon GL, Lennernäs H, Shah VP, Crison JR. A theoretical basis for a biopharmaceutics drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm Res. 1995;12:413–20.
Benet LZ, Larregieu CA. The FDA should eliminate the ambiguities in the current BCS biowaiver guidance and make public the drugs for which BCS biowaivers have been granted. Clin Pharmacol Ther. 2010;88:405–7.
Chen ML, Amidon GL, Benet LZ, Lennernäs H, Yu LX. The BCS, BDDCS and regulatory guidances. Pharm Res. 2011;28:1774–8.
Lennernäs H. Intestinal permeability and its relevance for absorption and elimination. Xenobiotica. 2007;37:1015–51.
European Medicines Agency. Guideline on the investigation of bioequivalence. Doc. Ref.:CPMP/EWP/QWP/1401/98 Rev.1/Corr. http://www.ema.europa.eu/pdfs/human/qwp/140198enrev1fin.pdf. Accessed 28 March 2010.
Chen ML, Yu L. The use of drug metabolism for prediction of intestinal permeability. Mol Pharmaceut. 2009;6:74–81.
Fraga S, Pinho MJ, Soares-da-Silva P. Expression of LAT1 and LAT2 amino acid transporters in human and rat intestinal epithelial cells. Amino Acids. 2005;29:229–33.
Lyrica™ package insert. http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=42134#nlm34090-1.
Shugarts S, Benet LZ. The role of transporters in the pharmacokinetics of orally administered drugs. Pharm Res. 2009;26:2039–54.
Matthews EJ, Krulak NL, Benz RD, Contrera JF. Assessment of the health effects of chemicals in humans: I. QSAR estimation of the maximum recommended therapeutic dose (MRTD) and no effect level (NOEL) of organic chemicals based on clinical trial data. Curr Drug Discov Technol. 2004;1:61–76.
Benet LZ. The drug transporter-metabolism alliance: uncovering and defining the interplay. Mol Pharmaceut. 2009;6:1631–43.
Benet LZ. Predicting drug disposition via application of a biopharmaceutics drug disposition classification system. Basic Clin Pharmacol Toxicol. 2010;106:162–7.
Benet LZ, Cummins CL, Wu C-Y. Transporter-enzyme interactions: implications for predicting drug–drug interactions from in vitro data. Curr Drug Metab. 2003;4:393–8.
Fleischer D, Li C, Zhou Y, Pao LH, Karim A. Drug, meal and formulation interactions influencing drug absorption after oral administration, clinical implications. Clin Pharmacokinet. 1999;36:233–54.
Custodio JM, Wu C-Y, Benet LZ. Predicting drug disposition, absorption/elimination/transporter interplay and the role of food on drug absorption. Adv Drug Deliv Rev. 2008;60:717–33.
Wu C-Y, Benet LZ. Disposition of tacrolimus in isolated perfused rat liver: influence of troleandomycin, cyclosporine, and GG918. Drug Metab Dispos. 2003;31:1292–5.
Lau Y, Huang Y, Frassetto L, Benet LZ. Effect of OATP1B transporter inhibition on the pharmacokinetics of atorvastatin in healthy volunteers. Clin Pharmacol Ther. 2007;81:194–204.
Zheng HX, Huang Y, Frassetto Y, Benet LZ. Elucidating rifampin’s inducing and inhibiting effects on glyburide pharmacokinetics and blood glucose in healthy volunteers: unmasking the differential effects of enzyme induction and transporter inhibition for a drug and its primary metabolite. Clin Pharmacol Ther. 2009;85:78–85.
Lau YY, Okochi H, Huang Y, Benet LZ. Multiple transporters affect the disposition of atorvastatin and its two active hydroxy metabolites: application of in vitro and ex situ systems. J Pharmacol Exp Ther. 2006;316:762–71.
Lau YY, Okochi H, Huang Y, Benet LZ. Pharmacokinetics of atorvastatin and its hydroxy metabolites in rats and the effects of concomitant rifampicin single doses: relevance of first-pass effect from hepatic uptake transporters, and intestinal and hepatic metabolism. Drug Metab Dispos. 2006;34:1175–81.
Frymoyer A, Shugarts S, Browne M, Wu AH, Frassetto L, Benet LZ. Effect of single-dose rifampin on the pharmacokinetics of warfarin in healthy volunteers. Clin Pharmacol Ther. 2010;88:540–7.
Sussman NL, Waltershied M, Butler T, Cali JJ, Riss T, Kelly JH. The predictive nature of high throughput toxicity screening using a human hepatocyte cell line. Cell Notes. 2002;3:7–10.
Wadelius M, Sorlin K, Wallerman O, Karlsson J, Yue Q-Y, Magnusson PKE, et al. Warfarin sensitivity related to CYP2C9, CYP3A5, ABCB1 (MDR1) and other factors. Pharmacogenomics J. 2004;4:40–8.
Oprea TI, Tropsha A, Faulon J-L, Rintoul MD. Systems chemical biology. Nature Chem Biol. 2007;3:447–50.
Oprea TI, May EE, Leitão A, Tropsha A. Computational systems chemical biology. Meth Mol Biol. 2011;672:459–88.
