Abstract
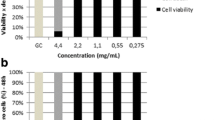
Houttuynia cordata Thunb. is a medicinal and edible plant that has been used to treat phlegm, asthma, cough, carbuncle, swelling, and sore in China for thousands of years. Many previous studies have reported the chemical compositions and pharmacological acivities of hydrodistillation extracted Houttuynia volatile oil. However, no scientific studies on the inhibition activity against virulence factors of Staphylococcus aureus (S. aureus) have been published. So this article analyze the chemical compositions of Houttuynia volatile oil, and evaluate the anti-α-hemolysin (Hla) activity of Houttuynia volatile oil, its three compositions (nonanol, α-terpineol, 4-terpineol) and eleven analogues. In addition, inhibition activity of three compositions to the A549 cells infection of S. aureus was evaluated in vitro. The results is thirty-four ion chromatographic peaks with matching degree ≥90% were identified, and main chemical compositions of Houttuynia volatile oil were β-myrcene (24.48%) and β-phellandrene (11.28%). The MIC of lauraldehyde against S. aureus ATCC 29213 was found to be 8 μg/mL, and houttuynin showed the strongest inhibition activity against the six S. aureus strains with the MIC of 64 or 128 μg/mL. Besides, growth curve analysis revealed the non-bactericidal activity of nonanol, α-terpilenol, and 4-terpineol at sub-MIC. The result of anti-virulence study demonstrated that dodecanol, undecanol, and decanol showed the strongest anti-Hla activity by significantly inhibiting hemolysis of rabbit blood. Nonanol, α-terpilenol, and 4-terpineol can obviously protect A549 cells from α-Hla-mediated injury at 8, 16, and 32 μg/mL in vitro, respectively.



Similar content being viewed by others
REFERENCES
Bhakdi, S. and Tranum-Jensen, J., Microbiol. Rev., 1991, vol. 55, pp. 733–751. https://doi.org/10.1128/mr.55.4.733-751.1991
Dajcs, J.J., Thibodeaux, B.A., Girgis, D.O., and O’Callaghan, R.J., DNA Cell Biol., 2002, vol. 21, pp. 375–382. https://doi.org/10.1089/10445490260099656
Parish, H.J. and Clark, W., J. Pathol. Bacteriol., 2005, vol. 35, pp. 251–258. https://doi.org/10.1002/path.1700350211
Rogolsky, M., Microbiol. Rev., 1979, vol. 43, pp. 320–360. https://doi.org/10.1007/BF02013529
Khodaverdian, V., Pesho, M., Truitt, B., Bollinger, L., Patel, P., Nithianantham, S., Yu, G., Delaney, E., Jankowsky, E. and Shoham, M., Antimicrob. Agents Chemother., 2013, vol. 57, pp. 3645–3652. https://doi.org/10.1128/AAC.00269-13
Xu, Z., Li, K., Pan, T., Liu, J., Li, B., Li, C., Wang, S., Diao, Y., and Liu, X., J. Ethnopharmacol., 2019, vol. 239, p. 111909. https://doi.org/10.1016/j.jep.2019.111909
Xuewen, H., Ping, O., Zhongwei, Y., Zhongqiong, Y., Hualin, F., Juchun, L., Changliang, H., Gang, S., Zhixiang, Y. and Xu, S., World J. Microbiol. Biotechnol., 2018, vol. 34, p. 64. https://doi.org/10.1007/s11274-018-2446-3
Yan, C., Tangjuan, L., Ke, W., Changchun, H., Shuangqi, C., Yingying, H., Zhongye, D., Hong, H., Jinliang, K., and Yiqiang, C., PLoS One, 2016, vol. 11, p. e0153468. https://doi.org/10.1371/journal.pone.0153468
Tang, F., Li, L., Meng, X.-M., Li, B., Wang, C.-Q., Wang, S.-Q., Wang, T.-W., and Tian, Y.-M., Microb. Pathog., 2018, vol. 127, pp. 85–90. https://doi.org/10.1016/j.micpath.2018.11.027
Bing, Z., Zihao, T., Xianhe, L., Gejin, L., Xuming, D., Xiaodi, N., and Jianfeng, W., Front. Microbiol., 2017, vol. 8, pp. 1715. https://doi.org/10.3389/fmicb.2017.01715
Silva, L.N., Hora, G., Soares, T.A., Bojer, M.S., Ingmer, H., Macedo, A.J., and Trentin, D.S., Scientific Reports, 2017, vol. 7, p. 2823. https://doi.org/10.1038/s41598-017-02712-1
Lakshmi, S.A., Bhaskar, J.P., Krishnan, V., Sethupathy, S., and Pandian, S.K., J. Biotechnol., 2020, vol. 317. https://doi.org/10.1016/j.jbiotec.2020.04.014
Commission, C.P., Beijing: People’s Medical Publishing House, 2015, vol. 04, pp. 132–135.
