Abstract
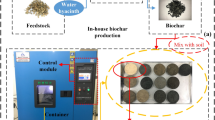
Temperature greatly determines biochar’s physicochemical characteristics during the pyrolysis of a biowaste. This study aimed to investigate how pyrolysis temperature alters the physicochemical characteristics of water hyacinth (WH) biochar as a soil amendment. WH biomass was slowly pyrolyzed at three temperatures (350, 550, and 750 °C) for 2 h. Results show that biochar yield lessened from 51.0 to 33.3% with a temperature rise. When pyrolysis temperature increased biochar’s pH (9.24–11.2), electrical conductivity (28.0–44.7 mS cm−1), liming capacity (17.7–33.0% CaCO3 equivalence), ash content (33.3–52.4%), available nutrients (Ca, Mg, K, P), surface area (1.1–29.8 m2 g−1), pore volume, C/N ratio (15.9–20.3), and water holding capacity increased. However, C, H, N, H/C (0.89–0.11) and O/C (0.62–0.49) ratios, cation exchange capacity (CEC) (44.4–2.3 cmol+kg−1), and pore width decreased. Surface functional groups shrank when pyrolysis temperature increased. As the temperature rises, WH biochar becomes structured, porous, and recalcitrant. All WH biochar samples show high alkalinity, particularly developed at 550 °C and 750 °C could replace liming materials in soil acidity alleviation. Biochar produced at 350 °C and 550 °C could improve agricultural soil fertility and nutrient retention capacity due to the lower C/N ratio, high N content, and CEC. Biochar produced at 550°C and 750 °C can sequester carbon in the soil. Biochar developed at 750 °C be applied to amend soil physical properties due to its comparably high surface area and porosity. Hence, the thermal conversion of WH biowaste to biochar helps obtain suitable biochar properties for soil amendment.






Similar content being viewed by others
Data availability
The data used in this study are available within the article.
References
Lehmann J, Joseph S (2015) Biochar for environmental management: science, technology and implementation. Routledge. https://doi.org/10.4324/9780203762264
Paz-Ferreiro J, Gascó G, Gutiérrez B, Méndez A (2011) Soil biochemical activities and the geometric mean of enzyme activities after application of sewage sludge and sewage sludge biochar to soil. Biol Fertil Soils 48:511–517. https://doi.org/10.1007/s00374-011-0644-3
Ding Y, Liu Y, Liu S, Li Z, Tan X, Huang X, Zeng G, Zhou L, Zheng B (2016) Biochar to improve soil fertility. Agron Sustain Dev 36:1–18. https://doi.org/10.1007/s13593-016-0372-z
Agegnehu G, Nelson PN, Bird MI (2016) Crop yield, plant nutrient uptake and soil physicochemical properties under organic soil amendments and nitrogen fertilization on Nitisols. Soil Tillage Res 160:1–13. https://doi.org/10.1016/j.still.2016.02.003
Yf Y, He Xh, Gao R, Hl Ma, Ys Y (2014) Effects of rice straw and its biochar addition on soil labile carbon and soil organic carbon. J Integr Agric 13:491–498. https://doi.org/10.1016/s2095-3119(13)60704-2
Pandit NR, Mulder J, Hale SE, Zimmerman AR, Pandit BH, Cornelissen G (2018) Multi-year double cropping biochar field trials in Nepal: finding the optimal biochar dose through agronomic trials and cost-benefit analysis. Sci Total Environ 637–638:1333–1341. https://doi.org/10.1016/j.scitotenv.2018.05.107
Pokharel P, Ma Z, Chang SX (2020) Biochar increases soil microbial biomass with changes in extra- and intracellular enzyme activities: a global meta-analysis. Biochar 2:65–79. https://doi.org/10.1007/s42773-020-00039-1
