Abstract
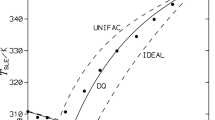
Solid–liquid equilibria (SLE) of binary mixtures of several n-alkanes (n-octadecane, n-eicosane, n-tetracosane, n-pentacosane, n-triacontane) and dibenzofuran covering the whole composition range were measured by differential scanning calorimetry (DSC) in a temperature range of 301–356 K. The dibenzofuran and the n-alkanes are completely miscible in the liquid state but non-miscible in the solid state, and in the context of this work, they seem to exhibit eutectic behavior. A linear trend is obtained for the eutectic temperature and eutectic composition versus the number of carbon atoms of n-alkane. The experimental data were compared to predictions made by using the ideal solution model, the DISQUAC model and several versions of the UNIFAC model, including the classical UNIFAC, the modified versions of Lyngby and Dortmund, and the recently proposed modified UNIFAC (NIST) model, to account for non-ideality in the liquid phase. Moreover, the experimental data were also compared with COSMO-RS predictions that utilize quantum chemical calculations and statistical thermodynamics to interpret the behavior of molecular interactions in the binary mixture. A good agreement was obtained between the predicted and the experimental temperatures.







Similar content being viewed by others
References
Jennings, D.W., Weispfennig, K.: Experimental solubility data of various n-alkane waxes: effects of alkane chain length, alkane odd versus even carbon number structures, and solvent chemistry on solubility. Fluid Phase Equilib. 227, 27–35 (2005). https://doi.org/10.1016/j.fluid.2004.10.021
Burger, E., Perkins, T., Striegler, J.: Studies of wax deposition in the trans Alaska pipeline. J. Petrol. Technol. 33, 1075–1086 (1981). https://doi.org/10.2118/8788-PA
Roenningsen, H.P., Bjoerndal, B., Baltzer Hansen, A., Batsberg Pedersen, W.: Wax precipitation from North Sea crude oils: 1. Crystallization and dissolution temperatures, and Newtonian and non-Newtonian flow properties. Energy Fuels 5, 895–908 (1991). https://doi.org/10.1021/ef00030a019
Coutinho, J.A., Edmonds, B., Moorwood, T., Szczepanski, R., Zhang, X.: Reliable wax predictions for flow assurance. In: European Petroleum Conference, OnePetro, (2002). https://doi.org/10.2118/78324-MS
Coto, B., Martos, C., Espada, J.J., Robustillo, M.D., Peña, J.L.: Analysis of paraffin precipitation from petroleum mixtures by means of DSC: iterative procedure considering solid–liquid equilibrium equations. Fuel 89, 1087–1094 (2010). https://doi.org/10.1016/j.fuel.2009.12.010
Hammami, A., Raines, M.: Paraffin deposition from crude oils: comparison of laboratory results with field data. SPE J. 4, 9–18 (1999). https://doi.org/10.2118/54021-PA
Tuntawiroon, J., Mahidol, C., Navasumrit, P., Autrup, H., Ruchirawat, M.: Increased health risk in Bangkok children exposed to polycyclic aromatic hydrocarbons from traffic-related sources. Carcinogenesis 28, 816–822 (2007). https://doi.org/10.1093/carcin/bgl175
Djordjevic, N.: Solubilities of polycyclic aromatic hydrocarbon solids in n-octadecane. Thermochim. Acta 177, 109–118 (1991). https://doi.org/10.1016/0040-6031(91)80089-2
Aoulmi, A., Bouroukba, M., Solimando, R., Rogalski, M.: Thermodynamics of mixtures formed by polycyclic aromatic hydrocarbons with long chain alkanes. Fluid Phase Equilib. 110, 283–297 (1995). https://doi.org/10.1016/0378-3812(95)02759-8
Mahmoud, R., Solimando, R., Bouroukba, M., Rogalski, M.: Solid−liquid equilibrium and excess enthalpy measurements in binary dibenzofuran or xanthene+normal long-chain alkane systems. J. Chem. Eng. Data 45, 433–436 (2000). https://doi.org/10.1021/je9902084
Hafsaoui, S., Mahmoud, R.: Solid-liquid equilibria of binary systems containing n-tetracosane with naphthalene or dibenzofuran: prediction with UNIFAC model. J. Therm. Anal. Calorim. 88, 565–570 (2007). https://doi.org/10.1007/s10973-006-8084-2
Burley, S., Petsko, G.: Weakly polar interactions in proteins. Adv. Protein Chem. 39, 125–189 (1988). https://doi.org/10.1016/S0065-3233(08)60376-9
