Abstract
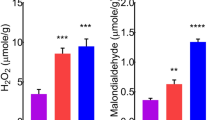
The objective of this study was to create a method to evaluate the aging of coir dust. A rainproof plastic house was used to monitor the fresh coir dust for 24 weeks, and the samples were collected every 2 weeks during aging. The aging effects on coir dust were investigated with the changes in mineral nutrient contents and plug seedling growth in four different vegetables. Experiments were conducted to determine the appropriate degree of aging by analyzing the changes of total phenol and tannin contents, color, and near-infrared spectral characteristics as influenced by the aging duration of coir dust. The pH, electrical conductivity (EC), and cation exchange capacity (CEC) values of the non-aged coir dust were 5.04, 9.19 dS m−1, and 75.2 cmol+ kg−1, respectively. The concentrations of Cl and K in the non-aged coir dust were 4061.8 and 3487.4 mg kg−1, respectively. The contents of total phenol and tannin in the non-aged coir dust were 168.8 and 38.8 mg kg−1, respectively. The pH rose continuously to 5.94 at 24 weeks, and the EC decreased to 1.8 dS m−1 after 4 weeks of aging and then remained at a similar level. The concentrations of elements decreased drastically except for NH4+ and NO3− until 4 weeks of aging, and later they remained at a similar level. The total phenol content decreased greatly at 2 weeks of aging, and the tannin content decreased continuously from 38.8 mg kg−1 in week 0 to 0.15 mg kg−1 at 24 weeks of aging. The color indicators b* and C* decreased until 12 weeks of aging, and after that, they remained at a similar level. The optimum aging of coir dust was judged based on b* and C* values. The spectral analysis showed that the coir dust aged over 18 weeks could be distinguished from the earlier stage of aging with 91% accuracy. When the coir dust with varied aging durations was used as a root substrate, the fresh weights of radish, Korean cabbage, tomato, and red pepper increased quadratically as the aging period of the coir dust became longer (R2 = 0.7824, 0.7529, 0.4718, and 0.5458 in radish, Chinese cabbage, tomato, and red pepper, respectively; p ≤ 0.001).








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abad M, Noguera P, Puchades R, Maquiera A, Noguera V (2002) Physio-chemical properties of some coconut coir dust for use as a peat substitute for containerised ornamental plants. Bioresour Technol 82:241–245. https://doi.org/10.1016/S0960-8524(01)00189-4
Abad M, Carrión C, Noguera V, Noguera P, Maquieira A, Puchades R (2005) Physical properties of various coconut coir dust compared to peat. HortScience 40:2138–2144
Aendekerk TGL, Cevat H, Dolmans N, van Elderen C, Kipp JA, de Kreij C et al (2000) International substrate manual: analysis, characteristics, recommendations. Elsevier, Dordrecht
Bunt AC (1988) Media and mixes for container: grown plants, 2nd edn. Unwin Hyman Ltd, London
Carlile WR, Cattivello C, Zaccheo P (2015) Organic growing media: constituents and properties. Vadose Zone J 14:1–13. https://doi.org/10.2136/vzj2015.04.0057
Evans MR, Konduru S, Stamps RH (1996) Source variation in physical and chemical properties of coconut coir dust. HortScience 6:965–967
Fornes F, Belda RM, Abad M, Noguera P, Puchades R, Maquieira A, Noguera V (2003) The microstructure of coconut coir dusts for use as alternatives to peat in soilless growing media. Aust J Exp Agric 43:1171–1179. https://doi.org/10.1071/EA02128
Francis FJ (1980) Color quality evaluation of horticultural crops. HortScience 15:58–59
Gorinstein S, Weisz M, Zemser M, Tilis K, Stiller A, Flam I, Gat Y (1993) Spectroscopic analysis of polyphenols in white wines. J Ferment Bioeng 75:115–120