Karimi A, Moezzi A, Chorom M, Enayatizamir N (2020) Application of biochar changed the status of nutrients and biological activity in a calcareous soil. J Soil Sci Plant Nutr 20:450–459. https://doi.org/10.1007/s42729-019-00129-5
Verheijen FGA, Zhuravel A, Silva FC, Amaro A, Ben-Hur M, Keizer JJ (2019) The influence of biochar particle size and concentration on bulk density and maximum water holding capacity of sandy vs sandy loam soil in a column experiment. Geoderma 347:194–202. https://doi.org/10.1016/j.geoderma.2019.03.044
Baiamonte G, Crescimanno G, Parrino F, De Pasquale C (2019) Effect of biochar on the physical and structural properties of a sandy soil. CATENA 175:294–303. https://doi.org/10.1016/j.catena.2018.12.019
Atkinson CJ, Aitkenhead M (2018) How good is the evidence that soil-applied biochar improves water-holding capacity? Soil Use Manag 34:177–186. https://doi.org/10.1111/sum.12413
Hale SE, Alling V, Martinsen V, Mulder J, Breedveld GD, Cornelissen G (2013) The sorption and desorption of phosphate-P, ammonium-N and nitrate-N in cacao shell and corn cob biochars. Chemosphere 91:1612–1619. https://doi.org/10.1016/j.chemosphere.2012.12.057
Ye L, Camps-Arbestain M, Shen Q, Lehmann J, Singh B, Sabir M, Condron LM (2020) Biochar effects on crop yields with and without fertilizer: a meta-analysis of field studies using separate controls. Soil Use Manag 36:2–18. https://doi.org/10.1111/sum.12546
Uras Ü, Carrier M, Hardie AG, Knoetze JH (2012) Physico-chemical characterization of biochars from vacuum pyrolysis of South African agricultural wastes for application as soil amendments. J Anal Appl Pyrol 98:207–213. https://doi.org/10.1016/j.jaap.2012.08.007
Li S, Chen G (2018) Thermogravimetric, thermochemical, and infrared spectral characterization of feedstocks and biochar derived at different pyrolysis temperatures. Waste Manag 78:198–207. https://doi.org/10.1016/j.wasman.2018.05.048
Balmuk G, Videgain M, Manyà JJ, Duman G, Yanik J (2023) Effects of pyrolysis temperature and pressure on agronomic properties of biochar. J Anal Appl Pyrol 169. https://doi.org/10.1016/j.jaap.2023.105858
Tomczyk A, Sokołowska Z, Boguta P (2020) Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Biotechnol 19:191–215. https://doi.org/10.1007/s11157-020-09523-3
Guo J, Zheng L, Li Z, Zhou X, Cheng S, Zhang L, Zhang Q (2021) Effects of various pyrolysis conditions and feedstock compositions on the physicochemical characteristics of cow manure-derived biochar. J Clean Prod 311:127458. https://doi.org/10.1016/j.jclepro.2021.127458
Wang Y, Hu Y, Zhao X, Wang S, Xing G (2013) Comparisons of biochar properties from wood material and crop residues at different temperatures and residence times. Energy Fuels 27:5890–5899. https://doi.org/10.1021/ef400972z
Sun Y, Gao B, Yao Y, Fang J, Zhang M, Zhou Y, Chen H, Yang L (2014) Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem Eng J 240:574–578. https://doi.org/10.1016/j.cej.2013.10.081
Sohi S, Lopez-Capel E, Krull E, Bol R (2009) Biochar, climate change and soil: a review to guide future research. CSIRO Land and Water Sci. Rep 5:17–31
Zhang Z, Zhu Z, Shen B, Liu L (2019) Insights into biochar and hydrochar production and applications: a review. Energy 171:581–598. https://doi.org/10.1016/j.energy.2019.01.035
Ahmed A, Abu Bakar MS, Hamdani R, Park YK, Lam SS, Sukri RS, Hussain M, Majeed K, Phusunti N, Jamil F, Aslam M (2020) Valorization of underutilized waste biomass from invasive species to produce biochar for energy and other value-added applications. Environ Res 186:109596. https://doi.org/10.1016/j.envres.2020.109596