Mahmoud, R., Solimando, R., Rogalski, M.: Solid–liquid equilibria of systems containing pyrene and long chain normal-alkanes. Fluid Phase Equilib. 148, 139–146 (1998). https://doi.org/10.1016/S0378-3812(98)00204-0
Boudouh, I., Djemai, I., González, J.A., Barkat, D.: Solid–liquid equilibria of biphenyl binary systems. J. Mol. Liq. 216, 764–770 (2016). https://doi.org/10.1016/j.molliq.2016.02.010
Boudouh, I., Hafsaoui, S.L., Mahmoud, R., Barkat, D.: Measurement and prediction of solid–liquid phase equilibria for systems containing biphenyl in binary solution with long-chain n-alkanes. J. Therm. Anal. Calorim. 125, 793–801 (2016). https://doi.org/10.1007/s10973-016-5407-9
Boudouh, I., Barkat, D., González, J.A., Djemai, I.: Solid-liquid equilibria of indole binary systems. Thermochim. Acta 644, 13–19 (2016). https://doi.org/10.1016/j.tca.2016.10.003
Boudouh, I., González, J.A., Djemai, I., Barkat, D.: Solid-liquid equilibria of eicosane, tetracosane or biphenyl + 1-octadecanol, or + 1-eicosanol mixtures. Fluid Phase Equilib. 442, 28–37 (2017). https://doi.org/10.1016/j.fluid.2017.03.012
Boudouh, I., González, J.A., Coto, B., Moussaoui, A., Kasmi, A., Djemai, I., Hadj-Kali, M.K.: Solid-liquid equilibria for dibenzofuran or Xanthene + heavy hydrocarbons: Experimental measurements and modeling. J. Mol. Liq. 116536, 335 (2021). https://doi.org/10.1016/j.molliq.2021.116536
Boudouh, I., Tamura, K., Djemai, I., Robustillo-Fuentes, M.D., Hadj-Kali, M.K.: Solid-liquid equilibria for biphenyl+ n-tetracosane binary mixtures and n-tetracosane+ dibenzofuran+ biphenyl ternary mixtures: experimental data and prediction with UNIFAC models. Int. J. Thermophys. 43, 1–18 (2022). https://doi.org/10.1007/s10765-022-03025-w
Hansen, H.K., Rasmussen, P., Fredenslund, A., Schiller, M., Gmehling, J.: Vapor–liquid equilibria by UNIFAC group contribution. 5. Revision and extension. Ind. Eng. Chem. Res. 30, 2352–2355 (1991). https://doi.org/10.1021/ie00058a017
Weidlich, U., Gmehling, J.: A modified UNIFAC model. 1. Prediction of VLE, hE, and. \({\gamma^{\infty}}\). Ind. Eng. Chem. Res. 26, 1372–1381 (1987). https://doi.org/10.1021/ie00067a018
Gmehling, J., Li, J., Schiller, M.: A modified UNIFAC model. 2. Present parameter matrix and results for different thermodynamic properties. Ind. Eng. Chem. Res. 32, 178–193 (1993). https://doi.org/10.1021/ie00013a024
Fredenslund, A., Jones, R.L., Prausnitz, J.M.: Group-contribution estimation of activity coefficients in nonideal liquid mixtures. AIChE J. 21, 1086–1099 (1975)
Larsen, B.L., Rasmussen, P., Fredenslund, A.: A modified UNIFAC group-contribution model for prediction of phase equilibria and heats of mixing. Ind. Eng. Chem. Res. 26, 2274–2286 (1987). https://doi.org/10.1021/ie00071a018
Kang, J.W., Diky, V., Frenkel, M.: New modified UNIFAC parameters using critically evaluated phase equilibrium data. Fluid Phase Equilib. 388, 128–141 (2015). https://doi.org/10.1016/j.fluid.2014.12.042
Kehiaian, H.V.: Group contribution methods for liquid mixtures: A critical review. Fluid Phase Equilib. 13, 243–252 (1983). https://doi.org/10.1016/0378-3812(83)80098-3
Klamt, A.: Conductor-like screening model for real solvents: a new approach to the quantitative calculation of solvation phenomena. J. Phys. Chem. 99, 2224–2235 (1995)
Prausnitz, J.M., Lichtenthaler, R.N., De Azevedo, E.G.: Molecular Thermodynamics of Fluid Phase Equilibria. Pearson Education, New Jersey (1998)
Guggenheim, E.A.: Mixtures. Oxford University Press, Oxford (1952)
Bondi, A.A.: Physical properties of Molecular Crystals Liquids, and Glasses. Wiley, New York (1968)
Kehiaian, H.V., Grolier, J.P.E., Benson, G.C.: Thermodynamics of organic mixtures. A generalized quasichemical theory in terms of group surface interactions. J. Chim. Phys. 75, 1031–1048 (1978). https://doi.org/10.1051/jcp/1978751031
González, J., de la Fuente, I.G., Cobos, J., Casanova, C., Ait-Kaci, A.: Application of the zeroth approximation of the DISQUAC model to cyclohexane+ n-alkane mixtures using different combinatorial entropy terms. Fluid Phase Equilib. 112, 63–87 (1995). https://doi.org/10.1016/0378-3812(95)02783-B