Grubešić RJ, Vuković J, Kremer D, Vladimir-Knežević S (2005) Spectrophotometric method for polyphenols analysis: prevalidation and application on Plantago L. species. J Pharm Biomed Anal 39:837–842
Handreck KA (1993) Properties of coir dust, and its use in the formulation of soilless potting media. Commun Soil Sci Plant Anal 24:349–363. https://doi.org/10.1080/00103629309368804
Israel AU, Ogali RE, Akaranta O, IB Obot (2011) Extraction and characterization of coconut (Cocos nucifera L.) coir dust. Songklanakarin J Sci Technol 33:717–724
Khatiwora E, Adsul VB, Kulkarni MM, Deshpande NR, Kashalkar RV (2010) Spectroscopic determination of total phenol and flavonoid contents of Ipomoea carnea. Int J ChemTech Res 2:1698–1701
Kim DY, Cho BK (2015) Rapid monitoring of the fermentation process for Korean traditional rice wine ‘Makgeolli’ using FT-NIR spectroscopy. Infrared Phys Technol 73:95–102
Kim CH, Park IS, Choi JM (2018) Fertilization levels affect growth of red leaf lettuce and chemical characteristics of re-circulated solution and root media. Hortic Sci Technol 36:478–486. https://doi.org/10.12972/kjhst.20180048
Konduru S, Evans MR, Stamps RH (1999) Coconut husk and processing effects on chemical and physical properties of coconut coir dust. HortScience 34:88–90
Lim MY, Jeong HJ, Choi SH, Choi GL, Kim SH (2020) Effect of planting density by cultivars on the growth and yield of melons (Cucumis melo L.) in hydroponics using coir substrates. Hortic Sci Technol 38:850–859. https://doi.org/10.7235/HORT.20200077
Ma YB, Nichols DG (2004) Phytotoxicity and detoxification of fresh coir dust and coconut shell. Commun Soil Sci Plant Anal 35:205–218. https://doi.org/10.1081/CSS-120027644
Marschner P (2012) Marschner’s mineral nutrition of higher plants, 3rd edn. Academic Press Inc., San Diego
Nelson PV (2012) Greenhouse operation and management, 7th edn. Prentice Hall, Englewood Cliffs
NIAST (2000) Analysis methods of soil and plant. National Institute of Agricultural Science and Technology RDA, Suwon
Nichols D, Yazaki Y (1979) Phytotoxicity of Eucalyptus camaldulensis and E. regnan sawdusts. Aust For Res 9:35–39
Politycka B, Wojcik-Wojtkowiak D, Pudelski T (1985) Phenolic compounds as a cause of phytotoxicity in greenhouse substrates repeatedly used in cucumber growing. Acta Hortic 156:89–94
Prasad M, Maher MJ (2013) Colour of peat as an indicator of chemical, biological and physical properties of peats. Acta Hortic 1013:89–94
Radjagukguk B, Soekotjo A, Soeseno OH, Santoso HJ (1983) A comparative study of peats and other media for containerized forest tree seedlings. Acta Hortic 150:445–458
Raviv M, Lieth JH (2008) Soilless culture; theory and practice. Elsevier, NY
Yazaki Y, Nichols D (1978) Phytotoxicity components of Pinus radiato bark. Aust For Res 8:185–198
Acknowledgements
This work was carried out with the support of “The Cooperative Research Program for Agriculture Science & Technology Development (Project No. PJ01188602)”, Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Contributions
YSK performed experiments, collected data and analyzed data. ISP assisted in collecting data and amending the manuscript. ESP assisted in data analysis. CWL designed the experiments and analyzed the data. BKC and JMC designed the experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Myung-Min Oh.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, Y.S., Park, I.S., Park, E. et al. Changes in the chemical properties of coir dust with increasing aging time and development of a method for determining moderate aging degree. Hortic. Environ. Biotechnol. 62, 547–557 (2021). https://doi.org/10.1007/s13580-021-00339-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-021-00339-w