Villamagna AM, Murphy BR (2010) Ecological and socio-economic impacts of invasive water hyacinth (Eichhornia crassipes): a review. Freshw Biol 55:282–298. https://doi.org/10.1111/j.1365-2427.2009.02294.x
Firehun Y, Struik P, Lantinga E, Taye T (2013) Joint use of insects and fungal pathogens in the management of water hyacinth (Eichhornia crassipes): perspectives for Ethiopia. J Aquat Plant Manage 51:109–121
Wassie A, Minwuyelet M, Ayalew W, Dereje T, Woldegebrael WK, Addisalem A, Wondie E (2014) Water hyacinth coverage survey report on Lake Tana. Tech Rep Ser 1. https://www.bdu.edu.et/sites/default/files/publication/Water_Hyacinth_Lake%20Tana_Report%20Series%201.pdf
Dersseh MG, Melesse AM, Tilahun SA, Abate M, Dagnew DC (2019) Water hyacinth: review of its impacts on hydrology and ecosystem services—lessons for management of Lake Tana. Extreme hydrology and climate variability, pp 237–251. https://doi.org/10.1016/B978-0-12-815998-9.00019-1
Tewabe D, Asmare E, Zelalem W, Mohamed B (2017) Identification of impacts, some biology of water hyacinth (Eichhornia crassipes) and its management options in Lake Tana, Ethiopia. Net J Agric Sci 5:8–15
Enyew BG, Assefa WW, Gezie A (2020) Socioeconomic effects of water hyacinth (Echhornia Crassipes) in Lake Tana. PLoS One 15:e0237668. https://doi.org/10.1371/journal.pone.0237668
Gezie A, Assefa WW, Getnet B, Anteneh W, Dejen E, Mereta ST (2018) Potential impacts of water hyacinth invasion and management on water quality and human health in Lake Tana watershed, Northwest Ethiopia. Biol Invasions 20:2517–2534. https://doi.org/10.1007/s10530-018-1717-0
Maruyama DO, Si A, Sekine M, Fujita A, Habtu NG, Sato S, Toda T (2023) Enhancement of water hyacinth juice treatment in an anaerobic sequential batch reactor with coffee husk–derived biochar. Bioenergy Res. https://doi.org/10.1007/s12155-023-10593-8
Bottezini L, Dick DP, Wisniewski A, Knicker H, Carregosa ISC (2021) Phosphorus species and chemical composition of water hyacinth biochars produced at different pyrolysis temperature. Bioresour Technol Rep 14:1–8. https://doi.org/10.1016/j.biteb.2021.100684
Masto RE, Kumar S, Rout TK, Sarkar P, George J, Ram LC (2013) Biochar from water hyacinth (Eichornia crassipes) and its impact on soil biological activity. CATENA 111:64–71. https://doi.org/10.1016/j.catena.2013.06.025
Sindhu R, Binod P, Pandey A, Madhavan A, Alphonsa JA, Vivek N, Gnansounou E, Castro E, Faraco V (2017) Water hyacinth a potential source for value addition: an overview. Bioresour Technol 230:152–162. https://doi.org/10.1016/j.biortech.2017.01.035
Bordoloi S, Garg A, Sreedeep S, Lin P, Mei G (2018) Investigation of cracking and water availability of soil-biochar composite synthesized from invasive weed water hyacinth. Bioresour Technol 263:665–677. https://doi.org/10.1016/j.biortech.2018.05.011
Jutakanoke R, Intaravicha N, Charoensuksai P, Mhuantong W, Boonnorat J, Sichaem J, Phongsopitanun W, Chakritbudsabong W, Rungarunlert S (2023) Alleviation of soil acidification and modification of soil bacterial community by biochar derived from water hyacinth Eichhornia crassipes. Sci Rep 13:397. https://doi.org/10.1038/s41598-023-27557-9
