Abstract
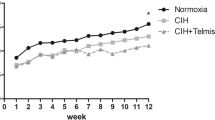
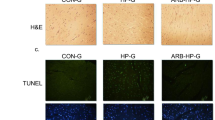
Obstructive sleep apnea syndrome is generally associated with multiple cardiovascular disorders, such as myocardial hypertrophy. Autophagy is strictly modulated to maintain cardiac homeostasis. Post-injury autophagy is closely associated with pathological cardiac hypertrophy. BCL2 interacting protein 3 (BNIP3) and BNIP3-like protein (BNIP3L) can cause cell death and are important for hypoxia-elicited autophagy. Here, we evaluated whether BNIP3 could mitigate functional remodeling and cardiac hypertrophy through regulation of autophagy. Male WT rats or rats with BNIP3 knockout were subjected to chronic intermittent hypoxia (CIH) for 8 h/day over 5 weeks. Echocardiography and morphology were employed to assess the cardioprotective effects. Autophagy was assessed via transmission electron microscopy and detection of LC3, p62, and Beclin-1. Terminal deoxynucleotidyl transferase dUTP nick end labeling and the Bax/Bcl2 ratio were used to monitor apoptosis. Biochemical evaluations were performed to assess oxidative stress. Additionally, BNIP3-knockdown H9c2 cells that were subjected to CIH were used to examine autophagy and apoptosis to confirm the findings of the animal study. The CIH group showed elevated heart weight/body weight and left ventricle weight/body weight proportions, along with left ventricular remodeling. CIH-exposed rats exhibited dramatically higher fractional shortening and ejection fractions than the controls. In addition, the levels of autophagy markers Beclin-1 and LC3-II/I were increased, whereas the level of p62 was reduced by CIH treatment. The oxidative marker levels and the apoptosis index in the CIH group were markedly increased. Knockout of BNIP3 significantly aggravated the impairment in cardiac function, apoptosis, oxidative stress, and hypertrophy of CIH rats, while significantly reducing autophagy. The autophagy-associated PI3K/Akt/mTOR pathway was also deactivated by BNIP3 knockout. At the cellular level, CIH treatment significantly upregulated autophagy and apoptosis; however, BNIP3 silencing reduced autophagy and promoted apoptosis. CIH treatment-mediated upregulation of BNIP3 expression plays a crucial role in autophagy by targeting the PI3K/Akt/mTOR pathway, alleviating cardiac hypertrophy.








Similar content being viewed by others
References
Huang J, Pinto SJ, Yuan H, Katz ES, Karamessinis LR, Bradford RM, et al. Upper airway collapsibility and genioglossus activity in adolescents during sleep. Sleep. 2012;35(10):1345–52. https://doi.org/10.5665/sleep.2110.
Capone C, Faraco G, Coleman C, Young CN, Pickel VM, Anrather J, et al. Endothelin 1-dependent neurovascular dysfunction in chronic intermittent hypoxia. Hypertension. 2012;60(1):106–13. https://doi.org/10.1161/hypertensionaha.112.193672.
Gottlieb DJ, Yenokyan G, Newman AB, O’Connor GT, Punjabi NM, Quan SF, et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the sleep heart health study. Circulation. 2010;122(4):352–60. https://doi.org/10.1161/circulationaha.109.901801.
Klusonová P, Reháková L, Borchert G, Vagnerová K, Neckár J, Ergang P, et al. Chronic intermittent hypoxia induces 11beta-hydroxysteroid dehydrogenase in rat heart. Endocrinology. 2009;150(9):4270–7. https://doi.org/10.1210/en.2008-1493.
Jordan AS, McSharry DG, Malhotra A. Adult obstructive sleep apnoea. Lancet. 2014;383(9918):736–47. https://doi.org/10.1016/s0140-6736(13)60734-5.
Yeung HM, Hung MW, Lau CF, Fung ML. Cardioprotective effects of melatonin against myocardial injuries induced by chronic intermittent hypoxia in rats. J Pineal Res. 2015;58(1):12–25. https://doi.org/10.1111/jpi.12190.
