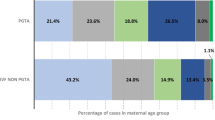
Abstract
In this retrospective study, 120 heterozygous Robertsonian translocation carriers undergoing preimplantation genetic testing (PGT) were included, between January 2018 and September 2021. Meiotic segregation patterns of 462 embryos from 51 female carriers and 69 male carriers were analyzed according to chromosome type, carrier’s sex, and female age. The proportion of alternate embryos in female carriers was slightly lower than that in male carriers [P < 0.001, odds ratio (OR) = 0.512]. By contrast, no difference was observed among Rob (13;14), Rob (14;21), and rare RobT groups. Stratification analysis of female carriers’ age doses showed no significant increase in unbalanced chromosomal abnormalities. Reproductive outcomes of 144 frozen-thawed cycles were analyzed. All 144 blastocysts were transferred, and there were no significant differences in the clinical pregnancy rates per transfer (CPR), miscarriage rates (MR), live birth rates per transfer (LBR), and cumulative live birth rates between female carriers and male carriers. In addition, couples in Rob (13;14), Rob (14;21), and rare RobTs groups had comparative clinical pregnancy rates per transfer (CPR), miscarriage rates (MR), live birth rates per transfer (LBR), and cumulative live birth rates. Our study demonstrated that the meiotic segregation pattern of Robertsonian translocations carriers is associated with the carrier’s sex, but not the carrier’s translocation type and female age. In addition, the sex of translocation carriers only affects the meiotic segregation pattern but does not influence the subsequent viability of normal embryos and live birth.
Similar content being viewed by others
Data Availability
Data available on request from the authors.
References
Nielsen J, Wohlert M. Chromosome abnormalities found among 34,910 newborn children: results from a 13-year incidence study in Arhus. Denmark Hum Genet. 1989;87(1):81–3. https://doi.org/10.1007/BF01213097.
Therman E, Susman B, Denniston C. The nonrandom participation of human acrocentric chromosomes in Robertsonian translocations. Ann Hum Genet. 1989;53(1):49–65. https://doi.org/10.1111/j.1469-1809.1989.tb01121.x.
Anahory T, Hamamah S, Andréo B, Hédon B, Claustres M, Sarda P, et al. Sperm segregation analysis of a (13;22) Robertsonian translocation carrier by FISH: a comparison of locus-specific probe and whole chromosome painting. Hum Reprod. 2005;20(7):1850–4. https://doi.org/10.1093/humrep/deh886.
Anton E, Blanco J, Egozcue J, Vidal F. Sperm FISH studies in seven male carriers of Robertsonian translocation t(13;14)(q10;q10). Hum Reprod. 2004;19(6):1345–51. https://doi.org/10.1093/humrep/deh232.
Chen Y, Huang J, Liu P, Qiao J. Analysis of meiotic segregation patterns and interchromosomal effects in sperm from six males with Robertsonian translocations. J Assist Reprod Genet. 2007;24(9):406–11. https://doi.org/10.1007/s10815-007-9137-6.
Escudero T, Lee M, Carrel D, Blanco J, Munné S. Analysis of chromosome abnormalities in sperm and embryos from two 45, XY, t(13;14)(q10;q10) carriers. PrenatDiagn. 2000;20(7):599–602. https://doi.org/10.1002/1097-0223(200007)20:7%3c599::aid-pd883%3e3.3.co;2-h.
Frydman N, Romana S, Le Lorc’h M, Vekemans M, Frydman R, Tachdjian G. Assisting reproduction of infertile men carrying a Robertsonian translocation. Hum Reprod. 2001;16(11):2274–7. https://doi.org/10.1093/humrep/16.11.2274.
Hatakeyama C, Gao H, Harmer K, Ma S. Meiotic segregation patterns and ICSI pregnancy outcome of a rare (13;21) Robertsonian translocation carrier: a case report. Hum Reprod. 2005;21(4):976–9. https://doi.org/10.1093/humrep/dei427.
Moradkhani K, Puechberty J, Bhatt S, Vago P, Janny L, Lefort G, et al. Meiotic segregation of rare Robertsonian translocations: sperm analysis of three t(14q;22q) cases. Hum Reprod. 2006;21(5):1166–71. https://doi.org/10.1093/humrep/dei477.