Zhang F, Wang X, Yin D, Peng B, Tan C, Liu Y, Tan X, Wu S (2015) Efficiency and mechanisms of Cd removal from aqueous solution by biochar derived from water hyacinth (Eichornia crassipes). J Environ Manage 153:68–73. https://doi.org/10.1016/j.jenvman.2015.01.043
Mimmo T, Panzacchi P, Baratieri M, Davies CA, Tonon G (2014) Effect of pyrolysis temperature on miscanthus (Miscanthus × giganteus) biochar physical, chemical and functional properties. Biomass Bioenerg 62:149–157. https://doi.org/10.1016/j.biombioe.2014.01.004
Singh B, Dolk MM, Shen Q, Camps-Arbestain M (2017) Biochar pH, electrical conductivity and liming potential. In: Singh B, Camps-Arbestain M, Lehmann J (eds) Biochar: a guide to analytical methods. CRC Press/Taylor and Francis Group LLC, pp 23–38
Hien TTT, Tsubota T, Taniguchi T, Shinogi Y (2020) Enhancing soil water holding capacity and provision of a potassium source via optimization of the pyrolysis of bamboo biochar. Biochar 3:51–61. https://doi.org/10.1007/s42773-020-00071-1
Wang T, Camps-Arbestain M, Hedley M, Bishop P (2012) Predicting phosphorus bioavailability from high-ash biochars. Plant Soil 357:173–187. https://doi.org/10.1007/s11104-012-1131-9
Camps-Arbestain M, Amonette JE, Singh B, Wang T, Schmidt HP (2015) A biochar classification system and associated test methods. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science, technology and implementation. Routledge, pp 165–193
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319
Ahmed M, Nigussie A, Addisu S, Belay B, Sato S (2021) Valorization of animal bone into phosphorus biofertilizer: effects of animal species, thermal processing method, and production temperature on phosphorus availability. Soil Sci. Plant Nutr 67:471–481. https://doi.org/10.1080/00380768.2021.1945403
Li F, Shen K, Long X, Wen J, Xie X, Zeng X, Liang Y, Wei Y, Lin Z, Huang W, Zhong R (2016) Preparation and characterization of biochars from Eichornia crassipes for cadmium removal in aqueous solutions. PLoS ONE 11:e0148132. https://doi.org/10.1371/journal.pone.0148132
Mašek O, Brownsort P, Cross A, Sohi S (2013) Influence of production conditions on the yield and environmental stability of biochar. Fuel 103:151–155. https://doi.org/10.1016/j.fuel.2011.08.044
Novak JM, Lima I, Xing B, Gaskin JW, Steiner C, Das K, Ahmedna M, Rehrah D, Watts DW, Busscher WJ (2009) Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann Reg Sci 3:195–206
Reza MS, Afroze S, Bakar MSA, Saidur R, Aslfattahi N, Taweekun J, Azad AK (2020) Biochar characterization of invasive Pennisetum purpureum grass: effect of pyrolysis temperature. Biochar 2:239–251. https://doi.org/10.1007/s42773-020-00048-0
Narayanan M, Kandasamy G, Kandasamy S, Natarajan D, Devarayan K, Alsehli M, Elfasakhany A, Pugazhendhi A (2021) Water hyacinth biochar and Aspergillus niger biomass amalgamation potential in removal of pollutants from polluted lake water. J Environ Chem Eng 9:105574. https://doi.org/10.1016/j.jece.2021.105574
Dhar SA, Sakib TU, Hilary LN (2022) Effects of pyrolysis temperature on production and physicochemical characterization of biochar derived from coconut fiber biomass through slow pyrolysis process. Biomass Convers Biorefin 12:2631–2647. https://doi.org/10.1007/s13399-020-01116-y
Zhang X, Zhao B, Liu H, Zhao Y, Li L (2022) Effects of pyrolysis temperature on biochar’s characteristics and speciation and environmental risks of heavy metals in sewage sludge biochars. Environ Technol Innov 26. https://doi.org/10.1016/j.eti.2022.102288
