Abstract
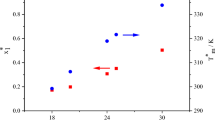
Fatty compounds have significant industrial applications as coatings, plastics, cleaning products, emulsifiers, structuring, and gelling agents. Due to these applications, it is important to understand their physical and chemical properties, such as melting behavior, crystallization processes, and phase transitions, all of them related to solid–liquid equilibrium (SLE). In this study, six binary mixtures composed of tristearin (SSS) plus fatty acids (capric acid, lauric acid, and myristic acid) and tristearin plus fatty alcohol compounds (1-decanol, 1-dodecanol, and 1-tetradecanol) were investigated using Differential Scanning Calorimetry (DSC) and optical microscopy analysis. The phase diagrams formed by tristearin + fatty acids presented a monotectic behavior and the phase diagrams formed by tristearin + fatty alcohol compounds, on the other hand, exhibited a solid solution formation on the entire composition range, although the liquidus line shape was very similar to that observed in the systems formed by fatty acids. The liquidus line of all systems were well-described by NRTL and three-suffix Margules models, and also by ideal assumption. The best result was observed for the three-suffix Margules model, with a root mean square deviation (RMSD) equal to 0.26.
Graphical Abstract













Similar content being viewed by others
Availability of data and material
The authors declare that the data and material are available.
Code availability
Not applicable.
Abbreviations
- ARD:
-
Average relative deviation
- DSC:
-
Differential scanning calorimetry
- NRTL:
-
Non-random two-liquid model
- RMSD:
-
Root mean square deviation
- SLE:
-
Solid–liquid equilibrium
- SSS:
-
Tristearin
- TAG:
-
Triacylglycerols
References
Carareto NDD, Costa MC, Meirelles AJA, Pauly J (2015) High pressure solid-liquid equilibrium of fatty alcohols binary systems from 1-Dodecanol, 1-Tetradecanol, 1-Hexadecanol, and 1-Octadecanol. J Chem Eng Data 60:2966–2973
Carareto NDD, Dos Santos AO, Rolemberg MP, Cardoso LP, Costa MC, Meirelles AJA (2014) ‘On the solid-liquid phase diagrams of binary mixtures of even saturated fatty alcohols: Systems exhibiting peritectic reaction’, Thermochimica Acta. Elsevier B.V., 589, pp 137–147. https://doi.org/10.1016/j.tca.2014.05.022.
Costa MC, Rolemberg MP, Boros LAD, Krähenbühl MA, de Oliveira MG, Meirelles AJA (2007) Solid−liquid equilibrium of binary fatty acid mixtures. J Chem Eng Data 52:30–36. https://doi.org/10.1021/je060146z
Costa MC, Rolemberg MP, Dos Santos AO, Cardoso LP, Krähenbühl MA, Meirelles AJA (2010) Solid-liquid equilibrium of tristearin with refined rice bran and palm oils. J Chem Eng Data 55:5078–5082. https://doi.org/10.1021/je100647b
Costa MC, Boros LAD, Souza JA, Rolemberg MP, Krähenbühl MA, Meirelles AJA (2011) Solid–liquid equilibrium of binary mixtures containing fatty acids and triacylglycerols. J Chem Eng Data 56:3277–3284. https://doi.org/10.1021/je200033b
Craven RJ, Lencki RW (2011) Binary phase behavior of diacid 1,3-diacylglycerols. J Am Oil Chem Soc 88:1125–1134. https://doi.org/10.1007/s11746-011-1777-0
Da Silva RC, De Martini Soares FAS, Maruyama JM, Dagostinho NR, Silva YA, Ract JNR, Gioielli LA (2016) Microscopic approach of the crystallization of tripalmitin and tristearin by microscopy. Chem Phys Lipid 198:1–9. https://doi.org/10.1016/j.chemphyslip.2016.04.004
de Matos FC, da Costa MC, Meirelles AJ, Batista EAC (2015) Binary solid-liquid equilibrium systems containing fatty acids, fatty alcohols and triolein by differential scanning calorimetry. Fluid Phase Equilib 404:1–8. https://doi.org/10.1016/j.fluid.2015.06.015
de Matos FC, da Costa MC, Meirelles AJ, Batista EAC (2016) Binary solid-liquid equilibrium systems containing fatty acids, fatty alcohols and trilaurin by differential scanning calorimetry. Fluid Phase Equilib 423:74–83. https://doi.org/10.1016/j.fluid.2016.04.008
Dorighello Carareto ND, Costa MC, Meirelles AJA, Pauly J (2015) High pressure solid-liquid equilibrium of fatty alcohols binary systems from 1-DODECANOL, 1-Tetradecanol, 1-Hexadecanol, and 1-Octadecanol. J Chem Eng Data 60:2966–2973. https://doi.org/10.1021/acs.jced.5b00330
