Abstract
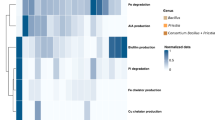
Bacillus subtilis and Rhodopseudomonas palustris are two widely used beneficial microbial inoculums in agriculture. However, whether they have synergistic effects on plant growth and their non-target effects on the root-associated microbial communities is still unknown. In this study, we aimed at understanding the effect of the synergism of two microorganisms on rice root microorganisms and related C, N, and P cycling genes. We used a pot experiment and looked at how the growth of rice plants, soil functions, and the bacterial community in the root zone responded to the single and co-inoculation of B. subtilis and R. palustris. The findings demonstrated that both single and co-inoculation of B. subtilis and R. palustris increased the rice yields and seed setting rates by 0.3%,13.7%, 13.3%, and 17.2%, respectively, and the co-inoculation presented better performance than the single inoculation. Microbial inoculation significantly changed the structure and composition of bacterial communities in the rhizosphere and rhizoplane, and rhizoplane showed more significant changes than those observed in the rhizosphere. Putative functional gene abundances related to carbon, nitrogen, and phosphorus cycling were also enhanced with the inoculation, which were significantly related to the rice seed setting rates, including genes related to arabinofuranosidase, beta-galactosidase, pullulanase, glucoamylase, and chitinase, and nirK, nirA, ureC, nasA, and napA, gdh, phoD, and pqqC (r = 0.58 ~ 0.72; p < 0.05). These genes were possibly carried by Geobacter and Caulobacter. These findings expand the current understanding of the microbial inoculation on the underlying microbial mechanisms in agricultural ecosystem.







Similar content being viewed by others
Data Availability
The raw sequencing reads of the 16S rRNA gene were deposited in the Genome Sequence Archive (publicly accessible at http://bigd.big.ac.cn/gsa) under accession number CRA007857.
References
Ai C, Liang G, Sun J, Wang X, Zhou W (2012) Responses of extracellular enzyme activities and microbial community in both the rhizosphere and bulk soil to long-term fertilization practices in a fluvo-aquic soil. Geoderma 173-174:330–338. https://doi.org/10.1016/j.geoderma.2011.07.020
Arashida H, Kugenuma T, Watanabe M, IJJoB M, Bioengineering (2019) Nitrogen fixation in Rhodopseudomonas palustris co-cultured with Bacillus subtilis in the presence of air. J Biosci Bio 127(5):589–593. https://doi.org/10.1016/j.jbiosc.2018.10.010
Aslam Z, Yasir M, Yoon HS, Jeon CO, Chung YR (2013) Diversity of the bacterial community in the rice rhizosphere managed under conventional and no-tillage practices. J Microbiol 6:747–756. https://doi.org/10.1007/s12275-013-2528-8
Atieno M, Herrmann L, Okalebo R, Lesueur D (2012) Efficiency of different formulations of Bradyrhizobium japonicum and effect of co-inoculation of Bacillus subtilis with two different strains of Bradyrhizobium japonicum. World J Microbiol Biotechnol 7:2541–2550. https://doi.org/10.1007/s11274-012-1062-x
Bashan Y, Holguin G, de-Bashan LE (2004) Azospirillum-plant relationships: physiological, molecular, agricultural, and environmental advances (1997-2003). Can J Microbiol 8:521–577. https://doi.org/10.1139/w04-035
Bei Q, Peng J, Liesack W (2021) Shedding light on the functional role of the Ignavibacteria in Italian rice field soil: a meta-genomic/transcriptomic analysis. Soil Biol Biochem 163:108444. https://doi.org/10.1016/j.soilbio.2021.108444