Lataf A, Jozefczak M, Vandecasteele B, Viaene J, Schreurs S, Carleer R, Yperman J, Marchal W, Cuypers A, Vandamme D (2022) The effect of pyrolysis temperature and feedstock on biochar agronomic properties. J Anal Appl Pyrol 168:105728. https://doi.org/10.1016/j.jaap.2022.105728
Zhang X, Zhang P, Yuan X, Li Y, Han L (2020) Effect of pyrolysis temperature and correlation analysis on the yield and physicochemical properties of crop residue biochar. Bioresour Technol 296:122318. https://doi.org/10.1016/j.biortech.2019.122318
Tu P, Zhang G, Wei G, Li J, Li Y, Deng L, Yuan H (2022) Influence of pyrolysis temperature on the physicochemical properties of biochars obtained from herbaceous and woody plants. Bioresour Bioprocess 9:131. https://doi.org/10.1186/s40643-022-00618-z
Windeatt JH, Ross AB, Williams PT, Forster PM, Nahil MA, Singh S (2014) Characteristics of biochars from crop residues: potential for carbon sequestration and soil amendment. J Environ Manage 146:189–197. https://doi.org/10.1016/j.jenvman.2014.08.003
He X, Liu Z, Niu W, Yang L, Zhou T, Qin D, Niu Z, Yuan Q (2018) Effects of pyrolysis temperature on the physicochemical properties of gas and biochar obtained from pyrolysis of crop residues. Energy 143:746–756. https://doi.org/10.1016/j.energy.2017.11.062
Rodriguez JA, Filho JFL, Melo LCA, de Assis IR, de Oliveira TS (2020) Influence of pyrolysis temperature and feedstock on the properties of biochars produced from agricultural and industrial wastes. J Anal Appl Pyrol 149:104839. https://doi.org/10.1016/j.jaap.2020.104839
McBeath AV, Smernik RJ (2009) Variation in the degree of aromatic condensation of chars. Org Geochem 40:1161–1168. https://doi.org/10.1016/j.orggeochem.2009.09.006
Claoston N, Samsuri AW, Husni MHA, Amran MSM (2014) Effects of pyrolysis temperature on the physicochemical properties of empty fruit bunch and rice husk biochars. Waste Manag Res 32:331–339. https://doi.org/10.1177/0734242X14525822
Jin J, Li Y, Zhang J, Wu S, Cao Y, Liang P, Zhang J, Wong MH, Wang M, Shan S, Christie P (2016) Influence of pyrolysis temperature on properties and environmental safety of heavy metals in biochars derived from municipal sewage sludge. J Hazard Mater 320:417–426. https://doi.org/10.1016/j.jhazmat.2016.08.050
Wu W, Yang M, Feng Q, McGrouther K, Wang H, Lu H, Chen Y (2012) Chemical characterization of rice straw-derived biochar for soil amendment. Biomass Bioenerg 47:268–276. https://doi.org/10.1016/j.biombioe.2012.09.034
Nguyen BT, Lehmann J, Hockaday WC, Joseph S, Masiello CA (2010) Temperature sensitivity of black carbon decomposition and oxidation. Environ Sci Technol 44:3324–3331. https://doi.org/10.1021/es903016y
International Biochar Initiatives (IBI) (2015) Standardized product definition and product testing guidelines for biochar that is used in soil version 2.1. https://biochar-international.org/wp-content/uploads/2023/01/IBI_Biochar_Standards_V2.1_Final2.pdf
Chen D, Yin L, Wang H, He P (2014) Pyrolysis technologies for municipal solid waste: a review. Waste Manag 34:2466–2486. https://doi.org/10.1016/j.wasman.2014.08.004
Yuan JH, Xu RK, Zhang H (2011) The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour Technol 102:3488–3497. https://doi.org/10.1016/j.biortech.2010.11.018
Liu C, Ye J, Lin Y, Wu J, Price GW, Burton D, Wang Y (2020) Removal of cadmium (II) using water hyacinth (Eichhornia crassipes) biochar alginate beads in aqueous solutions. Environ Pollut 264:114785. https://doi.org/10.1016/j.envpol.2020.114785