González, J.A., de la Fuentá, I.G., Cobos, J.C.: Thermodynamics of mixtures with strongly negative deviations from Raoult’s Law: Part 4. Application of the DISQUAC model to mixtures of 1-alkanols with primary or secondary linear amines. Comparison with Dortmund UNIFAC and ERAS results. Fluid Phase Equilib. 168, 31–58 (2000). https://doi.org/10.1016/S0378-3812(99)00326-X
González, J.A., Zawadzki, M., Domanska, U.: Thermodynamics of mixtures containing polycyclic aromatic hydrocarbons. J. Mol. Liq. 143, 134–140 (2008). https://doi.org/10.1016/j.molliq.2008.07.005
Marongiu, B., Piras, A., Porcedda, S., Tuveri, E.: Excess enthalpies of aromatic ether or aromatic ketone (1)+ n-heptane (2) mixtures DISQUAC analysis. J. Therm. Anal. Calorim. 92, 137–144 (2008). https://doi.org/10.1007/s10973-007-8752-x
Klamt, A., Jonas, V., Bürger, T., Lohrenz, J.C.: Refinement and parametrization of COSMO-RS. J. Phys. Chem. A 102, 5074–5085 (1998)
Klamt, A., Eckert, F.: COSMO-RS: a novel and efficient method for the a priori prediction of thermophysical data of liquids. Fluid Phase Equilib. 172, 43–72 (2000). https://doi.org/10.1016/S0378-3812(00)00357-5
Klamt, A.: COSMO-RS: from Quantum Chemistry to Fluid Phase Thermodynamics and Drug Design. Elsevier, Amsterdam (2005)
Eckert, F., Klamt, A.: Fast solvent screening via quantum chemistry: COSMO-RS approach. AIChE J. 48, 369–385 (2002). https://doi.org/10.1002/aic.690480220
Kniaź, K.: Influence of size and shape effects on the solubility of hydrocarbons: the role of the combinatorial entropy. Fluid Phase Equilib. 68, 35–46 (1991). https://doi.org/10.1016/0378-3812(91)85009-J
Khimeche, K., Boumrah, Y., Benziane, M., Dahmani, A.: Solid–liquid equilibria and purity determination for binary n-alkane + naphthalene systems. Thermochim. Acta 444, 166–172 (2006). https://doi.org/10.1016/j.tca.2006.03.011
Benziane, M., Khimeche, K., Dahmani, A., Nezar, S., Trache, D.: Experimental determination and prediction of (solid+ liquid) phase equilibria for binary mixtures of heavy alkanes and fatty acids. Mol. Phys. 110, 1383–1389 (2012). https://doi.org/10.1007/s10973-012-2654-2
Company, J.C.: Mésure et interprétation des équilibres de cristallisation de solutions de paraffines lourdes et d’hydrocarbures aromatiques. Chem. Eng. Sci. 28, 318–323 (1973). https://doi.org/10.1016/0009-2509(73)85117-6
Mondieig, D., Rajabalee, F., Metivaud, V., Oonk, H., Cuevas-Diarte, M.: n-Alkane binary molecular alloys. Chem. Mater. 16, 786–798 (2004). https://doi.org/10.1021/cm031169p
Acknowledgements
Dr. BOUDOUH wishes to express his gratitude to Pr. Juan Antonio González, Professor at the University of Valladolid, Spain, for having welcomed him to the G.E.T.E.F Laboratory. Dr. Hadj-Kali also expresses his appreciation to the Researchers Supporting Project (RSP2023R361), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Issam Boudouh: Conceptualization, Investigation, Writing – review & editing, Visualization, Validation. Baudilio Coto: Investigation, Writing – review & editing, Visualization, Software, Data curation, Validation, Supervision. Juan Antonio González: Conceptualization, Investigation, Writing – original draft, Visualization, Software, Data curation, Validation, Supervision. Irfan Wazeer: Investigation, Data curation, Writing – review & editing. M. Zulhaziman M. Salleh: Investigation, Writing – review & editing, Visualization, Software, Data curation, Validation. Ismahane Djemai: Writing – review & editing. Maria Dolores Robustillo: Investigation, Data curation, Writing – review & editing. Mohamed K. Hadj-Kali: Investigation, Writing – review & editing, Visualization, Software, Data curation, Validation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boudouh, I., Coto, B., González, J.A. et al. Experimental Data and Modeling of Solid–Liquid Equilibria of Binary Systems Containing Dibenzofuran and Long Chain n-alkanes. J Solution Chem 52, 1083–1106 (2023). https://doi.org/10.1007/s10953-023-01297-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-023-01297-w