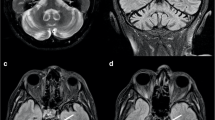
Abstract
Glutamate (Glu) has been reported to be closely related to the pathophysiology of Tic Disorders (TD). By using proton magnetic resonance spectroscopy (1H-MRS), we aimed to investigate the relationship between in vivo Glu levels and the severity of TD. We performed a cross-sectional study in medication-free patients with TD and healthy controls aged between 5 and 13 years using 1H-MRS at 3 T. First, we measured the Glu levels in both patients and controls and observed the difference in subgroups, including mild TD patients and moderate TD patients. We then examined the correlations between the Glu levels and clinical features of the patients. Finally, we assessed the diagnostic value of 1H-MRS and the influencing factors. Our results show that the Glu levels in the striatum of all patients with TD were not significantly different from those of the healthy controls. Subgroup analysis revealed that the Glu levels in the moderate TD group were higher than those in the mild TD group and healthy controls. The correlation analysis showed that Glu levels are strongly positive correlated with TD severity. The optimal cutoff value of Glu levels to differentiate mild tics from moderate tics was 1.244, with a sensitivity of 88.2% and a specificity of 94.7%. Multiple linear regression models revealed that the severity of TD is one of the important factors that affect Glu levels. We conclude that Glu levels are mainly associated with the severity of tics, thus it could serve as a key biomarker for TD classification.






Similar content being viewed by others
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Association AP (2013) The diagnostic and statistical manual of mental disorders (DSM-5), 5th edn. American Psychiatric Association, Arlington
Bloch MH, Peterson BS, Scahill L et al (2006) Adulthood outcome of tic and obsessive-compulsive symptom severity in children with Tourette syndrome. Arch Pediatr Adolesc Med 160(1):65–69. https://doi.org/10.1001/archpedi.160.1.65
Buonocore MH, Maddock RJ (2015) Magnetic resonance spectroscopy of the brain: a review of physical principles and technical methods. Rev Neurosci 26(6):609–632. https://doi.org/10.1515/revneuro-2015-0010
Cavanna AE, David K, Bandera V et al (2013) Health-related quality of life in Gilles de la Tourette syndrome: a decade of research. Behav Neurol 27(1):83–93. https://doi.org/10.3233/ben-120296
Chung YL (2017) Magnetic resonance spectroscopy (MRS)-based methods for examining cancer metabolism in response to oncogenic kinase drug treatment. Methods Mol Biol 1636:393–404. https://doi.org/10.1007/978-1-4939-7154-1_25
Di Lazzaro V, Pilato F, Dileone M et al (2006) GABAA receptor subtype specific enhancement of inhibition in human motor cortex. J Physiol 575(3):721–726. https://doi.org/10.1113/jphysiol.2006.114694
Dou W, Kaufmann J, Li M, Zhong K, Walter M, Speck O (2015) The separation of Gln and Glu in STEAM: a comparison study using short and long TEs/TMs at 3 and 7 T. MAGMA 28(4):395–405. https://doi.org/10.1007/s10334-014-0479-7
Fan S, Cath DC, van den Heuvel OA et al (2017) Abnormalities in metabolite concentrations in tourette’s disorder and obsessive-compulsive disorder-A proton magnetic resonance spectroscopy study. Psychoneuroendocrinology 77:211–217. https://doi.org/10.1016/j.psyneuen.2016.12.007
Floresco SB, Todd CL, Grace AA (2001) Glutamatergic afferents from the hippocampus to the nucleus accumbens regulate activity of ventral tegmental area dopamine neurons. J Neurosci 21(13):4915–4922. https://doi.org/10.1523/jneurosci.21-13-04915.2001
Floresco SB, West AR, Ash B, Moore H, Grace AA (2003) Afferent modulation of dopamine neuron firing differentially regulates tonic and phasic dopamine transmission. Nat Neurosci 6(9):968–973. https://doi.org/10.1038/nn1103
Freed RD, Coffey BJ, Mao X et al (2016) Decreased anterior cingulate cortex γ-aminobutyric acid in youth with Tourette’s disorder. Pediatr Neurol 65:64–70. https://doi.org/10.1016/j.pediatrneurol.2016.08.017
Hirschtritt ME, Lee PC, Pauls DL et al (2015) Lifetime prevalence, age of risk, and genetic relationships of comorbid psychiatric disorders in Tourette syndrome. JAMA Psychiat 72(4):325–333. https://doi.org/10.1001/jamapsychiatry.2014.2650
Kanaan AS, Gerasch S, García-García I et al (2017) Pathological glutamatergic neurotransmission in Gilles de la Tourette syndrome. Brain 140(1):218–234. https://doi.org/10.1093/brain/aww285
Leckman JF, Riddle MA, Hardin MT et al (1989) The Yale Global Tic Severity Scale: initial testing of a clinician-rated scale of tic severity. J Am Acad Child Adolesc Psychiatry 28(4):566–573. https://doi.org/10.1097/00004583-198907000-00015
Leckman JF, Bloch MH, Smith ME, Larabi D, Hampson M (2010) Neurobiological substrates of Tourette’s disorder. J Child Adolesc Psychopharmacol 20(4):237–247. https://doi.org/10.1089/cap.2009.0118
Lee P, Adany P, Choi IY (2017) Imaging based magnetic resonance spectroscopy (MRS) localization for quantitative neurochemical analysis and cerebral metabolism studies. Anal Biochem 529:40–47. https://doi.org/10.1016/j.ab.2017.01.007