Domingues RR, Trugilho PF, Silva CA, Melo I, Melo LCA, Magriotis ZM, Sanchez-Monedero MA (2017) Properties of biochar derived from wood and high-nutrient biomasses with the aim of agronomic and environmental benefits. PLoS ONE 12:e0176884. https://doi.org/10.1371/journal.pone.0176884
Chin-Pampillo JS, Alfaro-Vargas A, Rojas R, Giacomelli CE, Perez-Villanueva M, Chinchilla-Soto C, Alcañiz JM, Domene X (2020) Widespread tropical agrowastes as novel feedstocks for biochar production: characterization and priority environmental uses. Biomass Convers Biorefin 11:1775–1785. https://doi.org/10.1007/s13399-020-00714-0
Ding W, Dong X, Ime IM, Gao B, Ma LQ (2014) Pyrolytic temperatures impact lead sorption mechanisms by bagasse biochars. Chemosphere 105:68–74. https://doi.org/10.1016/j.chemosphere.2013.12.042
Zhao L, Cao X, Masek O, Zimmerman A (2013) Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J Hazard Mater 256–257:1–9. https://doi.org/10.1016/j.jhazmat.2013.04.015
Al-Wabel MI, Al-Omran A, El-Naggar AH, Nadeem M, Usman AR (2013) Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour Technol 131:374–379. https://doi.org/10.1016/j.biortech.2012.12.165
Cantrell KB, Hunt PG, Uchimiya M, Novak JM, Ro KS (2012) Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour Technol 107:419–428. https://doi.org/10.1016/j.biortech.2011.11.084
Joseph S, Downie A, Munroe P, Crosky A, Lehmann J (2007) Biochar for carbon sequestration, reduction of greenhouse gas emissions and enhancement of soil fertility; a review of the materials science. https://www.researchgate.net/profile/Johannes-Lehmann-5/publication/228483265_Biochar_for_Carbon_Sequestration_Reduction_of_Greenhouse_Gas_Emissions_and_Enhancement_of_Soil_Fertility_A_Review_of_the_Materials_Science/links/53f501d20cf2888a74913ba8/Biochar-for-Carbon-Sequestration-Reduction-of-Greenhouse-Gas-Emissions-and-Enhancement-of-Soil-Fertility-A-Review-of-the-Materials-Science.pdf
Gaskin JW, Steiner C, Harris K, Das KC, Bibens B (2008) Effect of low-temperature pyrolysis conditions on biochar for agricultural use. Transactions of the ASABE 51:2061–2069. https://doi.org/10.13031/2013.25409
Zornoza R, Moreno-Barriga F, Acosta JA, Munoz MA, Faz A (2016) Stability, nutrient availability and hydrophobicity of biochars derived from manure, crop residues, and municipal solid waste for their use as soil amendments. Chemosphere 144:122–130. https://doi.org/10.1016/j.chemosphere.2015.08.046
Banik C, Lawrinenko M, Bakshi S, Laird DA (2018) Impact of pyrolysis temperature and feedstock on surface charge and functional group chemistry of biochars. J Environ Qual 47:452–461. https://doi.org/10.2134/jeq2017.11.0432
Smider B, Singh B (2014) Agronomic performance of a high ash biochar in two contrasting soils. Agr Ecosyst Environ 191:99–107. https://doi.org/10.1016/j.agee.2014.01.024
Van Zwieten L, Kimber S, Morris S, Chan KY, Downie A, Rust J, Joseph S, Cowie A (2010) Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 327:235–246. https://doi.org/10.1007/s11104-009-0050-x
Singh B, Singh BP, Cowie AL (2010) Characterisation and evaluation of biochars for their application as a soil amendment. Soil Res 48:516–525. https://doi.org/10.1071/SR10058
Bolan N, Sarmah AK, Bordoloi S, Bolan S, Padhye LP, Van Zwieten L, Sooriyakumar P, Khan BA, Ahmad M, Solaiman ZM, Rinklebe J, Wang H, Singh BP, Siddique KHM (2023) Soil acidification and the liming potential of biochar. Environ Pollut 317:120632. https://doi.org/10.1016/j.envpol.2022.120632
