Abstract
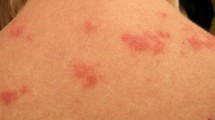
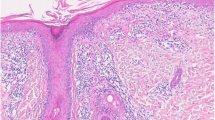
Though more common earlier in life, increasing attention is being focused on the development of cutaneous lupus erythematosus (CLE) in patients with advancing age. Studies show that CLE is more common in older populations than previously thought, and all CLE subtypes are possible in this group. Just like patients in the third or fourth decade of life, CLE may appear alongside or independent of systemic lupus erythematosus. Older populations manifesting CLE for the first time seem to have a lower risk of progression to systemic disease than younger peers, and are more commonly White. CLE must be carefully distinguished from other skin conditions that have a predilection for presentation in older populations, including rosacea, lichen planus, and other autoimmune conditions such as dermatomyositis or pemphigus/pemphigoid. It is thought that most CLE in older populations is drug-induced, with drug-induced subacute cutaneous lupus erythematosus being the most common subtype. Management of CLE in older patients focuses on eliminating unnecessary medications known to induce CLE, and otherwise treatment proceeds similarly to that in younger patients, with a few special considerations.



Similar content being viewed by others
References
Lu Q, et al. Guideline for the diagnosis, treatment and long-term management of cutaneous lupus erythematosus. J Autoimmun. 2021;123: 102707. https://doi.org/10.1016/j.jaut.2021.102707.
Filotico R, Mastrandrea V. Cutaneous lupus erythematosus: clinico-pathologic correlation. Ital J Dermatol Venereol. 2018;153(2):Art. No. 2. https://doi.org/10.23736/S0392-0488.18.05929-1.
Chang AY, Werth VP. Treatment of cutaneous lupus. Curr Rheumatol Rep. 2011;13(4):300–7. https://doi.org/10.1007/s11926-011-0180-z.
Okon LG, Werth VP. Cutaneous lupus erythematosus: Diagnosis and treatment. Best Pract Res Clin Rheumatol. 2013;27(3):391–404. https://doi.org/10.1016/j.berh.2013.07.008.
Renner R, Sticherling M. The different faces of cutaneous lupus erythematosus. G Ital Dermatol E Venereol Organo Uff Soc Ital Dermatol E Sifilogr. 2009;144(2):135–47.
Cohen MR, Crosby D. Systemic disease in subacute cutaneous lupus erythematosus: a controlled comparison with systemic lupus erythematosus. J Rheumatol. 1994;21(9):1665–9.
Sassi RH, et al. Age of onset influences on clinical and laboratory profile of patients with systemic lupus erythematosus. Clin Rheumatol. 2017;36(1):89–95. https://doi.org/10.1007/s10067-016-3478-4.
Jarukitsopa S, et al. Epidemiology of Systemic Lupus Erythematosus and Cutaneous Lupus Erythematosus in a Predominantly White Population in the United States: Incidence and Prevalence of SLE and CLE in a US Population. Arthritis Care Res. 2015;67(6):817–28. https://doi.org/10.1002/acr.22502.
Pons-Estel GJ, Ugarte-Gil MF, Alarcón GS. Epidemiology of systemic lupus erythematosus. Expert Rev Clin Immunol. 2017;13(8):799–814. https://doi.org/10.1080/1744666X.2017.1327352.
Chakravarty EF, Bush TM, Manzi S, Clarke AE, Ward MM. Prevalence of adult systemic lupus erythematosus in California and Pennsylvania in 2000: estimates obtained using hospitalization data. Arthritis Rheum. 2007;56(6):2092–4. https://doi.org/10.1002/art.22641.
Lahita RG. The role of sex hormones in systemic lupus erythematosus. Curr Opin Rheumatol. 1999;11(5):352–6. https://doi.org/10.1097/00002281-199909000-00005.
Simard JF, Costenbader KH. What can epidemiology tell us about systemic lupus erythematosus? Int J Clin Pract. 2007;61(7):1170–80. https://doi.org/10.1111/j.1742-1241.2007.01434.x.
Cozier YC, Barbhaiya M, Castro-Webb N, Costenbader KH, Rosenberg L. A prospective study of reproductive factors in relation to risk of systemic lupus erythematosus among black women. Lupus. 2021;30(2):204–10. https://doi.org/10.1177/0961203320973074.
Avilés Izquierdo JA, Cano Martínez N, Lázaro Ochaita P. Epidemiological characteristics of patients with cutaneous lupus erythematosus. Actas Dermosifiliogr. 2014;105(1):69–73. https://doi.org/10.1016/j.ad.2013.09.005.
Jarrett P, Werth VP. A review of cutaneous lupus erythematosus: improving outcomes with a multidisciplinary approach. J Multidiscip Healthc. 2019;12:419–28. https://doi.org/10.2147/JMDH.S179623.
Grönhagen CM, Fored CM, Linder M, Granath F, Nyberg F. Subacute cutaneous lupus erythematosus and its association with drugs: a population-based matched case-control study of 234 patients in Sweden. Br J Dermatol. 2012;167(2):296–305. https://doi.org/10.1111/j.1365-2133.2012.10969.x.
