Abstract
Purpose of Review
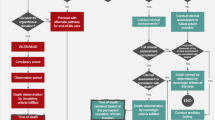
Due to the persistent sparsity of donor organs, today, organ donation after circulatory death (DCD) has become a promising solution to alleviate the burden of waitlist mortality.
Recent Findings
With survival rates comparable to that of standard donor lungs, DCD utilization is growing worldwide. However, the barriers to DCD lung transplantation adoption should be explored.
Summary
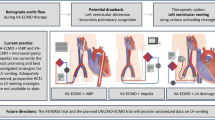
The utilization of DCD lungs is currently a viable option for lung transplantation. In this review, we weigh up the ethical, legal, and clinical barriers facing this alternative, then suggest some solutions for the current setbacks. We also look at cutting-edge innovations accompanying DCD utilization and comment on prospective research directions in this field that may change future practice standards.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as:
•• Of major importance
World Health Organization (WHO). Global Observation on Donation and Transplantation. 2019. Available from: http://www.transplant-observatory.org/data-charts-and-tables/chart/. Accessed 7 Apr 2021.
Dominguez-Gil B, et al. Expanding controlled donation after the circulatory determination of death: Statement from an international collaborative. Intensive Care Med. 2021;47(3):265–81.
Bernat JL, et al. The circulatory-respiratory determination of death in organ donation. Crit Care Med. 2010;38(3):963–70.
Westphal GA, et al. Guidelines for the assessment and acceptance of potential brain-dead organ donors. Rev Bras Ter Intensiva. 2016;28(3):220–55.
Erasmus ME, et al. DCD lung donation: Donor criteria, procedural criteria, pulmonary graft function validation, and preservation. Transpl Int. 2016;29(7):790–7.
Valapour M, et al. OPTN/SRTR 2019 Annual data report: Lung. Am J Transplant. 2021;21(Suppl 2):441–520.
de Antonio DG, et al. Results of clinical lung transplant from uncontrolled non-heart-beating donors. J Heart Lung Transplant. 2007;26(5):529–34.
Goff RR, Daly RC, Lease ED. Ex vivo lung perfusion on donor lungs in the United States: National trends and post-transplant outcomes. J Heart Lung Transplant. 2020;39.
Zych B, et al. Lungs from donation after circulatory death donors: An alternative source to brain-dead donors? Midterm results at a single institution. Eur J Cardiothorac Surg. 2012;42(3):542–9.
Saxena P, et al. Procurement of lungs for transplantation following donation after circulatory death: The Alfred technique. J Surg Res. 2014;192(2):642–6.
Cypel M, et al. International society for heart and lung transplantation donation after circulatory death registry report. J Heart Lung Transplant. 2015;34(10):1278–82.
Sabashnikov A, et al. Long-term results after lung transplantation using organs from circulatory death donors: a propensity score-matched analysis dagger. Eur J Cardiothorac Surg. 2016;49(1):46–53.
Mooney JJ, et al. Lung quality and utilization in controlled donation after circulatory determination of death within the United States. Am J Transplant. 2016;16(4):1207–15.
van Suylen V, et al. A multicenter study on long-term outcomes after lung transplantation comparing donation after circulatory death and donation after brain death. Am J Transplant. 2017;17(10):2679–86.
Villavicencio MA, et al. Lung transplantation from donation after circulatory death: United States and single-center experience. Ann Thorac Surg. 2018;106(6):1619–27.
•• Van Raemdonck D, et al. Donation after circulatory death in lung transplantation-five-year follow-up from ISHLT Registry. J Heart Lung Transplant. 2019;38(12):1235–45. Analysis of the ISHLT registry revealed comparable 5-year survival between DCD and DBD lung transplant recipients. This study also demonstrated a significant 20-fold increase in DCD lung transplantation globally.
Copeland H, et al. Donor heart and lung procurement: A consensus statement. J Heart Lung Transplant. 2020;39(6):501–17.
Krutsinger D, et al. Lung transplantation from donation after cardiocirculatory death: A systematic review and meta-analysis. J Heart Lung Transplant. 2015;34(5):675–84.
Van Raemdonck DE, et al. Warm ischemic tolerance in collapsed pulmonary grafts is limited to 1 hour. Ann Surg. 1998;228(6):788–96.
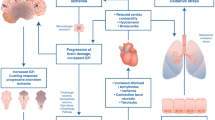
Avlonitis VS, et al. The hemodynamic mechanisms of lung injury and systemic inflammatory response following brain death in the transplant donor. Am J Transplant. 2005;5(4 Pt 1):684–93.
Neyrinck AP, et al. Comparative study of donor lung injury in heart-beating versus non-heart-beating donors. Eur J Cardiothorac Surg. 2006;30(4):628–36.
Jin Z, et al. Review 1: Lung transplant-from donor selection to graft preparation. J Anesth. 2020;34(4):561–74.
Barklin A, et al. Insulin alters cytokine content in two pivotal organs after brain death: A porcine model. Acta Anaesthesiol Scand. 2008;52(5):628–34.
Inci I. Donors after cardiocirculatory death and lung transplantation. J Thorac Dis. 2017;9(8):2660–9.
•• Ortega-Deballon I, Hornby L, Shemie SD. Protocols for uncontrolled donation after circulatory death: A systematic review of international guidelines, practices and transplant outcomes. Crit Care. 2015;19:268. There is wide variability in technical durations of uDCD organ procurement including variability in the definition of refractory CPR and the duration of the no-touch period.
Keshavamurthy S, Rodgers-Fischl P. Donation after circulatory death (DCD)-lung procurement. Indian J Thorac Cardiovasc Surg. 2021;1–8.
