Abstract
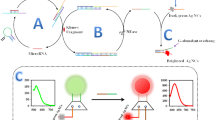
We describe here a target recycling transcription of lighting-up aptamer strategy for detecting ATP in human serums in a label-free means with high sensitivity. ATP molecules specifically recognize the binding aptamer and result in the structure switching of the DNA assembly probes to imitate the target ATP molecule recycling cycles through the toehold-mediated strand displacement reaction, which causes the formation of many dsDNAs containing the RNA promoter sequences for subsequent transcription generation of large amounts of lighting-up aptamers. The organic dye, malachite green, then associates with these lighting-up aptamers to produce significantly enhanced fluorescence signals, which can sensitively detect ATP within a dynamic range from 10 to 500 nM in a label-free way. The sensing approach shows a detection limit of 7.3 nM and also has an excellent selectivity for ATP analogue molecules. In addition, this method can detect ATP molecules in diluted human serum samples sensitively, which proves the promising potential to diagnose ATP-related diseases.





Similar content being viewed by others
References
Wang J, Jiang YX, Zhou CS, Fang XH. Aptamer-based ATP assay using a luminescent light switching complex. Anal Chem. 2005;77:3542–6.
Kucherenko IS, Didukh DY, Soldatkin OO, Soldatkin AP. Amperometric biosensor system for simultaneous determination of adenosine-5’-triphosphate and glucose. Anal Chem. 2014;86:5455–62.
Wang Y, Li Z, Hu D, Lin CT, Li J, Lin Y. Aptamer/graphene oxide nanocomplex for in situ molecular probing in living cells. J Am Chem Soc. 2010;132(27):9274–6.
Huang DW, Niu CG, Zeng GM, Ruan M. Time-resolved fluorescence biosensor for adenosine detection based on home-made europium complexes. Biosens Bioelectron. 2011;29:178–83.
Wang P, Cheng ZY, Chen Q, Qu LL, Miao XM, Feng QM. Construction of a paper-based electrochemical biosensing platform for rapid and accurate detection of adenosine triphosphate (ATP). Sens Actuators B. 2018;256:931–7.
Zhang P, Zhu MS, Luo H, Zhang Q, Guo LE, Li Z, Jiang YB. Aggregationswitching strategy for promoting fluorescent sensing of biologically relevant species: a simple near-infrared cyanine dye highly sensitive and selective for ATP. Anal Chem. 2017;89:6210–5.
Wang Y, Tang L, Li Z, Lin Y, Li J. In situ simultaneous monitoring of ATP and GTP using a graphene oxide nanosheet-based sensing platform in living cell. Nat Protoc. 2014;9(8):1944–55.
Llaudet E, Hatz S, Droniou M, Dale N. Microelectrode biosensor for real-time measurement of ATP in biological tissue. Anal Chem. 2005;77(10):3267.
He HZ, Ma VPY, Leung KH, Chan DSH, Yang H, Cheng Z, Leung CH, Ma DL. A label-free G-quadruplex-based switch-on fluorescence assay for the selective detection of ATP. Analyst. 2012;137(7):1538–40.
Wang K, Liao J, Yang X, Zhao M, Chen M, Yao W, Tan W, Lan X. A label-free aptasensor for highly sensitive detection of ATP and thrombin based on metal-enhanced PicoGreen fluorescence. Biosens Bioelectron. 2015;63:172–7.
Liu Z, Zhong Y, Hu Y, Yuan L, Luo R, Chen D, Wu M, Huang H, Li Y. Fluorescence strategy for sensitive detection of adenosine triphosphate in terms of evaluating meat freshness. Food Chem. 2019;270:573–8.
Liang A, Ouyang H, Jiang Z. Resonance scattering spectral detection of trace ATP based on label-free aptamer reaction and nanogold catalysis. Analyst. 2011;136:4514–9.
Scian M, Acchione M, Li M, Atkins WM. Reaction dynamics of ATP hydrolysis catalyzed by P-glycoprotein. Biochemistry. 2014;53:991–1000.
Yangyuoru PM, Dhakal S, Yu Z, Koirala D, Mwongela SM, Mao H. Singlemolecule measurements of the binding between small molecules and DNA aptamers. Anal Chem. 2012;84:5298–303.
Kim SU, Jo EJ, Noh Y, Mun H, Ahn YD, Kim MG. Adenosine triphosphate bioluminescence-based bacteria detection using targeted photothermal lysis by gold nanorods. Anal Chem. 2018;90:10171–8.
Khlyntseva SV, Vishnikin AB, Andruch V. Methods for the determination of adenosine triphosphate and other adenine nucleotides. J Anal Chem. 2009;64:657–73.
Wang DD, Qi GH, Zhou Y, Zhang Y, Wang B, Hu P, Jin YD. Single-cell ATP detection and content analyses in electrostimulus-induced apoptosis using functionalized glass nanopipettes. Chem Comm. 2020;56:1561–4.