Bernard KA, Pacheco AL, Burdz T, Wiebe D, Bernier AM (2020) Corynebacterium godavarianum Jani et al. 2018 and Corynebacterium hadale Wei et al. 2018 are both later heterotypic synonyms of Corynebacterium gottingense Atasayar et al. 2017, proposal of an emended description of Corynebacterium gottingense Atasayar et al. 2017. Int J Syst Evol Microbiol 5:3534–3540. https://doi.org/10.1099/ijsem.0.004153
Bharti N, Barnawal D (2019) Amelioration of salinity stress by PGPR: ACC deaminase and ROS scavenging enzymes activity. In: Amit K, Ajay K, Pawan K (eds) PGPR amelioration in sustainable agriculture. Woodhead Publishing, pp 85–106. https://doi.org/10.1016/C2017-0-03466-3
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 8:1621–1624. https://doi.org/10.1038/ismej.2012.8
Chamurliev OG, Borisenko IB, Zelenev AV, Chamurliev GO, Konstantinova TV, Feofilova LA (2022) Improving soil ecology when applying bacterial fertilizers. IOP Conf Ser: Earth Environ Sci 965:012002. https://doi.org/10.1088/1755-1315/965/1/012002
Ding LJ, Cui HL, Nie SA, Long XE, Duan GL, Zhu YG (2019) Microbiomes inhabiting rice roots and rhizosphere. FEMS Microbiol Ecol 95:fiz040. https://doi.org/10.1093/femsec/fiz040
Ge GF, Li ZJ, Zhang J, Wang LG, Xu MG, Zhang JB, Wang JK, Xie XL, Liang YC (2009) Geographical and climatic differences in long-term effect of organic and inorganic amendments on soil enzymatic activities and respiration in field experimental stations of China. Ecol Compl 4:421–431. https://doi.org/10.1016/j.ecocom.2009.02.001
Guo X, Zhang X, Qin Y, Liu YX, Zhang J, Zhang N, Wu K, Qu B, He Z, Wang X, Zhang X, Hacquard S, Fu X, Bai Y (2020) Host-associated quantitative abundance profiling reveals the microbial load variation of root microbiome. Plant Commun 1:100003. https://doi.org/10.1016/j.xplc.2019.100003
Hashem A, Tabassum B, Fathi Abd Allah E (2019) Bacillus subtilis: a plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J Biol Sci 6:1291–1297. https://doi.org/10.1016/j.sjbs.2019.05.004
Hinsinger P, Bengough AG, Vetterlein D, Young IM (2009) Rhizosphere: biophysics, biogeochemistry and ecological relevance. Plant Soil 1-2:117–152. https://doi.org/10.1007/s11104-008-9885-9
Iqbal A, He L, Ali I, Yuan P, Khan A, Hua Z, Wei S, Jiang L (2022) Partial substation of organic fertilizer with chemical fertilizer improves soil biochemical attributes, rice yields, and restores bacterial community diversity in a paddy field. Front Plant Sci 13:895230. https://doi.org/10.3389/fpls.2022.895230
Iturralde ET, Stocco MC, Faura A, Monaco CI, Cordo C, Perez-Gimenez J, Lodeiro AR (2020) Coinoculation of soybean plants with Bradyrhizobium japonicum and Trichoderma harzianum: coexistence of both microbes and relief of nitrate inhibition of nodulation. Biotechnol Rep (Amst) 26:e00461. https://doi.org/10.1016/j.btre.2020.e00461
Jiang M, He L, Fan B, Wang T, Yang N, Liu Y, Xu Y, Dong K, Hao G, Chen L, Ren A, Zhao N, Wang J, Gao Y (2022) Intraspecific more than interspecific diversity plays an important role on Inner Mongolia grassland ecosystem functions: a microcosm experiment. Sci Total Environ 826:154134. https://doi.org/10.1016/j.scitotenv.2022.154134
Kallow S, Garcia Zuluaga M, Fanega Sleziak N, Nugraha B, Mertens A, Janssens SB, Gueco L, Valle-Descalsota ML, Dang Vu T, Toan Vu D, Thi Li L, Vandelook F, Dickie JB, Verboven P, Swennen R, Panis B (2022) Drying banana seeds for ex situ conservation. Conserv Physiol 1:coab099. https://doi.org/10.1093/conphys/coab099