Eckert KA, Dasgupta S, Selge B, Ay P (2016) Solid liquid phase diagrams of binary fatty acid mixtures—palmitic/stearic with oleic/linoleic/linolenic acid mixture. Thermochim Acta 630:50–63. https://doi.org/10.1016/j.tca.2016.02.008
Fisk PR, Wildey RJ, Girling AE, Sanderson H, Belanger SE, Veenstra G, Nielsen A, Kasai Y, Willing A, Dyer SD, Stanton K (2009) Environmental properties of long chain alcohols. Part 1: physicochemical, environmental fate and acute aquatic toxicity properties. Ecotoxicol Environ Saf 72:980–995. https://doi.org/10.1016/j.ecoenv.2008.09.025
Garti N, Sato K (1988) Crystallization and polymorphism of fats and fatty acids. Marcel Dekker, Nova Iorque
Höhne GWH, Hemminger WF, Flammerseim HJ (2003) Differential Scanning Calorimetry, 2nd edn. Springer, New York
Humphrey KL, Narine SS (2005) Lipid phase behavior. In: Marangoni AG (ed) Fat crystal networks. Taylor & Francis, New York, p 854
Inoue T, Hisatsugu Y, Ishikawa R, Suzuki M (2004a) Solid-liquid phase behavior of binary fatty acid mixtures: 2. Mixtures of oleic acid with lauric acid, myristic acid, and palmitic acid. Chem Phys Lipid 127:161–173. https://doi.org/10.1016/j.chemphyslip.2003.10.013
Inoue T, Hisatsugu Y, Yamamoto R, Suzuki M (2004b) Solid-liquid phase behavior of binary fatty acid mixtures: 1. Oleic acid/stearic acid and oleic acid/behenic acid mixtures. Chem Phys Lipid 127:143–152. https://doi.org/10.1016/j.chemphyslip.2003.09.014
Johnson RW (1988) Fatty alcohols. Fatty acids in industry. Marcel Dekker, New York, pp 217–231
Kellens M, Reynaers H (1992) Study of the polymorphism of saturated monoacid triglycerides I: melting and crystallization behavior of tristearin. Fat Sci Technol 94:94–100. https://doi.org/10.1002/lipi.19920940304
Kellens M, Meeussen W, Gehrke R, Reynaers H (1991) Synchrotron radiation investigations of the polymorphic transitions of saturated monoacid triglycerides. Part 1: Tripalmitin and tristearin. Chem Phys Lipid 58:131–144. https://doi.org/10.1016/0009-3084(91)90119-V
Kogan A, Garti N (2006) Microemulsions as transdermal drug delivery vehicles. Adv Coll Interface Sci 123–126:369–385. https://doi.org/10.1016/j.cis.2006.05.014
Matovic M, Van Miltenburg JC, Los J, Gandolfo FG, Flöter E (2005) Thermal properties of tristearin by adiabatic and differential scanning calorimetry. J Chem Eng Data 50:1624–1630. https://doi.org/10.1021/je050092d
Maximo GJ, Costa MC, Meirelles AJA (2013) Solid-liquid equilibrium of triolein with fatty alcohols. Braz J Chem Eng 30:33–43. https://doi.org/10.1590/S0104-66322013000100005
Maximo GJ, Carareto NDD, Costa MC, dos Santos AO, Cardoso LP, Krähenbühl MA, Meirelles AJA (2014a) On the solid-liquid equilibrium of binary mixtures of fatty alcohols and fatty acids. Fluid Phase Equilib 366:88–98. https://doi.org/10.1016/j.fluid.2014.01.004
Maximo GJ, Costa MC, Meirelles AJA (2014b) The Crystal-T algorithm: a new approach to calculate the SLE of lipidic mixtures presenting solid solutions. Phys Chem Chem Phys 16:16740–16754. https://doi.org/10.1039/c4cp01529k
Maximo GJ, Carareto NDD, Costa MC (2019) Solid-Liquid Equilibrium in Food Processes. In: Thermodynamics of Phase Equilibria in Food Enhineering. 1st edn. Academic Press, Cambridge, p 684
Oh JH, McCurdy AR, Clark S, Swanson BG (2002) Characterization and thermal stability of polymorphic forms of synthesized tristearin. J Food Sci 67:911–2917. https://doi.org/10.1111/j.1365-2621.2002.tb08837.x
Pelaquim FP, Cardoso de Matos F, Cardoso LP, Caldas Batista EA, de Almeida J, Meirelles A, Conceição da Costa M (2019) Solid-liquid phase equilibrium diagrams of binary mixtures containing fatty acids, fatty alcohol compounds and tripalmitin using differential scanning calorimetry. Fluid Phase Equilib 497:19–32. https://doi.org/10.1016/j.fluid.2019.05.020
Pernetti M, van Malssen KF, Flöter E, Bot A (2007) Structuring of edible oils by alternatives to crystalline fat. Curr Opin Colloid Interface Sci 12:221–231. https://doi.org/10.1016/j.cocis.2007.07.002
Persson M (2008) Alpha stable fats. Lipid Technol 20:13–16. https://doi.org/10.1002/lite.200700099
Prausnitz JM, Lichtenthaler RN, de Azevedo EG (1999) Molecular thermodynamics of fluid-phase equilibria. 3rd edn, Nopardazan. Prentice Hall, Nova Jersey. https://doi.org/10.1016/0021-9614(70)90078-9