Del Rio R, Moya EA, Parga MJ, Madrid C, Iturriaga R. Carotid body inflammation and cardiorespiratory alterations in intermittent hypoxia. Eur Respir J. 2012;39(6):1492–500. https://doi.org/10.1183/09031936.00141511.
Ding W, Zhang X, Huang H, Ding N, Zhang S, Hutchinson SZ, et al. Adiponectin protects rat myocardium against chronic intermittent hypoxia-induced injury via inhibition of endoplasmic reticulum stress. PLoS ONE. 2014;9(4):e94545. https://doi.org/10.1371/journal.pone.0094545.
Gustafsson AB, Gottlieb RA. Autophagy in ischemic heart disease. Circ Res. 2009;104(2):150–8. https://doi.org/10.1161/circresaha.108.187427.
Levine B, Deretic V. Unveiling the roles of autophagy in innate and adaptive immunity. Nat Rev Immunol. 2007;7(10):767–77. https://doi.org/10.1038/nri2161.
Shintani T, Klionsky DJ. Autophagy in health and disease: a double-edged sword. Science. 2004;306(5698):990–5. https://doi.org/10.1126/science.1099993.
Mizushima N, Levine B, Cuervo AM, Klionsky DJ. Autophagy fights disease through cellular self-digestion. Nature. 2008;451(7182):1069–75. https://doi.org/10.1038/nature06639.
Hariharan N, Zhai P, Sadoshima J. Oxidative stress stimulates autophagic flux during ischemia/reperfusion. Antioxid Redox Signal. 2011;14(11):2179–90. https://doi.org/10.1089/ars.2010.3488.
Mariño G, Pietrocola F, Kong Y, Eisenberg T, Hill JA, Madeo F, et al. Dimethyl α-ketoglutarate inhibits maladaptive autophagy in pressure overload-induced cardiomyopathy. Autophagy. 2014;10(5):930–2. https://doi.org/10.4161/auto.28235.
Lavandero S, Chiong M, Rothermel BA, Hill JA. Autophagy in cardiovascular biology. J Clin Invest. 2015;125(1):55–64. https://doi.org/10.1172/jci73943.
Thomas RL, Roberts DJ, Kubli DA, Lee Y, Quinsay MN, Owens JB, et al. Loss of MCL-1 leads to impaired autophagy and rapid development of heart failure. Genes Dev. 2013;27(12):1365–77. https://doi.org/10.1101/gad.215871.113.
Li Z, Song Y, Liu L, Hou N, An X, Zhan D, et al. miR-199a impairs autophagy and induces cardiac hypertrophy through mTOR activation. Cell Death Differ. 2017;24(7):1205–13. https://doi.org/10.1038/cdd.2015.95.
Bellot G, Garcia-Medina R, Gounon P, Chiche J, Roux D, Pouysségur J, et al. Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol Cell Biol. 2009;29(10):2570–81. https://doi.org/10.1128/mcb.00166-09.
Willis SN, Adams JM. Life in the balance: how BH3-only proteins induce apoptosis. Curr Opin Cell Biol. 2005;17(6):617–25. https://doi.org/10.1016/j.ceb.2005.10.001.
Boyd JM, Malstrom S, Subramanian T, Venkatesh LK, Schaeper U, Elangovan B, et al. Adenovirus E1B 19 kDa and Bcl-2 proteins interact with a common set of cellular proteins. Cell. 1994;79(2):341–51. https://doi.org/10.1016/0092-8674(94)90202-x.
Webster KA, Graham RM, Bishopric NH. BNip3 and signal-specific programmed death in the heart. J Mol Cell Cardiol. 2005;38(1):35–45. https://doi.org/10.1016/j.yjmcc.2004.11.007.
Lee H, Paik SG. Regulation of BNIP3 in normal and cancer cells. Mol Cells. 2006;21(1):1–6.
Guan P, Lin XM, Yang SC, Guo YJ, Li WY, Zhao YS, et al. Hydrogen gas reduces chronic intermittent hypoxia-induced hypertension by inhibiting sympathetic nerve activity and increasing vasodilator responses via the antioxidation. J Cell Biochem. 2019;120(3):3998–4008. https://doi.org/10.1002/jcb.27684.