Mahjoub M, Mehdi M, Brahem S, Elghezal H, Ibala S, Saad A. Chromosomal segregation in spermatozoa of five Robertsonian translocation carriers t(13;14). J Assist Reprod Genet. 2011;28(7):607–13. https://doi.org/10.1007/s10815-011-9560-6.
Rives N, Ravel C, Duchesne V, Siffroi JP, Mousset-Siméon N. Macé B (2005) Molecular cytogenetics analysis with whole chromosome paint probes of sperm nuclei from a (13;15) Robertsonian translocation carrier 2005. J Hum Genet. 2005;50(7):360–4. https://doi.org/10.1007/s10038-005-0259-3.
Faraut T, Mermet MA, Demongeot J, Cohen O. Cooperation of selection and meiotic mechanisms in the production of imbalances in reciprocal translocations. Cytogenet Cell Genet. 2000;88(1–2):15–21. https://doi.org/10.1159/000015476.
Ford CE, Clegg HM. Reciprocal translocations. Br Med Bull. 1969;25(1):110–4. https://doi.org/10.1093/oxfordjournals.bmb.a070659.
Anton E, Blanco J, Vidal F. Meiotic behavior of three D;G Robertsonian translocations: segregation and interchromosomal effect. J Hum Genet. 2010;55(8):541–5. https://doi.org/10.1038/jhg.2010.67.
Ogur G, Van Assche E, Vegetti W, Verheyen G, Tournaye H, Bonduelle M, et al. Chromosomal segregation in spermatozoa of 14 Robertsonian translocation carriers. Mol Hum Reprod. 2006;12(7):209–15. https://doi.org/10.1093/molehr/gah253.
Shaffer LG. Risk estimates for uniparental disomy following prenatal detection of a nonhomologous Robertsonian translocation. Prenat Diagn. 2006;26(4):303–7. https://doi.org/10.1002/pd.1384.
Lejeune J. Autosomal disorders. Pediatrics. 1963;32:326–37.
Rousseaux S, Chevret E, Monteil M, Cozzi J, Pelletier R, Delafontaine D, et al. Sperm nuclei analysis of a Robertsonian t(14q21q) carrier, by FISH, using three plasmids and two YAC probes. Hum Genet. 1995;96(6):655–60. https://doi.org/10.1007/BF00210294.
Gianaroli L, Magli MC, Ferraretti AP, Munné S, Balicchia B, Escudero T, et al. Possible interchromosomal effect in embryos generated by gametes from translocation carriers. Hum Reprod. 2002;17(12):3201–7. https://doi.org/10.1093/humrep/17.12.3201.
Alfarawati S, Fragouli E, Colls P, Wells D. Embryos of robertsonian translocation carriers exhibit a mitotic interchromosomal effect that enhances genetic instability during early development. PLoS Genet. 2012;8(10):e1003025. https://doi.org/10.1371/journal.pgen.1003025.
Mateu-Brull E, Rodrigo L, Peinado V, Mercader A, Campos-Galindo I, Bronet F, et al. Interchromosomal effect in carriers of translocations and inversions assessed by preimplantation genetic testing for structural rearrangements (PGT-SR). J Assist Reprod Genet. 2019;36(12):2547–55. https://doi.org/10.1007/s10815-019-01593-9.
Zhang S, Lei C, Wu J, Zhou J, Xiao M, Zhu S, et al. Meiotic heterogeneity of trivalent structure and interchromosomal effect in blastocysts with Robertsonian translocations. Front Genet. 2021;12:609563. https://doi.org/10.3389/fgene.2021.609563.
Zhang S, Zhao D, Zhang J, Mao Y, Kong L, Zhang Y, et al. BasePhasing: a highly efficient approach for preimplantation genetic haplotyping in clinical application of balanced translocation carriers. BMC Med Genomics. 2019;12(1):52. https://doi.org/10.1186/s12920-019-0495-6.
