Abstract
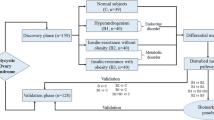
Polycystic ovary syndrome (PCOS) is one of the most common endocrinopathies in reproductive-aged women. The occurrence of PCOS was reported to be associated with the alteration of gut microbiota. Microbiota-derived indoles may possibly play a key role in glycemic control. The purpose of this work is to reveal the alteration of plasma indoles in PCOS patients and to investigate the correlation between indoles levels and glucose metabolism. Sixty-five patients with PCOS and twenty-eight age-matched women were enrolled in this work. The concentrations of plasma indoles, including indoxyl sulfate (IS), indole-3-acetic acid (IAA), indole-3-propionate (IPA), indole (IND), and 3-methylindole (3-MI), were measured by HPLC with the fluorescence detection. The plasma levels of IS, IAA, and IND were significantly elevated in patients with PCOS compared to those in the control group (p < 0.05). Furthermore, the plasma levels of IS, IAA, and IND were positively correlated with fasting glucose, fasting insulin, and the homeostatic model of insulin resistance index (HOMA-IR) (p < 0.05). Besides, the 3-MI level in the plasma was positively correlated with the fasting glucose level, whereas plasma levels of IS, IAA, IND, and 3-MI were negatively correlated with glucagon-like peptide 1 (p < 0.05). Moreover, IS and IND were considered to be risk factors for PCOS after age, BMI, T, LH, and HOMA-IR adjustment. The area under the receiver-operating characteristic curve of the combined index of five indoles was 0.867 for PCOS diagnosis. Additionally, plasma indoles altered in PCOS, which was closely associated with the glucose metabolism.

Similar content being viewed by others
Data Availability
All data generated or analyzed during this study will be provided on request.
Code Availability
Not applicable.
References
Lizneva D, Suturina L, Walker W, et al. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106:6–15.
Muscogiuri G, Barrea L, Caprio M, et al. Nutritional guidelines for the management of insulin resistance. Crit Rev Food Sci Nutr. 2021;2:1–14.
Joham AE, Norman RJ, Stener-Victorin E, et al. Polycystic ovary syndrome. Lancet Diabetes Endocrinol. 2022;10:668–80.
Greenwood EA, Huddleston HG. Insulin resistance in polycystic ovary syndrome: concept versus cutoff. Fertil Steril. 2019;112:827–8.
Joham AE, Teede HJ. PCOS - a metabolic condition with health impacts on women and men. Nat Rev Endocrinol. 2022;18(4):197–8.
Siddiqui S, Mateen S, Ahmad R, et al. A brief insight into the etiology, genetics, and immunology of polycystic ovarian syndrome (PCOS). J Assist Reprod Genet. 2022;39(11):2439–73.
Giampaolino P, Foreste V, Di Filippo C, et al. Microbiome and PCOS: state-of-art and future aspects. Int J Mol Sci. 2021;22(4):2048.
Wang L, Zhou J, Gober HJ, et al. Alterations in the intestinal microbiome associated with PCOS affect the clinical phenotype. Biomed Pharmacother. 2021;133:110958.
Vrieze A, Van Nood E, Holleman F, et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143(913–916):e917.
Pedersen HK, Gudmundsdottir V, Nielsen HB, et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature. 2016;535:376–81.
Agus A, Clément K, Sokol H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut. 2021;70(6):1174–82.
Lee JH, Wood TK, Lee J. Roles of indole as an interspecies and interkingdom signaling molecule. Trends Microbiol. 2015;23(11):707–18.
Koppe L, Pillon NJ, Vella RE, et al. p-Cresyl sulfate promotes insulin resistance associated with CKD. J Am Soc Nephrol. 2013;24(1):88–99.
Roager HM, Licht TR. Microbial tryptophan catabolites in health and disease. Nat Commun. 2018;9(1):3294.
Pernomian L, Duarte-Silva M, de Barros Cardoso CR. The Aryl Hydrocarbon Receptor (AHR) as a potential target for the control of intestinal inflammation: insights from an immune and bacteria Sensor Receptor. Clin Rev Allergy Immunol. 2020;59:382–90.
Abildgaard A, Elfving B, Hokland M, et al. The microbial metabolite indole-3-propionic acid improves glucose metabolism in rats, but does not affect behaviour. Arch Physiol Biochem. 2018;124:306–12.
Kwon Y, Jang M, Lee Y, et al. Metabolomic analysis of the improvements in insulin secretion and resistance after sleeve gastrectomy: implications of the novel biomarkers. Obes Surg. 2021;31:43–52.
Sehgal R, de Mello VD, Männistö V, et al. Indolepropionic acid, a gut bacteria-produced tryptophan metabolite and the risk of type 2 diabetes and non-alcoholic fatty liver disease. Nutrients. 2022;14(21):4695.
Rotterdam EA. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod. 2004;19:41–7.
Calori G, Lattuada G, Piemonti L, et al. Prevalence, metabolic features, and prognosis of metabolically healthy obese Italian individuals: the Cremona study. Diabetes Care. 2011;34:210–5.
Ożegowska K, Plewa S, Mantaj U, et al. Serum metabolomics in PCOS women with different body mass index. J Clin Med. 2021;10(13):2811.
Yang K, Zhou Y, Zhang X, et al. Simultaneous determination of plasma indoles by HPLC with fluorescence detection: application in polycystic ovary syndrome patients with/without depression. Liq Chromatogr R T. 2022;2:124–267.
Liu Z, Hao C, Song D, et al. Androgen receptor coregulator CTBP1-AS is associated with polycystic ovary syndrome in Chinese women: a preliminary study. Reprod Sci. 2015;22(7):829–37.