Levvey B, et al. Influence of lung donor agonal and warm ischemic times on early mortality: Analyses from the ISHLT DCD Lung Transplant Registry. J Heart Lung Transplant. 2019;38(1):26–34.
Motta ED. The ethics of heparin administration to the potential non-heart-beating organ donor. J Prof Nurs. 2005;21(2):97–102.
Tsui SSL, Oniscu GC. Extending normothermic regional perfusion to the thorax in donors after circulatory death. Curr Opin Organ Transplant. 2017;22(3):245–50.
Manara A, et al. Maintaining the permanence principle for death during in situ normothermic regional perfusion for donation after circulatory death organ recovery: A United Kingdom and Canadian proposal. Am J Transplant. 2020;20(8):2017–25.
Gries CJ, et al. An official American thoracic society/international society for heart and lung transplantation/society of critical care medicine/association of organ and procurement organizations/united network of organ sharing statement: Ethical and policy considerations in organ donation after circulatory determination of death. Am J Respir Crit Care Med. 2013;188(1):103–9.
Egan TM, et al. A strategy to increase the donor pool: Use of cadaver lungs for transplantation. Ann Thorac Surg. 1991;52(5):1113–20. Discussion 1120–1.
Steen S, Ingemansson R, Eriksson L, Pierre L, Algotsson L, Wierup P, et al. First human transplantation of a nonacceptable donor lung after reconditioning ex vivo. Ann Thorac Surg. 2007;83(6):2191-4.
Keshavjee SH, et al. A method for safe twelve-hour pulmonary preservation. J Thorac Cardiovasc Surg. 1989;98(4):529–34.
Suberviola B, et al. Excellent long-term outcome with lungs obtained from uncontrolled donation after circulatory death. Am J Transplant. 2019;19(4):1195–201.
De Oliveira NC, et al. Lung transplantation with donation after cardiac death donors: Long-term follow-up in a single center. J Thorac Cardiovasc Surg. 2010;139(5):1306–15.
Ruttens D, et al. Short- and long-term outcomes after lung transplantation from circulatory-dead donors: A single-center experience. Transplantation. 2017;101(11):2691–4.
Zeriouh M, et al. Utilization of the organ care system for bilateral lung transplantation: Preliminary results of a comparative study. Interact Cardiovasc Thorac Surg. 2016;23(3):351–7.
Valenza F, et al. Ex vivo lung perfusion to improve donor lung function and increase the number of organs available for transplantation. Transpl Int. 2014;27(6):553–61.
Warnecke G, et al. Normothermic ex-vivo preservation with the portable organ care system lung device for bilateral lung transplantation (INSPIRE): A randomised, open-label, non-inferiority, phase 3 study. Lancet Respir Med. 2018;6(5):357–67.
Sanchez PG, Davis RD, D’Ovidio F, et al. The NOVEL lung trial one-year outcomes. J Heart and Lung Transplant. 2014;33.
Tian D, et al. Outcomes of marginal donors for lung transplantation after ex vivo lung perfusion: A systematic review and meta-analysis. J Thorac Cardiovasc Surg. 2020;159(2):720–30 e6.
Luo Q, et al. The conversional efficacy of ex vivo lung perfusion and clinical outcomes in patients undergoing transplantation of donor lungs by ex vivo lung perfusion: A meta-analysis. Ann Transplant. 2019;24:647–60.
Andreasson AS, et al. Profiling inflammation and tissue injury markers in perfusate and bronchoalveolar lavage fluid during human ex vivo lung perfusion. Eur J Cardiothorac Surg. 2017;51(3):577–86.
Kakishita T, et al. Suppression of inflammatory cytokines during ex vivo lung perfusion with an adsorbent membrane. Ann Thorac Surg. 2010;89(6):1773–9.
Aboelnazar NS, et al. Negative pressure ventilation decreases inflammation and lung edema during normothermic ex-vivo lung perfusion. J Heart Lung Transplant. 2018;37(4):520–30.
Andreasson ASI, et al. The role of interleukin-1beta as a predictive biomarker and potential therapeutic target during clinical ex vivo lung perfusion. J Heart Lung Transplant. 2017;36(9):985–95.
Buchko MT, Boroumand N, Cheng JC, van Staalduinen F, Freed DH, Nagendran J. Initial successful clinical transplantation using negative pressure ventilation ex-situ lung perfusion with extended criteria donor lungs. J Heart Lung Transplant. 2020;39.
Pagano F, et al. Cytoprotective and antioxidant effects of steen solution on human lung spheroids and human endothelial cells. Am J Transplant. 2017;17(7):1885–94.
Wang X, et al. Experimental ex vivo lung perfusion with sevoflurane: Effects on damaged donor lung grafts. Interact Cardiovasc Thorac Surg. 2018;26(6):977–84.
Loor G, et al. Prolonged EVLP using OCS lung: Cellular and acellular perfusates. Transplantation. 2017;101(10):2303–11.
Loor G, et al. Portable normothermic ex-vivo lung perfusion, ventilation, and functional assessment with the Organ Care System on donor lung use for transplantation from extended-criteria donors (EXPAND): A single-arm, pivotal trial. Lancet Respir Med. 2019;7(11):975–84.
Sage AT, et al. Fractal circuit sensors enable rapid quantification of biomarkers for donor lung assessment for transplantation. Sci Adv. 2015;1(7):e1500417.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
A. Alnajar, A. Sayed, O. Shazly, S.H. Roberts, A. Ghodsizad, and S.A. Hirji declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Lung Transplant
Rights and permissions
About this article
Cite this article
Alnajar, A., Sayed, A., Shazly, O. et al. Donation After Circulatory Death Donors for Lung Transplantation: the US and Worldwide Status. Curr Pulmonol Rep 10, 149–157 (2021). https://doi.org/10.1007/s13665-021-00281-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13665-021-00281-3