Singh SK, Jalali AF, Aldén M (1999) Modulated temperature differential scanning calorimetry for examination of tristearin polymorphism: 2. Isothermal Crystallization of Metastable Forms. J Am Oil Chem Soc 76:507–510. https://doi.org/10.1007/s11746-999-0031-5
Tadie M, Bahadur I, Reddy P, Ngema PT, Naidoo P, Deenadayalu N, Ramjugernath D (2013) Solid-liquid equilibria measurements for binary systems comprising (butyric acid + propionic or pentanoic acid) and (heptanoic acid + propionic or butyric or pentanoic or hexanoic acid). J Chem Thermodyn 57:485–492. https://doi.org/10.1016/j.jct.2012.09.019
Timms RE (1984) Phase behaviour of fats and their mixtures. Prog Lipid Res 23:1–38. https://doi.org/10.1016/0163-7827(84)90004-3
Vetterling WT, Teukolsky SA, Press WH, Flannery BP (1992) Numerical Recipes in F77. 2nd edn, Numerical Methods for Fortran 77: the art of scientific computing. 2nd edn. Press Syndicate of the University of Cambridge. http://www.papers3://publication/uuid/42C52282–20B5–4159–9E1B-2E89F2631AAA
Webster GR, Bisset NB, Cahill DM, Jones P, Killick A, Hawley A, Boyd BJ (2018) Tristearin as a model cuticle for high-throughput screening of agricultural adjuvant systems. ACS Omega 3:16672–16680. https://doi.org/10.1021/acsomega.8b02656
Wei D, Han S, Wang B (2014) Solid-liquid phase equilibrium study of binary mixtures of n-octadecane with capric, and lauric acid as phase change materials (PCMs). Fluid Phase Equilib 373:84–88. https://doi.org/10.1016/j.fluid.2014.04.020
Wesdorp LH (1990) Liquid-Multiple solid phase equilibria in fats. Universidade de Tecnologia de Delft
Wright AJ, Marangoni AG (2005) Physical Properties of Fats and Oils. Handbook of Functional Lipids. CRC Press, Florida, pp 135–162
Yui K, Itsukaichi Y, Kobayashi T, Tsuji T, Fukui K, Maeda K, Kuramochi H (2017) Solid-liquid equilibria in the binary systems of saturated fatty acids or triglycerides (C12 to C18) + hexadecane. J Chem Eng Data 62:35–43. https://doi.org/10.1021/acs.jced.6b00355
Zhang Z, Yuan Y, Zhang N, Cao X (2015) Thermophysical Properties of Some Fatty Acids/Surfactants as Phase Change Slurries for Thermal Energy Storage. J Chem Eng Data 60:2495–2501. https://doi.org/10.1021/acs.jced.5b00371
Zhao P, Yue Q, He H, Gao B, Wang Y, Li Q (2014) Study on phase diagram of fatty acids mixtures to determine eutectic temperatures and the corresponding mixing proportions. Appl Energy 115:483–490. https://doi.org/10.1016/j.apenergy.2013.10.048
Funding
The authors thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior–Brazil (CAPES)–Finance Code 001, FAPESP (2014/21252-0 and 2018/19198-9), CNPq (305870/2014-9, 406963/2014-4, 306666/2020-0, 309780/2014-4, and 132149/2017-6), and FAEPEX/UNICAMP for the financial support. The authors also thank Espaço da Escrita–Pró-Reitoria de Pesquisa–UNICAMP–for the language services provided.
Author information
Authors and Affiliations
Contributions
PFP and MFC designed research; CMC, BEAC, MAJA conducted review, editing, and provided funding acquisition, project administration, and resources; PFP wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
43153_2022_227_MOESM1_ESM.docx
Experimental data and Ideal assumption, NRTL and three-suffix Margules UNIFAC model plots to the systems studied: SSS+C10; SSS+C12; SSS+C14; SSS+C10OH; SSS+C12OH; SSS+C14OH (DOCX 301 KB)

SI Figure 1.
Hemolymph (N = 6–12) and urinary (N = 4-12) essential amino acid composition of immersed (0-h) and emersed crabs over a 160-hour time-course exposure. Where data in not available for urinary concentrations no detectable amino acid presence was found. Values are represented as the mean ± S.E.M. (JPG 43 KB)

SI Figure 2.
Hemolymph (N = 6–12) non-essential amino acid composition of immersed (0-h) and emersed crabs over a 160-hour time-course exposure. Where data in not available for urinary concentrations no detectable amino acid presence was found. Values are represented as the mean ± S.E.M. (JPG 80 KB)
Rights and permissions
About this article
Cite this article
Pelaquim, F.P., de Matos, F.C., Batista, E.A.C. et al. Solid–liquid phase equilibrium diagrams of binary mixtures containing fatty acids, fatty alcohol compounds, and tristearin. Braz. J. Chem. Eng. 40, 269–286 (2023). https://doi.org/10.1007/s43153-022-00227-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-022-00227-5