Chanprapaph K, Tubtieng I, Pratumchat N, Thadanipon K, Rattanakaemakorn P, Suchonwanit P. Cutaneous, systemic features and laboratory characteristics of late- versus adult-onset systemic lupus erythematosus in 1006 Thai patients. Lupus. 2021;30(5):785–94. https://doi.org/10.1177/0961203321991920.
Tomic-Lucic A, et al. Late-onset systemic lupus erythematosus: clinical features, course, and prognosis. Clin Rheumatol. 2013;32(7):1053–8. https://doi.org/10.1007/s10067-013-2238-y.
Anderson SR, Hynan LS, Chong BF. Late-onset cutaneous lupus erythematosus patients have distinctive clinical features and demographics versus early-onset patients. Lupus. 2022;31(12):1523–8. https://doi.org/10.1177/09612033221122254.
Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of ‘Lupus’: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509. https://doi.org/10.1155/2021/6610509.
Litt JZ. Rosacea: how to recognize and treat an age-related skin disease. Geriatrics. 1997;52(11):39–40 (42, 45–47).
Wollina U. Rosacea and rhinophyma in the elderly. Clin Dermatol. 2011;29(1):61–8. https://doi.org/10.1016/j.clindermatol.2010.07.009.
Van Onselen J. Rosacea: symptoms and support. Br J Nurs Mark Allen Publ. 2012;21(21):1252–5. https://doi.org/10.12968/bjon.2012.21.21.1252.
Sowell J, Pena SM, Elewski BE. Seborrheic dermatitis in older adults: pathogenesis and treatment options. Drugs Aging. 2022;39(5):315–21. https://doi.org/10.1007/s40266-022-00930-5.
Sanders MGH, Pardo LM, Franco OH, Ginger RS, Nijsten T. Prevalence and determinants of seborrhoeic dermatitis in a middle-aged and elderly population: the Rotterdam Study. Br J Dermatol. 2018;178(1):148–53. https://doi.org/10.1111/bjd.15908.
Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24(5):348–62. https://doi.org/10.1016/j.clindermatol.2006.07.014.
Magro CM, Crowson AN. The immunofluorescent profile of dermatomyositis: a comparative study with lupus erythematosus. J Cutan Pathol. 1997;24(9):543–52. https://doi.org/10.1111/j.1600-0560.1997.tb01458.x.
Dressler F, Maurer B. Dermatomyositis and juvenile dermatomyositis. Z Rheumatol. 2023;82(3):233–45. https://doi.org/10.1007/s00393-022-01205-5.
Walling HW, Sontheimer RD. Cutaneous lupus erythematosus: issues in diagnosis and treatment. Am J Clin Dermatol. 2009;10(6):365–81. https://doi.org/10.2165/11310780-000000000-00000.
Chi AC, Neville BW, Krayer JW, Gonsalves WC. Oral manifestations of systemic disease. Am Fam Physician. 2010;82(11):1381–8.
Fabbri P, Cardinali C, Giomi B, Caproni M. Cutaneous lupus erythematosus: diagnosis and management. Am J Clin Dermatol. 2003;4(7):449–65. https://doi.org/10.2165/00128071-200304070-00002.
Furukawa F, Muto M. Ethnic differences in immunogenetic features and photosensitivity of cutaneous lupus erythematosus. Arch Dermatol Res. 2009;301(1):111–5. https://doi.org/10.1007/s00403-008-0897-3.
Jore S, Gran JT. Subacute cutaneous lupus erythematosus. Tidsskr Den Nor Laegeforening Tidsskr Prakt Med Ny Raekke. 1997;117(14):2040–2.
Laurinaviciene R, Sandholdt LH, Bygum A. Drug-induced cutaneous lupus erythematosus: 88 new cases. Eur J Dermatol. 2017;27(1):28–33. https://doi.org/10.1684/ejd.2016.2912.
Werth VP. Clinical manifestations of cutaneous lupus erythematosus. Autoimmun Rev. 2005;4(5):296–302. https://doi.org/10.1016/j.autrev.2005.01.003.
Stull C, Sprow G, Werth VP. Cutaneous involvement in systemic lupus erythematosus: a review for the rheumatologist. J Rheumatol. 2023;50(1):27–35. https://doi.org/10.3899/jrheum.220089.
Yell JA, Mbuagbaw J, Burge SM. Cutaneous manifestations of systemic lupus erythematosus. Br J Dermatol. 1996;135(3):355–62.
Chlebus E, Wolska H, Blaszczyk M, Jablonska S. Subacute cutaneous lupus erythematosus versus systemic lupus erythematosus: diagnostic criteria and therapeutic implications. J Am Acad Dermatol. 1998;38(3):405–12. https://doi.org/10.1016/s0190-9622(98)70497-9.
de Berker D, Dissaneyeka M, Burge S. The sequelae of chronic cutaneous lupus erythematosus. Lupus. 1992;1(3):181–6. https://doi.org/10.1177/096120339200100310.
D. Saleh, H. Grubbs, T. Koritala, and J. S. Crane, “Tumid Lupus Erythematosus,” in StatPearls, Treasure Island (FL): StatPearls Publishing, 2023. Accessed 21 May 2023. [Online]. Available: http://www.ncbi.nlm.nih.gov/books/NBK482515/
Joseph AK, Windsor B, Hynan LS, Chong BF. Discoid lupus erythematosus skin lesion distribution and characteristics in Black patients: a retrospective cohort study. Lupus Sci Med. 2021;8(1): e000514. https://doi.org/10.1136/lupus-2021-000514.