Diamanti-Kandarakis E, Dunaif A. Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr Rev. 2012;33(6):981–1030.
Ozegowska K, Korman M, Szmyt A, et al. Heterogeneity of endocrinologic and metabolic parameters in reproductive age polycystic ovary syndrome (PCOS) women concerning the severity of hyperandrogenemia-a new insight on syndrome pathogenesis. Int J Environ Res Public Health. 2020;17(24):9291.
Pijls KE, Smolinska A, Jonkers DM, et al. A profile of volatile organic compounds in exhaled air as a potential non-invasive biomarker for liver cirrhosis. Sci Rep. 2016;6:19903.
Lindon J, Nicholson J, Holmes E. The handbook of metabonomics and metabolomics. 1st ed. Elsevier Science; 2007.
Guo J, Shao J, Yang Y, et al. Gut microbiota in patients with polycystic ovary syndrome: a systematic review. Reprod Sci. 2022;29(1):69–83.
France MM, Turner JR. The mucosal barrier at a glance. J Cell Sci. 2017;130:307–14.
Meijers B, Evenepoel P, Anders H-J. Intestinal microbiome and fitness in kidney disease. Nat Rev Nephrol. 2019;15:531–45.
Zhang D, Zhang L, Yue F, et al. Serum zonulin is elevated in women with polycystic ovary syndrome and correlates with insulin resistance and severity of anovulation. Eur J Endocrinol. 2015;172:29–36.
Matsumoto T, Takayanagi K, Kojima M, et al. Acute exposure to indoxyl sulfate impairs endothelium-dependent vasorelaxation in rat aorta. Int J Mol Sci. 2019;20(2):338.
Minakuchi H, Wakino S, Hosoya K, et al. The role of adipose tissue asymmetric dimethylarginine/dimethylarginine dimethylaminohydrolase pathway in adipose tissue phenotype and metabolic abnormalities in subtotally nephrectomized rats. Nephrol Dial Transplant. 2016;31:413–23.
Opdebeeck B, Maudsley S, Azmi A, et al. Indoxyl sulfate and p-cresyl sulfate promote vascular calcification and associate with glucose intolerance. J Am Soc Nephrol. 2019;30:751–66.
Li G, Yin P, Chu S, et al. Correlation analysis between GDM and gut microbial composition in late pregnancy. J Diabetes Res. 2021;2021:8892849.
Woting A, Pfeiffer N, Loh G, et al. Clostridium ramosum promotes high-fat diet-induced obesity in gnotobiotic mouse models. mBio. 2014;5:e01530-01514.
Chen L, Yang Y, Sun S, et al. Indolepropionic acid reduces obesity-induced metabolic dysfunction through colonic barrier restoration mediated via tuft cell-derived IL-25. FEBS J. 2022;289(19):5985–6004.
Jin M, Zhang X, Zhen Q, et al. An electrochemical sensor for indole in plasma based on MWCNTs-chitosan modified screen-printed carbon electrode. Biosens Bioelectron. 2017;98:392–7.
Trajkova M, Molan K, Zugan M, et al. Increased fecal indole concentration in women with gestational diabetes: a pilot study. Acta Diabetol. 2021;58:241–3.
Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8:728–42.
Rajeev SP, Wilding J. GLP-1 as a target for therapeutic intervention. Curr Opin Pharmacol. 2016;31:44–9.
Chimerel C, Emery E, Summers DK, et al. Bacterial metabolite indole modulates incretin secretion from intestinal enteroendocrine L cells. Cell Rep. 2014;9:1202–8.
Lipworth L, Adami HO, Trichopoulos D, et al. Serum steroid hormone levels, sex hormone-binding globulin, and body mass index in the etiology of postmenopausal breast cancer. Epidemiology. 1996;7:96–100.
Corbould A. Chronic testosterone treatment induces selective insulin resistance in subcutaneous adipocytes of women. J Endocrinol. 2007;192:585–94.
Malini NA, Roy GK. Evaluation of different ranges of LH:FSH ratios in polycystic ovarian syndrome (PCOS) - clinical based case control study. Gen Comp Endocrinol. 2018;260:51–7.
Taylor AE, Brian MC, Martin KA, et al. Determinants of abnormal gonadotropin secretion in clinically defined women with polycystic ovary syndrome. J Clin Endocrinol Metab. 1997;7:2248–56.
Sacchi S, D’Ippolito G, Sena P, et al. The anti-Mullerian hormone (AMH) acts as a gatekeeper of ovarian steroidogenesis inhibiting the granulosa cell response to both FSH and LH. J Assist Reprod Genet. 2016;33:95–100.
Capuzzo M, La Marca A. Use of AMH in the differential diagnosis of anovulatory disorders including PCOS. Front Endocrinol (Lausanne). 2020;11:616766.
Acknowledgements
The authors acknowledge all participants for cooperation in this study. We thank the contributions of physicians and other medical personnel in the First Affiliated Hospital of Chongqing Medical University to this study.
Funding
This work was supported by the National Nature Science Foundation of China (31700725), Natural Science Foundation of Chongqing (cstc2019jcyj-msxmX0103, cstc2020jcyj-msxmX0141), and the Science and Technology Research Program of Chongqing Municipal Education Commission (Grant No. KJQN201800437).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
The study was approved by the ethical committee of the First Affiliated Hospital of Chongqing Medical University, China.
Consent to Participate
Informed consents were obtained from all participants.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The first author and co‐first author of this article were Ke Yang and Xiaoqing Zhang.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, K., Zhang, X., Gui, W. et al. Alteration of Plasma Indoles in Polycystic Ovary Syndrome. Reprod. Sci. 31, 764–772 (2024). https://doi.org/10.1007/s43032-023-01377-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01377-8