Khuong NQ, Kantachote D, Onthong J, Xuan LNT, Sukhoom A (2018) Enhancement of rice growth and yield in actual acid sulfate soils by potent acid-resistant Rhodopseudomonas palustris strains for producing safe rice. Plant Soil 1-2:483–501. https://doi.org/10.1007/s11104-018-3705-7
Kumar M, Singh DP, Prabha R, Rai AK, Sharma L (2016) Role of microbial inoculants in nutrient use efficiency. Microbial Inocul Sustain Agric Produc 2:133–142
Langille MG, Zaneveld J, Caporaso JG, McDonald D, Knights D, Reyes JA, Clemente JC, Burkepile DE, Vega Thurber RL, Knight R, Beiko RG, Huttenhower C (2013) Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol 9:814–821. https://doi.org/10.1038/nbt.2676
Lu RK (1999) Analytical methods of soil and agricultural chemistry. China Agricultural Science and Technology Press, Beijing
Luo G, Xue C, Jiang Q, Xiao Y, Zhang F, Guo S, Shen Q, Ling N (2020) Soil carbon, nitrogen, and phosphorus cycling microbial populations and their resistance to global change depend on soil C:N:P stoichiometry. mSystems 5:3. https://doi.org/10.1128/mSystems.00162-20
Mahanty T, Bhattacharjee S, Goswami M, Bhattacharyya P, Das B, Ghosh A, Tribedi P (2017) Biofertilizers: a potential approach for sustainable agriculture development. Environ Sci Pollut Res Int 4:3315–3335. https://doi.org/10.1007/s11356-016-8104-0
Mawarda PC, Le Roux X, Dirk van Elsas J, Salles JF (2020) Deliberate introduction of invisible invaders: a critical appraisal of the impact of microbial inoculants on soil microbial communities. Soil Biol Biochem 148:107874. https://doi.org/10.1016/j.soilbio.2020.107874
Mickan BS, Abbott LK, Fan J, Hart MM, Siddique KHM, Solaiman ZM, Jenkins SN (2017) Application of compost and clay under water-stressed conditions influences functional diversity of rhizosphere bacteria. Biol Fert Soils 1:55–70. https://doi.org/10.1007/s00374-017-1238-5
Mohamed EAH, Farag AG, Youssef SA (2018) Phosphate Solubilization by <i> Bacillus subtilis </i> and <i> Serratia marcescens </i> isolated from tomato plant rhizosphere. J Environ Protec 3:266–277. https://doi.org/10.4236/jep.2018.93018
Nosheen S, Ajmal I, Song Y (2021) Microbes as biofertilizers, a potential approach for sustainable crop production. Sustainability 13(4):1868. https://doi.org/10.3390/su13041868
Ortiz A, Sansinenea E (2022) The role of beneficial microorganisms in soil quality and plant health. Sustainability 14(9):5358. https://doi.org/10.3390/su14095358
Radhakrishnan R, Hashem A, Abd Allah EF (2017) Bacillus: a biological tool for crop improvement through bio-molecular changes in adverse environments. Front Physiol 8:667. https://doi.org/10.3389/fphys.2017.00667
Roesti D, Gaur R, Johri B, Imfeld G, Sharma S, Kawaljeet K, Aragno M (2006) Plant growth stage, fertiliser management and bio-inoculation of arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria affect the rhizobacterial community structure in rain-fed wheat fields. Soil Biol Biochem 5:1111–1120. https://doi.org/10.1016/j.soilbio.2005.09.010
Santos-Medellin C, Liechty Z, Edwards J, Nguyen B, Huang B, Weimer BC, Sundaresan V (2021) Prolonged drought imparts lasting compositional changes to the rice root microbiome. Nat Plants 8:1065–1077. https://doi.org/10.1038/s41477-021-00967-1