McDaniel B, Sukumaran S, Koritala T, Tanner LS. Discoid Lupus Erythematosus. In: StatPearls, Treasure Island (FL): StatPearls Publishing, 2023. http://www.ncbi.nlm.nih.gov/books/NBK493145/. Accessed 21 May 2023.
Chong BF, Song J, Olsen NJ. Determining risk factors for developing systemic lupus erythematosus in patients with discoid lupus erythematosus. Br J Dermatol. 2012;166(1):29–35. https://doi.org/10.1111/j.1365-2133.2011.10610.x.
Bertoli AM, et al. Systemic lupus erythematosus in a multiethnic US cohort. XXXIII. Clinical [corrected] features, course, and outcome in patients with late-onset disease. Arthritis Rheum. 2006;54(5):1580–7. https://doi.org/10.1002/art.21765.
Medlin JL, Hansen KE, Fitz SR, Bartels CM. A systematic review and meta-analysis of cutaneous manifestations in late- versus early-onset systemic lupus erythematosus. Semin Arthritis Rheum. 2016;45(6):691–7. https://doi.org/10.1016/j.semarthrit.2016.01.004.
Izmirly P, et al. Population-based prevalence and incidence estimates of primary discoid lupus erythematosus from the Manhattan Lupus Surveillance Program. Lupus Sci Med. 2019;6(1): e000344. https://doi.org/10.1136/lupus-2019-000344.
Deligny C, Marie DS, Clyti E, Arfi S, Couppié P. Pure cutaneous lupus erythematosus in a population of African descent in French Guiana: a retrospective population-based description. Lupus. 2012;21(13):1467–71. https://doi.org/10.1177/0961203312458167.
He Y, Sawalha AH. Drug-induced lupus erythematosus: an update on drugs and mechanisms. Curr Opin Rheumatol. 2018;30(5):490–7. https://doi.org/10.1097/BOR.0000000000000522.
Pretel M, Marquès L, España A. Drug-induced lupus erythematosus. Actas Dermosifiliogr. 2014;105(1):18–30. https://doi.org/10.1016/j.ad.2012.09.007.
Borchers AT, Keen CL, Gershwin ME. Drug-induced lupus. Ann N Y Acad Sci. 2007;1108:166–82. https://doi.org/10.1196/annals.1422.019.
Vedove CD, Del Giglio M, Schena D, Girolomoni G. Drug-induced lupus erythematosus. Arch Dermatol Res. 2009;301(1):99–105. https://doi.org/10.1007/s00403-008-0895-5.
Szczęch J, Samotij D, Werth VP, Reich A. Trigger factors of cutaneous lupus erythematosus: a review of current literature. Lupus. 2017;26(8):791–807. https://doi.org/10.1177/0961203317691369.
Noël B. Lupus erythematosus and other autoimmune diseases related to statin therapy: a systematic review. J Eur Acad Dermatol Venereol JEADV. 2007;21(1):17–24. https://doi.org/10.1111/j.1468-3083.2006.01838.x.
Noël B, Panizzon RG. Lupus-like syndrome associated with statin therapy. Dermatol Basel Switz. 2004;208(3):276–7. https://doi.org/10.1159/000077320.
Deng Z, Guo A, Wu C, Wang C. Proton pump inhibitors-related subacute cutaneous lupus erythematosus: clinical characteristics, management, and outcome. J Cosmet Dermatol. 2022;21(12):7202–8. https://doi.org/10.1111/jocd.15450.
Lowe GC, Henderson CL, Grau RH, Hansen CB, Sontheimer RD. A systematic review of drug-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2011;164(3):465–72. https://doi.org/10.1111/j.1365-2133.2010.10110.x.
Marzano AV, Lazzari R, Polloni I, Crosti C, Fabbri P, Cugno M. Drug-induced subacute cutaneous lupus erythematosus: evidence for differences from its idiopathic counterpart. Br J Dermatol. 2011;165(2):335–41. https://doi.org/10.1111/j.1365-2133.2011.10397.x.
Mirali S, Mufti A, Lansang RP, Sachdeva M, Yeung J. Development of chronic cutaneous lupus erythematosus during biologic therapy: A systematic review. J Am Acad Dermatol. 2021;84(3):835–8. https://doi.org/10.1016/j.jaad.2020.09.087.
Brehon A, et al. Discoid drug-induced lupus erythematosus induced by antitumor necrosis factor agents is a very rare subtype of cutaneous lupus: three cases and literature review. Dermatol Ther. 2020;33(3): e13364. https://doi.org/10.1111/dth.13364.
Bataille P, et al. Cutaneous drug-induced lupus erythematosus: clinical and immunological characteristics and update on new associated drugs. Ann Dermatol Vénéréologie. 2021;148(4):211–20. https://doi.org/10.1016/j.annder.2021.02.006.
Subramanian S, Yajnik V, Sands BE, Cullen G, Korzenik JR. Characterization of patients with infliximab-induced lupus erythematosus and outcomes after retreatment with a second anti-TNF agent. Inflamm Bowel Dis. 2011;17(1):99–104. https://doi.org/10.1002/ibd.21370.