Khandelwal A, Bahadduri PM, Chang C, Polli JE, Swaan PW, Ekins S. Computational models to assign biopharmaceutics drug disposition classification from molecular structure. Pharm Res. 2007;24:2249–62.
Willmann S, Schmitt W, Keldenich J, Lippert J, Dressman JB. A physiological model for the estimation of the fraction dose absorbed in humans. J Med Chem. 2004;47:4022–31.
Zhao YH, Le J, Abraham MH, Hersey A, Edershaw PJ, Luscombe CN, et al. Evaluation of human intestinal absorption data and subsequent derivation of a quantitative structure–activity relationship (QSAR) with the Abraham descriptors. J Pharm Sci. 2001;90:749–84.
Meylan MW, Howard PH, Boethling RS. Improved method for estimating water solubility from octanol water partition coefficient. Environ Toxicol Chem. 1996;15:100–6.
Yazdanian M, Briggs K, Jankovsky C, Hawi A. The “high solubility” definition of the current FDA Guidance on Biopharmaceutical Classification System may be too strict for acidic drugs. Pharm Res. 2004;21:293–9.
Rinaki E, Dokoumetzidis A, Valsami G, Macheras P. Identification of biowaivers among class II drugs: theoretical justification and practical examples. Pharm Res. 2004;21:1567–72.
Oprea TI, Nielsen SK, Ursu O, Yang JJ, Taboureau O, Mathias SL, et al. Associating drugs, targets and clinical outcomes into an integrated network affords a new platform for computer-aided drug repurposing. Mol Inf. 2011;30:100–11.
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46:3–26.
Lipinski CA. Drug-like properties and the causes of poor solubility and poor permeability. J Pharmacol Toxicol Methods. 2000;44:235–49.
Lipinski CA. Chris Lipinski discusses life and chemistry after the Rule of Five. Drug Discov Today. 2003;8:12–6.
Leo AJ. Calculating log Poct from structures. Chem Rev. 1993;93:1281–306.
Clark DE. Rapid calculation of polar molecular surface area and its application to the prediction of transport phenomena. 1. Prediction of intestinal absorption. J Pharm Sci. 1999;88:807–14.
Cruciani G, Crivori P, Carrupt P-A, Testa B. Molecular fields in quantitative structure–permeation relationships: the VolSurf approach. TheoChem-J Mol Struc. 2000;503:17–30.
The VolSurf + program, version 1.0.4 is distributed by Molecular Discovery Ltd. http://www.moldiscovery.com.
Tetko IV. Computing chemistry on the web. Drug Discov Today. 2005;10:1497–500.
Ross DL, Riley CM. Aqueous solubilities of some variously substituted quinolone antimicrobials. Int J Pharmaceutics. 1990;63:237–50.
Rinaki E, Valsami G, Macheras P. Quantitative biopharmaceutics classification system: the central role of dose/solubility ratio. Pharm Res. 2003;20:1917–25.
Thummel KE, Shen DD, Isoherranen N. Design and optimization of dosage regimens: pharmacokinetic data. In: Brunton L, Chabner B, Knollman B, editors. Goodman and Gilman’s the pharmacological basis of therapeutics, vol. 12th. New York: McGraw-Hill; 2011. p. 1891–990 (Appendix II).
Olah M, Rad R, Ostopovici L, Bora A, Hadaruga N, Hadaruga D, et al. WOMBAT and WOMBAT-PK: bioactivity databases for lead and drug discovery. In: Schreiber SL, Kapoor TM, Wess G, editors. Chemical biology: from small molecules to systems biology and drug design. Weinheim: Wiley-VCH; 2007. p. 760–86.
Tetko IV, Oprea TI. Early ADME/T predictions: toy or tool? In: Varnek A, Tropsha A, editors. Chemoinformatics: an approach to virtual screening. London: Royal Society of Chemistry; 2008. p. 240–67.
Oprea TI. Current trends in lead discovery: are we looking at the appropriate properties? J Comput Aided Mol Design. 2002;16:325–34.
Acknowledgments
The authors were supported in part in preparation of the five tables and this manuscript by NIH grants GM-61390, GM-75900 and GM-90457 (LZB), and by GM-095952, MH-084690, and CA-118100 (TIO). We thank Molecular Discovery Ltd. and Professor Cruciani for the VolSurf+ suite license.
Supporting Info Available
An excel file is available as supporting info containing the following data for the 927 drugs dataset: name, BDDCS class, max dose strength value, max dose strength unit, formulation, route, measured solubility, dose number, % excreted unchanged in urine, MW drug, MW solution, pDose, measured LogS molar, measured LogP, measured LogD 7.4, ALOGPS 2.1 solubility, cDose Number (ALOGPS based), minVSLgS, cDose Number (minVSLgS based), cLogP, HBA,HBD, PSA, and violations to Rules of Five. Definitions for the terms used only in the supporting info file may be found at the end of that data set. In addition, box plots of minVSLgS, ALOGPS 2.1 solubility, MLogP, cLogP, MW, PSA parameters against BDDCS are provided.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benet, L.Z., Broccatelli, F. & Oprea, T.I. BDDCS Applied to Over 900 Drugs. AAPS J 13, 519–547 (2011). https://doi.org/10.1208/s12248-011-9290-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-011-9290-9