Ko DS, Cho JW, Lee HS, Kim JY, Kang IS, Yang KM, et al. Preimplantation g enetic diagnosis outcomes and meiotic segregation analysis of robertsonian translocation carriers. Fertil Steril. 2013;99(5):1369–76. https://doi.org/10.1016/j.fertnstert.2012.12.010.
Zhang L, Jiang W, Zhu Y, Chen H, Yan J, Chen ZJ. Effects of a carrier’s sex and age on the segregation patterns of the trivalent of Robertsonian translocations. J Assist Reprod Genet. 2019;36(9):1963–9. https://doi.org/10.1007/s10815-019-01534-6.
Yuan P, Zheng L, Ou S, Zhao H, Li R, Luo H, et al. Evaluation of chromosomal abnormalities from preimplantation genetic testing to the reproductive outcomes: a comparison between three different structural rearrangements based on next-generation sequencing. J Assist Reprod Genet. 2021;38(3):709–18. https://doi.org/10.1007/s10815-020-02053-5.
Keymolen K, Staessen C, Verpoest W, Michiels A, Bonduelle M, Haentjens P, et al. A proposal for reproductive counselling in carriers of Robertsonian translocations: 10 years of experience with preimplantation genetic diagnosis. Hum Reprod. 2009;24(9):2365–71. https://doi.org/10.1093/humrep/dep201.
Bint SM, Ogilvie CM, Flinter FA, Khalaf Y, Scriven PN. Meiotic segregation of Robertsonian translocations ascertained in cleavage-stage embryos–implications for preimplantation genetic diagnosis. Hum Reprod. 2011;26(6):1575–84. https://doi.org/10.1093/humrep/der080.
Jin H, Ping L, Jie Q, Ying L, Yongjian C. Translocation chromosome karyotypes of the Robertsonian translocation carriers’ embryos. Fertil Steril. 2010;93(4):1061–5. https://doi.org/10.1016/j.fertnstert.2008.11.020.
Miller DE. The interchromosomal effect: different meanings for different organisms. Genetics. 2020;216(3):621–31. https://doi.org/10.1016/j.fertnstert.2008.11.020.
Maithripala Savanie, Durland Ursula, Havelock Jon, Kashyap Sonya, Hitkari Jason, Tan Justin, et al. Prevalence and treatment choices for couples with recurrent pregnancy loss due to structural chromosomal anomalies. J Obstet Gynaecol Can. 2018;40(6):655–62. https://doi.org/10.1016/j.jogc.2017.09.024.
Keymolen K, Van Berkel K, Vorsselmans A, Staessen C, Liebaers I. Pregnancy outcome in carriers of Robertsonian translocations. Am J Med Genet A. 2011;155(10):2381–5. https://doi.org/10.1002/ajmg.a.33941.
Therman E, Susman B, Denniston C. The nonrandom participation of human acrocentric chromosomes in Robertsonian translocations. Ann Hum Genet. 1989;53(1):49–65. https://doi.org/10.1111/j.1469-1809.1989.tb01121.x.
Boue A, Gallano PA. collaborative study of the segregation of inherited chromosome structural rearrangements in 1356 prenatal diagnoses. Prenat Diagn. 1984;4:45–67. https://doi.org/10.1002/pd.1970040705.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the ethics committee of the Northwest Women’s and Children’s Hospital (number 2022007).
Consent to Participate
All patients provided informed consent to participate in the study before their PGT samples were collected.
Consent for Publication
All authors consent to the publication of the paper “Retrospective analysis of meiotic segregation pattern and reproductive outcomes in blastocysts from robertsonian preimplantation genetic testing cycles.” We declare that the submitted manuscript does not contain previously published material and are not under consideration for publication elsewhere. Each author has made an important scientific contribution to the study and is thoroughly familiar with the primary data. All authors listed have read the complete manuscript and have approved the submission of the paper. The manuscript is truthful original work without fabrication, fraud, or plagiarism.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jia, M., Shi, J. & Xue, X. Retrospective Analysis of Meiotic Segregation Pattern and Reproductive Outcomes in Blastocysts from Robertsonian Preimplantation Genetic Testing Cycles. Reprod. Sci. 30, 2983–2989 (2023). https://doi.org/10.1007/s43032-023-01244-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01244-6