Antonov D, Kazandjieva J, Etugov D, Gospodinov D, Tsankov N. Drug-induced lupus erythematosus. Clin Dermatol. 2004;22(2):157–66. https://doi.org/10.1016/j.clindermatol.2003.12.023.
Stratton MA. Drug-induced systemic lupus erythematosus. Clin Pharm. 1985;4(6):657–63.
Goldman AL, Braman SS. Isoniazid: a review with emphasis on adverse effects. Chest. 1972;62(1):71–7. https://doi.org/10.1378/chest.62.1.71.
West SG, McMahon M, Portanova JP. Quinidine-induced lupus erythematosus. Ann Intern Med. 1984;100(6):840–2. https://doi.org/10.7326/0003-4819-100-6-840.
Drory VE, Korczyn AD. Hypersensitivity vasculitis and systemic lupus erythematosus induced by anticonvulsants. Clin Neuropharmacol. 1993;16(1):19–29. https://doi.org/10.1097/00002826-199302000-00002.
Sandholdt LH, Laurinaviciene R, Bygum A. Proton pump inhibitor-induced subacute cutaneous lupus erythematosus. Br J Dermatol. 2014;170(2):342–51. https://doi.org/10.1111/bjd.12699.
Zitouni NB, Arnault J-P, Dadban A, Attencourt C, Lok CC, Chaby G. Subacute cutaneous lupus erythematosus induced by nivolumab: two case reports and a literature review. Melanoma Res. 2019;29(2):212–5. https://doi.org/10.1097/CMR.0000000000000536.
Pinard J, Patel M, Granter SR, Vleugels RA, Merola JF. Subacute cutaneous lupus erythematosus induced by palbociclib. J Cutan Med Surg. 2018;22(3):341–3. https://doi.org/10.1177/1203475417752369.
Fumal I, Danchin A, Cosserat F, Barbaud A, Schmutz JL. Subacute cutaneous lupus erythematosus associated with tamoxifen therapy: two cases. Dermatology. 2005;210(3):251–2. https://doi.org/10.1159/000083798.
Andric M, Dixit S, Robaei D, Watchorn R, Verma N. A case of subacute cutaneous lupus erythematosus as a result of ranibizumab (Lucentis) treatment. Indian J Ophthalmol. 2013;61(12):752–4. https://doi.org/10.4103/0301-4738.121133.
Casado-Verrier B, Pérez-Santos S, Delgado-Mucientes C, Beato-Merino M. Subacute cutaneous lupus erythematosus induced by the new multikinase inhibitor pazopanib. Br J Dermatol. 2014;171(6):1559–61. https://doi.org/10.1111/bjd.13175.
Eriksson C, Engstrand S, Sundqvist K-G, Rantapää-Dahlqvist S. Autoantibody formation in patients with rheumatoid arthritis treated with anti-TNF alpha. Ann Rheum Dis. 2005;64(3):403–7. https://doi.org/10.1136/ard.2004.024182.
Klein R, Rosenbach M, Kim EJ, Kim B, Werth VP, Dunham J. Tumor necrosis factor inhibitor-associated dermatomyositis. Arch Dermatol. 2010;146(7):780–4. https://doi.org/10.1001/archdermatol.2010.142.
Ramos-Casals M, et al. Autoimmune diseases induced by TNF-targeted therapies: analysis of 233 cases. Medicine (Baltimore). 2007;86(4):242–51. https://doi.org/10.1097/MD.0b013e3181441a68.
Zattra E, Stan R, Russo I, Lo Nigro A, Peserico A, Alaibac M. TNF blockade and cutaneous lupus erythematosus: where do we stand and where are we going? Immunotherapy. 2013;5(8):791–4. https://doi.org/10.2217/imt.13.82.
Jean G, Souberbielle JC, Chazot C. Vitamin D in chronic kidney disease and dialysis patients. Nutrients. 2017;9(4):328. https://doi.org/10.3390/nu9040328.
Aguado P, et al. Low vitamin D levels in outpatient postmenopausal women from a rheumatology clinic in Madrid, Spain: their relationship with bone mineral density. Osteoporos Int J Establ Result Coop Eur Found Osteoporos Natl Osteoporos Found USA. 2000;11(9):739–44. https://doi.org/10.1007/s001980070052.
Yao P, et al. Vitamin D and calcium for the prevention of fracture: a systematic review and meta-analysis. JAMA Netw Open. 2019;2(12): e1917789. https://doi.org/10.1001/jamanetworkopen.2019.17789.
Feng Y, Cheng G, Wang H, Chen B. The associations between serum 25-hydroxyvitamin D level and the risk of total fracture and hip fracture. Osteoporos Int J Establ Result Coop Eur Found Osteoporos Natl Osteoporos Found USA. 2017;28(5):1641–52. https://doi.org/10.1007/s00198-017-3955-x.
Cutillas-Marco E, Marquina-Vila A, Grant WB, Vilata-Corell JJ, Morales-Suárez-Varela MM. Vitamin D and cutaneous lupus erythematosus: effect of vitamin D replacement on disease severity. Lupus. 2014;23(7):615–23. https://doi.org/10.1177/0961203314522338.