Lerner A, Bagic A, Simmons JM et al (2012) Widespread abnormality of the γ-aminobutyric acid-ergic system in Tourette syndrome. Brain 135(6):1926–1936. https://doi.org/10.1093/brain/aws104
Lisman JE, Grace AA (2005) The hippocampal-VTA loop: controlling the entry of information into long-term memory. Neuron 46(5):703–713. https://doi.org/10.1016/j.neuron.2005.05.002
Mahone EM, Puts NA, Edden R, Ryan M, Singer HS (2018) GABA and glutamate in children with Tourette syndrome: a 1H MR spectroscopy study at 7T. Psychiatry Res Neuroimaging 273:46–53. https://doi.org/10.1016/j.pscychresns.2017.12.005
Menzies L, Chamberlain SR, Laird AR, Thelen SM, Sahakian BJ, Bullmore ET (2008) Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: the orbitofronto-striatal model revisited. Neurosci Biobehav Rev 32(3):525–549. https://doi.org/10.1016/j.neubiorev.2007.09.005
Naaijen J, Forde NJ, Lythgoe DJ et al (2017) Fronto-striatal glutamate in children with Tourette’s disorder and attention-deficit/hyperactivity disorder. NeuroImage Clin 13:16–23. https://doi.org/10.1016/j.nicl.2016.11.013
Pauls DL, Abramovitch A, Rauch SL, Geller DA (2014) Obsessive-compulsive disorder: an integrative genetic and neurobiological perspective. Nat Rev Neurosci 15(6):410–424. https://doi.org/10.1038/nrn3746
Provencher SW (1993) Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med 30(6):672–679. https://doi.org/10.1002/mrm.1910300604
Puts NA, Harris AD, Crocetti D et al (2015) Reduced GABAergic inhibition and abnormal sensory symptoms in children with Tourette syndrome. J Neurophysiol 114(2):808–817. https://doi.org/10.1152/jn.00060.2015
Sanaei Nezhad F, Anton A, Michou E, Jung J, Parkes LM, Williams SR (2018) Quantification of GABA, glutamate and glutamine in a single measurement at 3 T using GABA-edited MEGA-PRESS. NMR Biomed 31(1):e3847. https://doi.org/10.1002/nbm.3847
Shprecher DR, Kious BM, Himle MH (2015) Advances in mechanistic understanding and treatment approaches to Tourette syndrome. Discov Med 20(111):295–301.
Silvestri PR, Chiarotti F, Baglioni V, Neri V, Cardona F, Cavanna AE (2016) Health-related quality of life in patients with Gilles de la Tourette syndrome at the transition between adolescence and adulthood. Neurol Sci 37(11):1857–1860. https://doi.org/10.1007/s10072-016-2682-y
Singer HS (2010) Treatment of Tics and Tourette syndrome. Curr Treat Options Neurol 12(6):539–561. https://doi.org/10.1007/s11940-010-0095-4
Singer HS, Morris C, Grados M (2010) Glutamatergic modulatory therapy for Tourette syndrome. Med Hypotheses 74(5):862–867. https://doi.org/10.1016/j.mehy.2009.11.028
Specht MW, Woods DW, Piacentini J et al (2011) Clinical characteristics of children and adolescents with a primary Tic Disorder. J Dev Phys Disabil 23(1):15–31. https://doi.org/10.1007/s10882-010-9223-z
Acknowledgements
We acknowledge the funds of the National Natural Science Foundation of China (No. 81874486; No. 81873893; No. 82004418), Shanghai Science and Technology Commission Research Project (No. 18401902700; No. 17401931200), Shanghai Municipal Health Commission (No. 2018LQ010), Shanghai Municipal Science and Technology Major Project (No. 2018SHZDZX01), Zhejiang Medical Health Science and Technology Program (No. 2022RC280), Shanghai Jiaotong University School of Medicine high level local college construction project (No. 18zxy006), Shaoxing University college-level research projects (No. 20205038; No. 2020LG1007).
Funding
This study was supported by the grant awarded by the National Natural Science Foundation of China (No. 81874486; No. 81873893; No. 82004418), Shanghai Science and Technology Commission Research Project (No. 18401902700; No. 17401931200), Shanghai Municipal Health Commission (No. 2018LQ010), Shanghai Municipal Science and Technology Major Project (No. 2018SHZDZX01). Zhejiang Medical Health Science and Technology Program (No. 2022RC280). Shanghai Jiaotong University School of Medicine high level local college construction project (No. 18zxy006). Shaoxing University college-level research projects (No. 20205038; No. 2020LG1007).
Author information
Authors and Affiliations
Contributions
Authors JH, XZ, KJ, X-Y Z and MW designed the study, enrolled the participants, analyzed data and wrote the manuscript. Author ZZ did the MR examinations. Authors YL and X-YZ processed the 1H-MRS data and edited the manuscript critically. All authors contributed to have approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent to Publish
All presentations have consent for publication.
Ethical Approval
This study was approved by the Xinhua Hospital Ethics Committee affiliated to Shanghai JiaoTong University School of Medicine (Approval No. XHEC-C-2018-046-2).
Rights and permissions
About this article
Cite this article
Hao, J., Zhang, X., Liu, Y. et al. Cross-sectional Exploration of the Relationship Between Glutamate Abnormalities and Tic Disorder Severity Using Proton Magnetic Resonance Spectroscopy. Phenomics 3, 138–147 (2023). https://doi.org/10.1007/s43657-022-00064-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-022-00064-z