Gao XM, Dart AM, Dewar E, Jennings G, Du XJ. Serial echocardiographic assessment of left ventricular dimensions and function after myocardial infarction in mice. Cardiovasc Res. 2000;45(2):330–8. https://doi.org/10.1016/s0008-6363(99)00274-6.
Xie S, Deng Y, Pan YY, Wang ZH, Ren J, Guo XL, et al. Melatonin protects against chronic intermittent hypoxia-induced cardiac hypertrophy by modulating autophagy through the 5′ adenosine monophosphate-activated protein kinase pathway. Biochem Biophys Res Commun. 2015;464(4):975–81. https://doi.org/10.1016/j.bbrc.2015.06.149.
Xie S, Deng Y, Pan YY, Ren J, Jin M, Wang Y, et al. Chronic intermittent hypoxia induces cardiac hypertrophy by impairing autophagy through the adenosine 5′-monophosphate-activated protein kinase pathway. Arch Biochem Biophys. 2016;606:41–52. https://doi.org/10.1016/j.abb.2016.07.006.
Guan P, Sun ZM, Wang N, Zhou J, Luo LF, Zhao YS, et al. Resveratrol prevents chronic intermittent hypoxia-induced cardiac hypertrophy by targeting the PI3K/AKT/mTOR pathway. Life Sci. 2019;233:116748. https://doi.org/10.1016/j.lfs.2019.116748.
Aoyagi T, Matsui T. Phosphoinositide-3 kinase signaling in cardiac hypertrophy and heart failure. Curr Pharm Des. 2011;17(18):1818–24. https://doi.org/10.2174/138161211796390976.
Myslinski W, Rekas-Wojcik A, Dybala A, Zakrzewski M, Barud W, Prystupa A, et al. Clinical characteristics of hypertensive patients with obstructive sleep apnoea syndrome developing different types of left ventricular geometry. Biomed Res Int. 2021;2021:6631500. https://doi.org/10.1155/2021/6631500.
Dutta D, Xu J, Kim JS, Dunn WA Jr, Leeuwenburgh C. Upregulated autophagy protects cardiomyocytes from oxidative stress-induced toxicity. Autophagy. 2013;9(3):328–44. https://doi.org/10.4161/auto.22971.
Zhu H, Tannous P, Johnstone JL, Kong Y, Shelton JM, Richardson JA, et al. Cardiac autophagy is a maladaptive response to hemodynamic stress. J Clin Invest. 2007;117(7):1782–93. https://doi.org/10.1172/jci27523.
Zhang X, Li ZL, Crane JA, Jordan KL, Pawar AS, Textor SC, et al. Valsartan regulates myocardial autophagy and mitochondrial turnover in experimental hypertension. Hypertension. 2014;64(1):87–93. https://doi.org/10.1161/hypertensionaha.113.02151.
Hang T, Huang Z, Jiang S, Gong J, Wang C, Xie D, et al. Apoptosis in pressure overload-induced cardiac hypertrophy is mediated, in part, by adenine nucleotide translocator-1. Ann Clin Lab Sci. 2006;36(1):88–95.
Yang H, Xie Y, Yang D, Ren D. Oxidative stress-induced apoptosis in granulosa cells involves JNK, p53 and Puma. Oncotarget. 2017;8(15):25310–22. https://doi.org/10.18632/oncotarget.15813.
Funding
This work was supported by Natural Science Foundation of Shanxi, Grant/Award No. 201901D211496 and Applied Basic Research Youth Project of Shanxi, Grant/Award No. 201901D211497.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethics approval
All animal experiments and protocols were approved by The Second Hospital of Shanxi Medical University of Chinese Medicine Animal Care and Use Committee.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Fig. S1
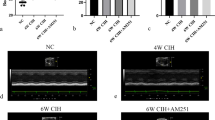
: Effects of BNIP3 knockout on chronic intermittent hypoxia (CIH)-induced left ventricle dysfunction assessed by echocardiography in CIH rats. Echocardiographic images were shown. (TIF 1597 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chi, R., Chai, C., Liu, G. et al. Chronic intermittent hypoxia-induced BNIP3 expression mitigates contractile dysfunction and myocardial injury in animal and cell model via modulating autophagy. Human Cell 36, 631–642 (2023). https://doi.org/10.1007/s13577-022-00851-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-022-00851-w