Yosipovitch G, Tang MBY. Practical management of psoriasis in the elderly: epidemiology, clinical aspects, quality of life, patient education and treatment options. Drugs Aging. 2002;19(11):847–63. https://doi.org/10.2165/00002512-200219110-00003.
Howell AN, Ghamrawi RI, Strowd LC, Feldman SR. Pharmacological management of atopic dermatitis in the elderly. Expert Opin Pharmacother. 2020;21(7):761–71. https://doi.org/10.1080/14656566.2020.1729738.
Pariser DM. Topical steroids: a guide for use in the elderly patient. Geriatrics. 1991;46(10):51–4 (57–60, 63).
Sárdy M, Ruzicka T, Kuhn A. Topical calcineurin inhibitors in cutaneous lupus erythematosus. Arch Dermatol Res. 2009;301(1):93–8. https://doi.org/10.1007/s00403-008-0894-6.
Barikbin B, Givrad S, Yousefi M, Eskandari F. Pimecrolimus 1% cream versus betamethasone 17-valerate 0.1% cream in the treatment of facial discoid lupus erythematosus: a double-blind, randomized pilot study. Clin Exp Dermatol. 2009;34(7):776–80. https://doi.org/10.1111/j.1365-2230.2008.03138.x.
Madan V, August PJ, Chalmers RJG. Efficacy of topical tacrolimus 0.3% in clobetasol propionate 0.05% ointment in therapy-resistant cutaneous lupus erythematosus: a cohort study. Clin Exp Dermatol. 2010;35(1):27–30. https://doi.org/10.1111/j.1365-2230.2009.03351.x.
Wahie S, et al. Clinical and pharmacogenetic influences on response to hydroxychloroquine in discoid lupus erythematosus: a retrospective cohort study. J Invest Dermatol. 2011;131(10):1981–6. https://doi.org/10.1038/jid.2011.167.
Chasset F, Arnaud L, Costedoat-Chalumeau N, Zahr N, Bessis D, Francès C. The effect of increasing the dose of hydroxychloroquine (HCQ) in patients with refractory cutaneous lupus erythematosus (CLE): an open-label prospective pilot study. J Am Acad Dermatol. 2016;74(4):693-699.e3. https://doi.org/10.1016/j.jaad.2015.09.064.
Chang AY. Response to antimalarial agents in cutaneous lupus erythematosus: a prospective analysis. Arch Dermatol. 2011;147(11):1261. https://doi.org/10.1001/archdermatol.2011.191.
Cavazzana I, et al. Treatment of lupus skin involvement with quinacrine and hydroxychloroquine. Lupus. 2009;18(8):735–9. https://doi.org/10.1177/0961203308101714.
Yokogawa N, et al. Effects of hydroxychloroquine in patients with cutaneous lupus erythematosus: a multicenter, double-blind, randomized, parallel-group trial. Arthritis Rheumatol. 2017;69(4):791–9. https://doi.org/10.1002/art.40018.
Finbloom DS, Silver K, Newsome DA, Gunkel R. Comparison of hydroxychloroquine and chloroquine use and the development of retinal toxicity. J Rheumatol. 1985;12(4):692–4.
Levy GD, Munz SJ, Paschal J, Cohen HB, Pince KJ, Peterson T. Incidence of hydroxychloroquine retinopathy in 1,207 patients in a large multicenter outpatient practice. Arthritis Rheum. 1997;40(8):1482–6. https://doi.org/10.1002/art.1780400817.
Petri M, Elkhalifa M, Li J, Magder LS, Goldman DW. Hydroxychloroquine blood levels predict hydroxychloroquine retinopathy. Arthritis Rheumatol. 2020;72(3):448–53. https://doi.org/10.1002/art.41121.
Melles RB, Marmor MF. The risk of toxic retinopathy in patients on long-term hydroxychloroquine therapy. JAMA Ophthalmol. 2014;132(12):1453–60. https://doi.org/10.1001/jamaophthalmol.2014.3459.
Yusuf IH, Sharma S, Luqmani R, Downes SM. Hydroxychloroquine retinopathy. Eye Lond Engl. 2017;31(6):828–45. https://doi.org/10.1038/eye.2016.298.
Mittal L, Werth VP. The quinacrine experience in a population of patients with cutaneous lupus erythematosus and dermatomyositis. J Am Acad Dermatol. 2017;77(2):374–7. https://doi.org/10.1016/j.jaad.2017.03.027.
Yan D, Borucki R, Sontheimer RD, Werth VP. Candidate drug replacements for quinacrine in cutaneous lupus erythematosus. Lupus Sci Med. 2020;7(1): e000430. https://doi.org/10.1136/lupus-2020-000430.
Blum FR, Sampath AJ, Foulke GT. Anifrolumab for treatment of refractory cutaneous lupus erythematosus. Clin Exp Dermatol. 2022;47(11):1998–2001. https://doi.org/10.1111/ced.15335.
Shaw K, et al. Assessment of clinical response to anifrolumab in patients with refractory discoid lupus erythematosus. JAMA Dermatol. 2023;159(5):560–3. https://doi.org/10.1001/jamadermatol.2023.0175.