Schwieger F, Tebbe CC (2000) Effect of field inoculation with Sinorhizobium meliloti L33 on the composition of bacterial communities in rhizospheres of a target plant (Medicago sativa) and a non-target plant (Chenopodium album)-linking of 16S rRNA gene-based single-strand conformation polymorphism community profiles to the diversity of cultivated bacteria. Appl Environ Microbiol 8:3556–3565. https://doi.org/10.1128/AEM.66.8.3556-3565.2000
Shen M, Li J, Dong Y, Zhang Z, Zhao Y, Li Q, Dang K, Peng J, Liu H (2021) The effects of microbial inoculants on bacterial communities of the rhizosphere soil of maize. Agric 11(5):389. https://doi.org/10.3390/agriculture11050389
Tang W, Li Z, Li C, Yu X, Wang F, Wan X, Wang Y, Ma L (2016) High-level expression and characterization of the Bacillus subtilis subsp. subtilis str. BSP1 YwaD aminopeptidase in Pichia pastoris. Protein Expr Purif 122:23–30. https://doi.org/10.1016/j.pep.2016.02.009
van der Bom F, Nunes I, Raymond NS, Hansen V, Bonnichsen L, Magid J, Nybroe O, Jensen LS (2018) Long-term fertilisation form, level and duration affect the diversity, structure and functioning of soil microbial communities in the field. Soil Biol Biochem 122:91–103. https://doi.org/10.1016/j.soilbio.2018.04.003
Xu J, Feng Y, Wang Y, Lin X (2018) Effect of rhizobacterium Rhodopseudomonas palustris inoculation on Stevia rebaudiana plant growth and soil microbial community. Pedosphere 5:793–803. https://doi.org/10.1016/s1002-0160(18)60043-8
Xu J, Feng Y, Wang Y, Luo X, Tang J, Lin X (2015) The foliar spray of Rhodopseudomonas palustris grown under Stevia residue extract promotes plant growth via changing soil microbial community. J Soils Sedi 3:916–923. https://doi.org/10.1007/s11368-015-1269-1
Ye T, Li Y, Zhang J, Hou W, Zhou W, Lu J, Xing Y, Li X (2019) Nitrogen, phosphorus, and potassium fertilization affects the flowering time of rice (Oryza sativa L.). Glo Ecol Conser 20:e00753. https://doi.org/10.1016/j.gecco.2019.e00753
Zhang Y, Xu J, Riera N, Jin T, Li J, Wang N (2017) Huanglongbing impairs the rhizosphere-to-rhizoplane enrichment process of the citrus root-associated microbiome. Microbiome 1:97. https://doi.org/10.1186/s40168-017-0304-4
Zhao Y, Sun Y, Pei M, Fu J, Ji H, Zhao L, Xiao X (2021) Enhanced rice yields are related to pronounced shifts in soil resident bacterial community structures in response to Rhodopseudomonas palustris and Bacillus subtilis inoculation. J Soils Sedi 6:2369–2380. https://doi.org/10.1007/s11368-021-02929-8
Zhong Y, Yang Y, Liu P, Xu R, Rensing C, Fu X, Liao H (2019) Genotype and rhizobium inoculation modulate the assembly of soybean rhizobacterial communities. Plant Cell Environ 6:2028–2044. https://doi.org/10.1111/pce.13519
Funding
This research work was supported by the National Natural Scientific Foundation of China (No. 42007027) and the Postgraduate Research & Practice Innovation Program of Jiangsu Province (SJCX211264, SJCX221387).
Author information
Authors and Affiliations
Contributions
Xian Xiao and Yuan Zhao designed the experiments, Yifeng Gui executed the experiments, Yifeng Gui and Caihong Gu analyzed the data, and Yifeng Gui, Yuexiang Gao, and Caihong Gu wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 896 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gui, Y., Gu, C., Xiao, X. et al. Microbial Inoculations Promoted the Rice Plant Growth by Regulating the Root-Zone Bacterial Community Composition and Potential Function. J Soil Sci Plant Nutr 23, 5222–5232 (2023). https://doi.org/10.1007/s42729-023-01394-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-023-01394-1