Abstract
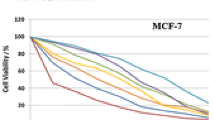
Mononuclear complexes of Co(II), Ni(II), Cu(II), Zr(IV), Pd(II), and Cd(II) with metformin hydrochloride (MF) and 1,4-diacetylbenzene (DAB) have been synthesized, and their structures have been elucidated on the basis of FT-IR, 1H NMR, and electronic spectra, molar conductance, magnetic susceptibility measurements, thermal, and elemental analysis. Molar conductance measurements indicate electrolytic nature of the complexes. According to FT-IR spectra, MF reacts with metal ions as a bidentate ligand through its two imino groups, and DAB reacts as a monodentate ligand through one of two equivalent keto groups. The complexes have octahedral structure, except Pd(II) complex which is square planar. TGA and DTG analyses support the suggested stereochemistry and subsequently the proposed mechanism of thermal decomposition. Coats–Redfern and Horowitz–Metzger equations have been used for calculating kinetic and thermodynamic parameters of the main decomposition step. Molecular modeling calculations confirm the structure of the complexes. The ligands and their complexes in vitro antimicrobial activity tests against some bacterial and fungal strains indicate higher activity of the complexes than that of MF and DAB.

Similar content being viewed by others
REFERENCES
Refat, M.S., Al-Azab, F.M., Al-Maydama, H.M.A., Amin, R.R., Jamil, Y.M.S., and Kobeasy, M.I., Spectrochim. Acta A, 2015, vol. 142, p. 392. https://doi.org/10.1016/j.saa.2015.01.096
Stepensky, D., Friedman, M., Srour, W., Raz, I., and Hoffman, A., J. Control. Release, 2001, vol. 71, p. 107.
Zhu, M., Lu, L., Yang, P., and Jin, X., Acta Cryst.E, 2002, vol. 58, p. 32. https://doi.org/10.1107/S1600536802005342
Shahabadi, N. and Heidari, L. Spectrochim. Acta A, 2012, vol. 97, p. 406. https://doi.org/10.1016/j.saa.2012.06.044
Shahabadi, N. and Heidari, L. Spectrochim. Acta A, 2014, vol. 128, p. 377. https://doi.org/10.1016/j.saa.2014.02.167
Mahmoud, M.A., Abdel-Salam, E.T., Abdel Aal, N.F., Showery, Z.M., and Sallamb, S.A., J. Coord. Chem., 2019, vol. 72, p. 749. https://doi.org/10.1080/00958972.2019.1569642
Mahmoud, M.A., Zaitone, S.A., Ammar, A.M., and Sallam, S.A., J. Mol. Struct., 2016, vol. 1108, p. 60. https://doi.org/10.1016/j.molstruc.2015.11.055.
Mahmoud, M.A., Abdel-Salam, E.T., Abou-Elmagd, M., and Sallam, S., J. Fluorescence, 2019, vol. 29, p. 319. https://doi.org/10.1007/s10895-018-02341-5
Sadeek, S.A., El-Attar, M.S., and Abd El-Hamid, S.M., J. Mol. Struct., 2013, vol. 1051, p. 30. https://doi.org/10.1016/j.molstruc.2013.07.053
Bracke, W., Macromolecules, 1969, vol. 2, p. 286. https://doi.org/10.1021/ma60009a015
Bauer, A.W., Kirby, W.M.M., Sherris, J.C., and Turck, M., AJCP, 1966, vol. 45, p. 493. https://doi.org/10.1093/ajcp/45.4_ts.493
Abd El-Hamid, S.M., Sadeek, S.A., Zordok, W.A., and El-Shwiniy, W.H., J. Mol. Struct., 2019, vol. 1176, p. 422. https://doi.org/10.1016/j.molstruc.2018.08.082.
Sadeek, S.A., El-Attar, M.S., and Abd El-Hamid, S.M., Int. J. Adv. Res., 2014, vol. 2, no. 7, p. 90.