Loncharich MF, Anderson CW. Interferon inhibition for lupus with anifrolumab: critical appraisal of the evidence leading to FDA approval. ACR Open Rheumatol. 2022;4(6):486–91. https://doi.org/10.1002/acr2.11414.
Werth VP, et al. Trial of anti-BDCA2 antibody litifilimab for cutaneous lupus erythematosus. N Engl J Med. 2022;387(4):321–31. https://doi.org/10.1056/NEJMoa2118024.
Fairley JL, Oon S, Saracino AM, Nikpour M. Management of cutaneous manifestations of lupus erythematosus: a systematic review. Semin Arthritis Rheum. 2020;50(1):95–127. https://doi.org/10.1016/j.semarthrit.2019.07.010.
Frankel HC, Sharon VR, Vleugels RA, Merola JF, Qureshi AA. Lower-dose thalidomide therapy effectively treats cutaneous lupus erythematosus but is limited by neuropathic toxicity. Int J Dermatol. 2013;52(11):1407–9. https://doi.org/10.1111/j.1365-4632.2011.05200.x.
Luo J, Gagne JJ, Landon J, Avorn J, Kesselheim AS. Comparative effectiveness and safety of thalidomide and lenalidomide in patients with multiple myeloma in the United States of America: a population-based cohort study. Eur J Cancer Oxf Engl 1990. 2017;70:22–33. https://doi.org/10.1016/j.ejca.2016.10.018.
Pelle MT, Werth VP. Thalidomide in cutaneous lupus erythematosus. Am J Clin Dermatol. 2003;4(6):379–87. https://doi.org/10.2165/00128071-200304060-00002.
Chasset F, Tounsi T, Cesbron E, Barbaud A, Francès C, Arnaud L. Efficacy and tolerance profile of thalidomide in cutaneous lupus erythematosus: a systematic review and meta-analysis. J Am Acad Dermatol. 2018;78(2):Art. No. 2. https://doi.org/10.1016/j.jaad.2017.09.059.
Klebes M, Wutte N, Aberer E. Dapsone as Second-Line Treatment for Cutaneous Lupus Erythematosus? A Retrospective Analysis of 34 Patients and a Review of the Literature. Dermatol Basel Switz. 2016;232(1):91–6. https://doi.org/10.1159/000441054.
Youngster I, et al. Medications and glucose-6-phosphate dehydrogenase deficiency: an evidence-based review. Drug Saf. 2010;33(9):713–26. https://doi.org/10.2165/11536520-000000000-00000.
Coleman MD. Dapsone-mediated agranulocytosis: risks, possible mechanisms and prevention. Toxicology. 2001;162(1):53–60. https://doi.org/10.1016/s0300-483x(01)00360-2.
Andersohn F, Konzen C, Garbe E. Systematic review: agranulocytosis induced by nonchemotherapy drugs. Ann Intern Med. 2007;146(9):657–65. https://doi.org/10.7326/0003-4819-146-9-200705010-00009.
Chang J, Werth VP. Therapeutic options for cutaneous lupus erythematosus: recent advances and future prospects. Expert Rev Clin Immunol. 2016;12(10):Art. No. 10. https://doi.org/10.1080/1744666X.2016.1188006.
Wenzel J, Brähler S, Bauer R, Bieber T, Tüting T. Efficacy and safety of methotrexate in recalcitrant cutaneous lupus erythematosus: results of a retrospective study in 43 patients. Br J Dermatol. 2005;153(1):157–62. https://doi.org/10.1111/j.1365-2133.2005.06552.x.
Boehm IB, Boehm GA, Bauer R. Management of cutaneous lupus erythematosus with low-dose methotrexate: indication for modulation of inflammatory mechanisms. Rheumatol Int. 1998;18(2):59–62. https://doi.org/10.1007/s002960050058.
Kreuter A, Tomi NS, Weiner SM, Huger M, Altmeyer P, Gambichler T. Mycophenolate sodium for subacute cutaneous lupus erythematosus resistant to standard therapy. Br J Dermatol. 2007;156(6):1321–7. https://doi.org/10.1111/j.1365-2133.2007.07826.x.
Pisoni CN, et al. Skin manifestations of systemic lupus erythematosus refractory to multiple treatment modalities: poor results with mycophenolate mofetil. Clin Exp Rheumatol. 2005;23(3):393–6.
Gammon B, Hansen C, Costner MI. Efficacy of mycophenolate mofetil in antimalarial-resistant cutaneous lupus erythematosus. J Am Acad Dermatol. 2011;65(4):717-721.e2. https://doi.org/10.1016/j.jaad.2010.08.011.
Callen JP, Spencer LV, Burruss JB, Holtman J. Azathioprine. An effective, corticosteroid-sparing therapy for patients with recalcitrant cutaneous lupus erythematosus or with recalcitrant cutaneous leukocytoclastic vasculitis. Arch Dermatol. 1991;127(4):515–22. https://doi.org/10.1001/archderm.127.4.515.
Raptopoulou A, Linardakis C, Sidiropoulos P, Kritikos HD, Boumpas DT. Pulse cyclophosphamide treatment for severe refractory cutaneous lupus erythematosus. Lupus. 2010;19(6):744–7. https://doi.org/10.1177/0961203309358601.