Cancel MS, Simpson LW, Leach JB, White RJ. Direct, real-time detection of adenosine triphosphate release from astrocytes in three-dimensional culture using an integrated electrochemical aptamer-based sensor. ACS Chem Neurosci. 2019;10:2070–9.
Feng G, Zhang BY, Wang SC, Ding ZY, Li G, Zhang XJ. Ratiometric ATP detection on gliding microtubules based on bioorthogonal fluorescence conjugation. Sens Actuators B Chem. 2019;301:127090.
Zhou XM, Li JM, Tan LL, Li Q, Shang L. Novel perylene probe-encapsulated metal–organic framework nanocomposites for ratiometric fluorescence detection of ATP. J Mater Chem B. 2020;8:3661–6.
Danesh NM, Ramezani M, Emrani AS, Abnous K, Taghdisi SM. A novel electrochemical aptasensor based on arch-shape structure of aptamer-complimentary strand conjugate and exonuclease I for sensitive detection of streptomycin. Biosens Bioelectron. 2016;75:123–8.
Gong X, Li JF, Zhou WJ, Xiang Y, Yuan R, Chai YQ. Target recycling amplification for label-free and sensitive colorimetric detection of adenosine triphosphate based on un-modified aptamers and DNAzymes. Anal Chim Acta. 2014;828:80–4.
Liu M, Song JP, Shuang SM, Dong C, Brennan JD, Li YF. A graphene-based biosensing platform based on the release of DNA probes and rolling circle amplification. ACS Nano. 2014;8:5564–73.
Liu ZB, Chen SS, Liu BW, Wu JP, Zhou YB, He LY, Ding JS, Liu JW. Intracellular detection of ATP using an aptamer beacon covalently linked to graphene oxide resisting nonspecific probe displacement. Anal Chem. 2014;86:12229–35.
Munzar JD, Ng A, Juncker D. Duplexed aptamers: history, design, theory, and application to biosensing. Chem Soc Rev. 2019;48:1390–419.
Paige JS, Wu KY, Jaffrey SR. RNA mimics of green fluorescent protein. Science. 2011;333:642–6.
Victoria G, Geneva L, Seth A, Emil FK. Fluorogenic RNA aptamers: a nano-platform for fabrication of simple and combinatorial logic gates. Nanomaterials. 2018;8:984.
Li DX, Yang F, Yuan R, Xiang Y. Lighting-up RNA aptamer transcription synchronization amplification for ultrasensitive and label-free imaging of microRNA in single cells. Anal Chim Acta. 2020;1102:84–90.
Pribnow D. Nucleotide sequence of an RNA polymerase binding site at an early T7 promoter. Proc Natl Acad Sci U S A. 1975;72:784–8.
Jiao K, Zhu B, Guo L, Zhou H, Wang F, Zhang X, Shi J, Li Q, Wang L, Li J, Fan C. Programming switchable transcription of topologically constrained DNA. J Am Chem Soc. 2020;142:10739–46.
Genot AJ, Zhang DY, Bath J, Turberfield AJ. Remote toehold: a mechanism for flexible control of DNA hybridization kinetics. J Am Chem Soc. 2011;133:2177–82.
Hu TX, Wen W, Zhang XH, Wang SF. Nicking endonuclease-assisted recycling of target–aptamer complex for sensitive electrochemical detection of adenosine triphosphate. Analyst. 2016;141:1506–11.
Liu Y, Lei J, Huang Y, Ju H. “Off-on” electrochemiluminescence system for sensitive detection of ATP via target-induced structure switching. Anal Chem. 2014;86:8735.
Song QW, Wang RH, Sun FF, Chen HK, Wang ZMK, Na N, Ouyang J. A nuclease-assisted label-free aptasensor for fluorescence turn-on detection of ATP based on the in situ formation of copper nanoparticles. Biosens Bioelectron. 2017;87:760–3.
Li X, Peng Y, Chai YQ, Yuan R, Xiang Y. A target responsive aptamer machine for label-free and sensitive non-enzymatic recycling amplification detection of ATP. Chem Commun. 2016;52:3673–6.
Wei YL, Chen YX, Li HH, Shuang SM, Dong C, Wang GF. An exonuclease I- based label-free fluorometricaptasensor for adenosine triphosphate (ATP) detection with a wide concentration range. Biosens Bioelectron. 2015;63:311–6.
Acknowledgements
This work was supported by National Natural Science Foundation of China (22004010), the Chongqing Science and Technology Commission of China (cstc2019jcyj-msxmX0196), the Science and Technology Research Program of Chongqing Municipal Education Commission (KJQN201901135) and the Scientific Research Foundation of Chongqing University of Technology (W. Zhou).
Author information
Authors and Affiliations
Corresponding authors
About this article
Cite this article
He, J.L., Jiang, B.Y., Zhou, W.J. et al. Target Recycling Transcription of Lighting-Up RNA Aptamers for Highly Sensitive and Label-Free Detection of ATP. J. Anal. Test. 5, 174–180 (2021). https://doi.org/10.1007/s41664-021-00170-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41664-021-00170-3