Zhu, L.-N., Liang, M., Wang, Q.-L., Wang, W.-Z., Liao, D.-Z., Jiang, Z.-H., Yan, S.-P., and Cheng, P., J. Mol. Struct., 2003, vol. 657, p.157. https://doi.org/10.1016/S0022-2860(03)00365-X
Vasantha, P., Kumar, B.S., Shekhar, B., and Lakshmi, P.V.A., Mater. Sci. Eng., 2018, vol. 90, p. 621. https://doi.org/10.1016/S0022-2860(03)00365-X
Olar, R., Badea, M., Marinescu, D., Chifiriuc, M.C., Bleotu, C., Grecu, M.N., Iorgulescu, E.E., and Lazar, V., Eur. J. Med. Chem., 2010, vol. 45, p. 3027. https://doi.org/10.1016/j.ejmech.2010.03.033
Sadeek, A.S, Abd El-Hamid, S.M., and El-Shwiniy, W.H., Res. Chem. Intermed., 2016, vol. 42, p. 3183. https://doi.org/10.1007/s11164-015-2205-0
Nandi, S.D., Tetrahedron, 1972, vol. 28, p. 845. https://doi.org/10.1016/0040-4020(72)84044-4
El-Shwiniy, W.H., Shehab, W.S., Mohamed, S.F., and Ibrahium, H.G., Appl. Organomet. Chem., 2018, vol. 32, p. 4503. https://doi.org/10.1002/aoc.4503
El-Shwiniy, W.H. and Sadeek, S.A., Spectrochim. Acta, Part A, 2015, vol. 137, p. 535. https://doi.org/10.1016/j.saa.2014.08.124
Khedr, A.M. and Draz, D.F., J. Coord. Chem., 2010, vol. 63, p. 1418. https://doi.org/10.1080/00958971003774241
Ahmed, F.M., Sadeek, S.A., and El-Shwiniy, W.H., Russ. J. Gen. Chem., 2019, vol. 89, p. 1874. https://doi.org/10.1134/S1070363219090238
Coats, A.W. and Redfern, J.P., Nature, 1964, vol. 201, p. 68. https://doi.org/10.1038/201068a0
Horowitz, H.H. and Metzger, G., Anal. Chem., 1963, vol. 35, no. 10, p. 1464. https://doi.org/10.1021/ac60203a013
Rehman, W., Baloch, M.K., and Badshah, A., Eur. J. Med. Chem., 2008, vol. 43, p. 2380. https://doi.org/10.1016/j.ejmech.2008.01.019
El-Shwiniy, W.H., Shehab, W.S., and Zordok, W.A., J. Mol. Struct., 2020, vol. 1199, p. 126993. https://doi.org/10.1016/j.molstruc.2019.126993.
Saif, M., Mashaly, M.M., Eid, M.F., and Fouad, R., Spectrochim. Acta Part A., 2012, vol. 92, p. 347. https://doi.org/10.1016/j.saa.2012.02.098
Frisch, M.J., et al., Gaussian98, Revision A.6, Inc., Pittsburgh PA, 1998.
Kohn, W. and Sham, L.J., Phys. Rev. A, 1965, vol. 140, p. 1133. https://doi.org/10.1103/PhysRev.140.A1133
Becke, A.D., Phys. Rev. A, 1988, vol. 38, p. 3098. https://doi.org/10.1103/PhysRevA.38.3098
Lee, C., Yang, W., and Parr, R.G., Phys. Rev. B, 1988, vol. 37, p. 785. https://doi.org/10.1103/PhysRevB.37.785
Flurry, R.L.Jr., Molecular Orbital Theory of Bonding in Organic Molecules, New York: Marcel Dekker, 1968. https://doi.org/10.1002/ange.19690810618
Saeed, A., Ashraf, S., Flörke, U., Espinoza, Z.Y.D., Erben, M.F., and Pérez, H., J. Mol. Struct., 2016, vol. 1111, p. 76. https://doi.org/10.1016/j.molstruc.2016.01.074
Daud, A.I., Khairul, W.M., Zuki, H.M., and Kubulat, K., J. Sulfur. Chem., 2014, vol. 35, p. 691. https://doi.org/10.1080/17415993.2014.954248
Kurtaran, R., Odabasoglu, S., Azizoglu, A., Kara, H., and Atakol, O., Polyhedron, 2007, vol. 26, p. 5069. https://doi.org/10.1016/j.poly.2007.07.021
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
El-Shwiniy, W.H., Abbass, L.M., Sadeek, S.A. et al. Synthesis, Structure, and Biological Activity of Some Transition Metal Complexes with the Mixed Ligand of Metformin and 1,4-Diacetylbenzene. Russ J Gen Chem 90, 483–488 (2020). https://doi.org/10.1134/S1070363220030238
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363220030238