Finelli PF, Naik K, DiGiuseppe JA, Prasad A. Primary lymphoma of CNS, mycophenolate mofetil and lupus. Lupus. 2006;15(12):886–8. https://doi.org/10.1177/0961203306071431.
Sakairi T, et al. Primary central nervous system lymphoma in a patient with neuropsychiatric systemic lupus erythematosus receiving mycophenolate mofetil: a case report and literature review. Mod Rheumatol Case Rep. 2022;6(1):36–40. https://doi.org/10.1093/mrcr/rxab012.
Maddox JS, Soltani K. Risk of nonmelanoma skin cancer with azathioprine use. Inflamm Bowel Dis. 2008;14(10):1425–31. https://doi.org/10.1002/ibd.20444.
Rollan MP, Cabrera R, Schwartz RA. Current knowledge of immunosuppression as a risk factor for skin cancer development. Crit Rev Oncol Hematol. 2022;177: 103754. https://doi.org/10.1016/j.critrevonc.2022.103754.
Godeau B, et al. Pneumocystis carinii pneumonia in the course of connective tissue disease: report of 34 cases. J Rheumatol. 1994;21(2):246–51.
Li J, Huang X-M, Fang W-G, Zeng X-J. Pneumocystis carinii pneumonia in patients with connective tissue disease. J Clin Rheumatol Pract Rep Rheum Musculoskelet Dis. 2006;12(3):114–7. https://doi.org/10.1097/01.rhu.0000221794.24431.36.
Danza A, Ruiz-Irastorza G. Infection risk in systemic lupus erythematosus patients: susceptibility factors and preventive strategies. Lupus. 2013;22(12):1286–94. https://doi.org/10.1177/0961203313493032.
Rackoff PJ, Rosen CJ. Pathogenesis and treatment of glucocorticoid-induced osteoporosis. Drugs Aging. 1998;12(6):477–84. https://doi.org/10.2165/00002512-199812060-00005.
Laugesen K, Jørgensen JOL, Sørensen HT, Petersen I. Systemic glucocorticoid use in Denmark: a population-based prevalence study. BMJ Open. 2017;7(5): e015237. https://doi.org/10.1136/bmjopen-2016-015237.
Grennan D, Wang S. Steroid side effects. JAMA. 2019;322(3):282. https://doi.org/10.1001/jama.2019.8506.
Ruzicka T, Meurer M, Bieber T. Efficiency of acitretin in the treatment of cutaneous lupus erythematosus. Arch Dermatol. 1988;124(6):897–902.
Shornick JK, Formica N, Parke AL. Isotretinoin for refractory lupus erythematosus. J Am Acad Dermatol. 1991;24(1):49–52. https://doi.org/10.1016/0190-9622(91)70008-P.
Tett SE. Clinical pharmacokinetics of slow-acting antirheumatic drugs. Clin Pharmacokinet. 1993;25(5):392–407. https://doi.org/10.2165/00003088-199325050-00005.
Jallouli M, et al. The association of systemic lupus erythematosus and myasthenia gravis: a series of 17 cases, with a special focus on hydroxychloroquine use and a review of the literature. J Neurol. 2012;259(7):1290–7. https://doi.org/10.1007/s00415-011-6335-z.
Krishna S, White NJ. Pharmacokinetics of quinine, chloroquine and amodiaquine. Clinical implications. Clin Pharmacokinet. 1996;30(4):263–99. https://doi.org/10.2165/00003088-199630040-00002.
Salako LA, Walker O, Iyun AO. Pharmacokinetics of chloroquine in renal insufficiency. Afr J Med Med Sci. 1984;13(3–4):177–82.
Chen N, et al. Pharmacokinetics of lenalidomide in subjects with various degrees of renal impairment and in subjects on hemodialysis. J Clin Pharmacol. 2007;47(12):1466–75. https://doi.org/10.1177/0091270007309563.
Kintzel PE, Dorr RT. Anticancer drug renal toxicity and elimination: dosing guidelines for altered renal function. Cancer Treat Rev. 1995;21(1):33–64. https://doi.org/10.1016/0305-7372(95)90010-1.
Al-Hasani H, Roussou E. Methotrexate for rheumatoid arthritis patients who are on hemodialysis. Rheumatol Int. 2011;31(12):1545–7. https://doi.org/10.1007/s00296-011-2041-5.
Bergan S, et al. Personalized Therapy for Mycophenolate: Consensus Report by the International Association of Therapeutic Drug Monitoring and Clinical Toxicology. Ther Drug Monit. 2021;43(2):150–200. https://doi.org/10.1097/FTD.0000000000000871.
Bach JF, Dardenne M. The metabolism of azathioprine in renal failure. Transplantation. 1971;12(4):253–9. https://doi.org/10.1097/00007890-197110000-00003.
Schusziarra V, Ziekursch V, Schlamp R, Siemensen HC. Pharmacokinetics of azathioprine under haemodialysis. Int J Clin Pharmacol Biopharm. 1976;14(4):298–302.
Krens SD, et al. Dose recommendations for anticancer drugs in patients with renal or hepatic impairment. Lancet Oncol. 2019;20(4):e200–7. https://doi.org/10.1016/S1470-2045(19)30145-7.
