Abstract
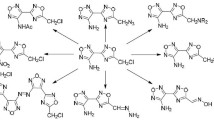
Heterocyclization of furan-2-carbonyl isothiocyanate 1 with a variety of aliphatic and aromatic nitrogen nucleophiles resulted in the formation of a new series of heterocycles including triazines, pyrimidines, oxadiazines, imidazolidines, thiadiazoles and their condensed candidates. The antibacterial screening for a group of the newly synthesized compounds showed that they possess moderate antibacterial activities against examples of Gram-positive and Gram-negative bacteria.






Similar content being viewed by others
References
Z. Arzehgar, S. Sajjadifar, H. Arandiyan, Asian J. Green Chem. 3, 43 (2019)
A. Ramazani, M. Khoobi, A. Torkaman, F.Z. Nasrabadi, H. Forootanfar, M. Shakibaie, M. Jafari, A. Ameri, S. Emami, M.A. Faramarzi, A. Foroumadi, A. Shafiee, Eur. J. Med. Chem. 78, 151 (2014)
H. Aghahosseini, A. Ramazani, N.S. Jalayer, Z. Ranjdoost, A. Souldozi, K. Slepokura, T. Lis, Org. Lett. 21, 22 (2019)
H. Parham, B.A. Esfahani, J. Iran. Chem. Soc. 5, 457 (2008)
I.E. Tolpygin, E.N. Shepelenko, G.S. Borodkin, A.D. Dubonosov, V.A. Bren, V.I. Minkin, Chem. Heterocycl. Compd. 46, 546 (2010)
F. Aydin, N. Tunoglu, D. Aykac, Asian J. Chem. 25, 2455 (2013)
M.M. Hemdan, A.F. Fahmy, N.F. Ali, E. Hegazi, A. Abd-Elhaleem, Chin. J. Chem. 25, 388 (2007)
M.G. Assy, H.Y. Moustafa, Phosphorous Sulfur Silicon Relat. Elem. 105, 213 (1995)
E.V. Sadchikova, V.A. Bakulev, J.O. Subbotina, D.L. Privalova, W. Dehaen, K.V. Hecke, K. Robeyns, L.V. Meervelt, Tetrahedron 69, 6992 (2013)
F. Piscitelli, C. Ballatore, A.B. Smith III, Bioorg. Med. Chem. Lett. 20, 648 (2010)
O.V. Shablykin, A.V. Golovchenko, V.S. Brovarets, B.S. Drach, Russ. J. Gen. Chem. 77, 935 (2007)
B.A. Trofimov, J. Heterocycl. Chem. 36, 1490 (1999)
R.V. Chambhare, B.G. Khadse, A.S. Bobde, R.H. Bahekar, Eur. J. Med. Chem. 38, 89 (2003)
M.M.H. Bhuiyan, K.M.M. Rahman, M.K. Hossain, M.A. Rahim, M.I. Hossain, Croat. Chem. Acta 78, 633 (2005)
M.A. Salem, M.I. Marzouk, N.F. Mahmoud, J. Serb. Chem. Soc. 79, 1059 (2014)
M. Hossam, D.S. Lasheen, N.S.M. Ismail, A. Esmat, A.M. Mansour, A.N.B. Singab, K.A.M. Abouzid, Eur. J. Med. Chem. 144, 330 (2018)
A.A. Abu-Hashem, M.M. Youssef, Molecules 16, 1956 (2011)
M.S. Frasinyuk, S.V. Gorelov, S.P. Bondarenko, V.P. Khilya, Chem. Heterocycl. Compd. 45, 1269 (2009)
E. Abdelghani, S.A. Said, M.G. Assy, A.M. Abdel Hamid, Arab. J. Chem. 10, S2926 (2017)
H.A. El-Sayed, A.M. Abdel Hamid, M.G. Assy, T.S. Farag, Synth. Commun. 48, 794 (2018)
A.M. Abdel Hamid, W. Shehta, J. Iran. Chem. Soc. 15, 2771 (2018)
S. Noura, M. Ghorbani, M.A. Zolfigol, M. Narimani, M. Yarie, M. Oftadeh, J. Mol. Liq. 271, 778 (2018)
F. Karimi, M.A. Zolfigol, M. Yarie, Mol. Catal. 463, 20 (2019)
M. Yarie, Iran. J. Catal. 7, 85 (2017)
A.R. Katritzky, P.J. Steel, S.N. Denisenko, Tetrahedron 57, 3309 (2001)
B. Bonev, J. Hooper, J. Parisot, J. Antimicrob. Chemoth. 61, 1301 (2008)
M. Uher, D. Ilavsky, J. Foltin, K. Skvareninova, Collect. Czech. Chem. Commun. 46, 3128 (1981)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abdel Hamid, A.M. Addition–cyclization reactions of furan-2-carbonyl isothiocyanate with nitrogen nucleophiles as a synthetic route to novel azines and azoles of potential biological activity. J IRAN CHEM SOC 16, 1853–1861 (2019). https://doi.org/10.1007/s13738-019-01659-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01659-6