Yang, J., Chen, W.Y., Fu, Y., Yang, T., Luo, X.D., Wang, Y.H., and Wang, Y.H., J. Ethnopharmacol., 2020, vol. 249, p. 112430. https://doi.org/10.1016/j.jep.2019.112430
Laloo, D. and Hemalatha, S., Pharmacogn. Rev., 2011, vol. 5, no. 10, p. 147. https://doi.org/10.4103/0973-7847.91108
Kim, G.S., Kim, D.H., Lim, J.J., Lee, J.J., Han, D.Y., Lee, W.M., Jung, W.C., Min, W.G., Won, C.G., and Rhee, M.H., Biol. Pharm. Bull., 2008, vol. 31, p. 2012. https://doi.org/10.1248/bpb.31.2012
Cheng, B.H., Chan, Y.W., Chan, C.L., Lin, H.Q., Han, X.Q., Zhou, X., Wan, C.C., Wang, Y.F., Leung, P.C., Fung, P.K., and Polym, C., Carbohydr. Polym., 2014, vol. 69, pp. 244–249. https://doi.org/10.1016/j.carbpol.2013.12.048
Lau, K.M., Lee, K.M., Koon, C.M., Cheung, S.F., Lau, C.P., Ho, H.M., Lee, Y.H., Au, W.N., Cheng, H.K., and Lau, B.S., J. Ethnopharmacol., 2008, vol. 118, pp. 79–85. https://doi.org/10.1016/j.jep.2008.03.018
Zhu, H., Lu, X., Ling, L., Li, H., Ou, Y., Shi, X., Lu, Y., Zhang, Y., and Chen, D., J. Ethnopharmacol., 2018, vol. 28, pp. 28–33. https://doi.org/10.1016/j.jep.2018.02.016
Probstle, A. and Bauer, R., Planta Med., 1992, vol. 58, pp. 568–569. https://doi.org/10.1055/s-2006-961554
Li, W., Ping, Z., Zhang, Y., and He, L., J. Ethnopharmacol., 2011, vol. 133, pp. 922–927. https://doi.org/10.1016/j.jep.2010.10.048
Li, G.Z., Chai, O.H., Lee, M.S., Han, E.H., Kim, H.T., and Song, C.H., Biol. Pharm. Bull., 2005, vol. 28, pp. 1864–1868. https://doi.org/10.1248/bpb.28.1864
Nuengchamnong, N., Krittasilp, K., and Ingkaninan, K., Food Chem., 2009, vol. 117, pp. 750–756. https://doi.org/10.1016/j.foodchem.2009.04.071
Li, W., Fan, T., Zhang, Y., Fan, T., Zhou, P., Niu, X., and He, L., Phytother. Res., 2013, vol. 27, pp. 1629–1639. https://doi.org/10.1002/ptr.4905
Ling, L., Lu, Y.A., Zhang, Y., Zhu, H., Tu, P.A., Li, H.B., and Chen, D., J. Phytomed., 2020, vol. 67. https://doi.org/10.1016/j.phymed.2019.153150
Ma, Q., Wang, Z., Wei, R., Liu, W., and Huang, H., J. Ethnopharmacol., 2017, vol. 195, pp. 166–172. https://doi.org/10.1016/j.jep.2016.11.013
Qing-Song, X. and Sai-Jin, W., Chem. Nat. Compd., 2015, vol. 51, pp. 359–360. https://doi.org/10.1007/s10600-015-1283-y
Verma, R.S., Joshi, N., Padalia, R.C., Singh, V.R., Goswami, P., Kumar, A., Iqbal, H., Verma, R.K., Chanda, D., Chauhan, A., and Saikia, D., Chem. Biodiversity, 2017, vol. 14, p. e1700189. https://doi.org/10.1002/cbdv.201700189
Jianmei, P., Wujun, D., Yuhuan, L., Xuejun, X., Zhihua, L., Huazhen, H., Lingmin, J., and Yuling, L., Molecules, 2017, vol. 22, p. 293. https://doi.org/10.3390/molecules22020293
Lu, H.M., Liang, Y.Z., and Chen, S., J. Ethnopharmacol., 2006, vol. 105, pp. 436–440. https://doi.org/10.1016/j.jep.2005.11.018
Qing-Song, X., Zhan, W., and Hua-Ru, Y., J. Anal. Sci., 2012, vol. 28, pp. 28–33.
Menestrina, G., J. Membr. Biol., 1986, vol. 90, pp. 177–190. https://doi.org/10.1007/BF01869935
Rasmussen, T.B. and Givskov, M., Microbiology, 2006, vol. 152, pp. 895–904. https://doi.org/10.1099/mic.0.28601-0
Wardenburg, J.B. and Schneewind, O., J. Exp. Med., 2008, vol. 205, pp. 287–294. https://doi.org/10.1084/jem.20072208
Martínez, O.F., Cardoso, M.H., Ribeiro, S.M., and Franco, O.L., Front. Cell Infect. Microbiol., 2019, vol. 9, p. 74. https://doi.org/10.3389/fcimb.2019.00074
Zhou, L.L. and Yang, G.G., Chin. J. Chem., 2019, vol. 037, pp. 183–193.
Balamurugan, P., Krishna, V.P., Bharath, D., Lavanya, R., Vairaprakash, P., and Princy, S.A., Front. Microbiol., 2017, vol. 8, p. 1290. https://doi.org/10.3389/fmicb.2017.01290
Jóźwiak, M., Struga, M., Roszkowski, P., Filipek, A., Nowicka, G., and Olejarz, W., Biomed. Pharmacother., 2019, vol. 110, pp. 618–630. https://doi.org/10.1016/j.biopha.2018.12.005
Funding
This work was supported by the National Key Research and Development Project of China (Grant no. 2017YFD0501400).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
This article does not contain any studies involving animals or human participants performed by any of the authors.
Rights and permissions
About this article
Cite this article
Yingxue Yang, Wang, L., Zhao, B. et al. Chemical Compositions of Houttuynia cordata Thunb. Volatile Oil and Its Analogues Attenuate Staphylococcus aureus Virulence by Targeting α-Hemolysin. Russ J Bioorg Chem 48 (Suppl 1), S166–S177 (2022). https://doi.org/10.1134/S1068162023010284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023010284