Abouraya M, Sacco JC, Hayes K, Thomas S, Kitchens CS, Trepanier LA. Dapsone-associated methemoglobinemia in a patient with slow NAT2*5B haplotype and impaired cytochrome b5 reductase activity. J Clin Pharmacol. 2012;52(2):272–8. https://doi.org/10.1177/0091270010393343.
Lee SM, Geetha D. Dapsone induced hemolysis in a patient with ANCA associated glomerulonephritis and normal G6PD level and implications for clinical practice: case report and review of the literature. Springerplus. 2015;4:29. https://doi.org/10.1186/s40064-015-0816-y.
Mannemuddhu SS, Ali R, Kadhem S, Ruchi R. Unusual cause of persistent dyspnea in a patient with nephrotic syndrome: dapsone-induced methemoglobinemia. CEN Case Rep. 2021;10(3):336–40. https://doi.org/10.1007/s13730-020-00565-8.
Floyd J, Mirza I, Sachs B, Perry MC. Hepatotoxicity of chemotherapy. Semin Oncol. 2006;33(1):50–67. https://doi.org/10.1053/j.seminoncol.2005.11.002.
Gildea DT, Roswarski JL. Severe Lenalidomide-Associated Hyperbilirubinemia. Cureus. 2023. https://doi.org/10.7759/cureus.34408.
Zanella M-C, Rubbia-Brandt L, Giostra E, Chalandon Y, Hadengue A, Spahr L. A case of drug-induced hepatitis due to lenalidomide. Case Rep Gastroenterol. 2011;5(1):217–22. https://doi.org/10.1159/000326935.
Legendre DP, Muzny CA, Swiatlo E. Hansen’s disease (Leprosy): current and future pharmacotherapy and treatment of disease-related immunologic reactions. Pharmacotherapy. 2012;32(1):27–37. https://doi.org/10.1002/PHAR.1009.
Liu D, et al. Chloroquine and hydroxychloroquine are associated with reduced cardiovascular risk: a systematic review and meta-analysis. Drug Des Dev Ther. 2018;12:1685–95. https://doi.org/10.2147/DDDT.S166893.
McAllister HA, Ferrans VJ, Hall RJ, Strickman NE, Bossart MI. Chloroquine-induced cardiomyopathy. Arch Pathol Lab Med. 1987;111(10):953–6.
Nadeem U, et al. Chloroquine- and hydroxychloroquine-induced cardiomyopathy: a case report and brief literature review. Am J Clin Pathol. 2021;155(6):793–801. https://doi.org/10.1093/ajcp/aqaa253.
Doyno C, Sobieraj DM, Baker WL. Toxicity of chloroquine and hydroxychloroquine following therapeutic use or overdose. Clin Toxicol Phila Pa. 2021;59(1):12–23. https://doi.org/10.1080/15563650.2020.1817479.
Rabkin SW. Aging effects on QT interval: implications for cardiac safety of antipsychotic drugs. J Geriatr Cardiol JGC. 2014;11(1):20–5. https://doi.org/10.3969/j.issn.1671-5411.2014.01.005.
Rizvi SK, Chong BF. Cost minimization analysis of mainstay treatments in cutaneous lupus erythematous. Dermatol Ther. 2022;35(1): e15190. https://doi.org/10.1111/dth.15190.
Liu SW, et al. Willingness-to-pay stated preferences in cutaneous lupus erythematosus: a pilot study. Arch Dermatol Res. 2020;312(7):527–31. https://doi.org/10.1007/s00403-019-02006-8.
Chen CL, Kuppermann M, Caughey AB, Zane LT. A community-based study of acne-related health preferences in adolescents. Arch Dermatol. 2008. https://doi.org/10.1001/archderm.144.8.988.
Delfino M, Holt EW, Taylor CR, Wittenberg E, Qureshi AA. Willingness-to-pay stated preferences for 8 health-related quality-of-life domains in psoriasis: a pilot study. J Am Acad Dermatol. 2008;59(3):439–47. https://doi.org/10.1016/j.jaad.2008.05.032.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Dr Galen Foulke is supported by a Dermatology Foundation Medical Dermatology Career Development Award.
Conflicts of interest/competing interest
Dr Astia Allenzara’s spouse is employed by LatigoBio pharmaceuticals. Dr Galen Foulke has served as a consultant to Astra Zeneca, the manufacturer of anifrolumab. Dr Matthew Helm and Briana Heinly have no conflicts of interest to disclose.
Author contributions
Briana Heinly performed the full literature review and drafted all parts of the manuscript. Dr Foulke also did portions of the literature review, provided multiple rounds of edits, and played a major role in the revision of the manuscript. Dr Helm and Dr Allenzara both provided expert clinical critique of the paper, provided additional sources, and revised the manuscript prior to submission. All authors read and approved the final manuscript.
Ethics approval
Not applicable.
Patient consent to publish
Patients consented to the publication of their clinical photos.
Patient consent to participate
Not applicable.
Code availability
Not applicable.
Data/material availability
As this is a review, no original data were generated nor analyzed.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heinly, B., Allenzara, A., Helm, M. et al. Cutaneous Lupus Erythematosus: Review and Considerations for Older Populations. Drugs Aging 41, 31–43 (2024). https://doi.org/10.1007/s40266-023-01079-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-023-01079-5