Wang K, Peng N, Lu G, Dang Z (2020) Effects of pyrolysis temperature and holding time on physicochemical properties of swine-manure-derived biochar. Waste Biomass Valori 11:613–624. https://doi.org/10.1007/s12649-018-0435-2
Hossain MZ, Bahar MM, Sarkar B, Donne SW, Wade P, Bolan N (2021) Assessment of the fertilizer potential of biochars produced from slow pyrolysis of biosolid and animal manures. J Anal Appl Pyrol 155:105043. https://doi.org/10.1016/j.jaap.2021.105043
Jindo K, Mizumoto H, Sawada Y, Sanchez-Monedero MA, Sonoki T (2014) Physical and chemical characterization of biochars derived from different agricultural residues. Biogeosciences 11:6613–6621. https://doi.org/10.5194/bg-11-6613-2014
Chen B, Zhou D, Zhu L (2008) Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol 42:5137–5143. https://doi.org/10.1021/es8002684
Keiluweit M, Nico PS, Johnson MG, Kleber M (2010) Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ Sci Technol 44:1247–1253. https://doi.org/10.1021/es9031419
Vasseghian Y, Sezgin D, Nguyen DC, Hoang HY, Yilmaz MS (2023) A hybrid nanocomposite based on CuFe layered double hydroxide coated graphene oxide for photocatalytic degradation of trimethoprim. Chemosphere 322:138243. https://doi.org/10.1016/j.chemosphere.2023.138243
Ahmad M, Lee SS, Dou X, Mohan D, Sung JK, Yang JE, Ok YS (2012) Effects of pyrolysis temperature on soybean stover- and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour Technol 118:536–544. https://doi.org/10.1016/j.biortech.2012.05.042
Allam F, Elnouby M, El-Khatib KM, El-Badan DE, Sabry SA (2020) Water hyacinth (Eichhornia crassipes) biochar as an alternative cathode electrocatalyst in an air-cathode single chamber microbial fuel cell. Int J Hydrogen Energy 45:5911–5927. https://doi.org/10.1016/j.ijhydene.2019.09.164
Chatterjee R, Sajjadi B, Chen WY, Mattern DL, Hammer N, Raman V, Dorris A (2020) Effect of pyrolysis temperature on physicochemical properties and acoustic-based amination of biochar for efficient CO2 adsorption. Front Energy Res 8:1–18. https://doi.org/10.3389/fenrg.2020.00085
Liu X, Zhou Y, Zhou W, Li L, Huang S, Chen S (2015) Biomass-derived nitrogen self-doped porous carbon as effective metal-free catalysts for oxygen reduction reaction. Nanoscale 7:6136–6142. https://doi.org/10.1039/C5NR00013K
Vinayakumar K, Palliyarayil A, Prakash PS, Kumar NS, Sil S (2022) A facile one pot synthesis of biocarbon derived from water hyacinth and development of pellets for CO2 capture applications. Biomass Bioenerg 167:106648. https://doi.org/10.1016/j.biombioe.2022.106648
Lee JW, Kidder M, Evans BR, Paik S, Buchanan Iii AC, Garten CT, Brown RC (2010) Characterization of biochars produced from cornstovers for soil amendment. Environ Sci Technol 44:7970–7974. https://doi.org/10.1021/es101337x
Kalderis D, Kotti MS, Méndez A, Gascó G (2014) Characterization of hydrochars produced by hydrothermal carbonization of rice husk. Solid Earth 5:477–483. https://doi.org/10.5194/se-5-477-2014
Yang C, Liu J, Lu S (2021) Pyrolysis temperature affects pore characteristics of rice straw and canola stalk biochars and biochar-amended soils. Geoderma 397:115097. https://doi.org/10.1016/j.geoderma.2021.115097
Liang B, Lehmann J, Solomon D, Kinyangi J, Grossman J, O’Neill B, Skjemstad JO, Thies J, Luizão FJ, Petersen J, Neves EG (2006) Black carbon increases cation exchange capacity in soils. Soil Sci Soc Am J 70:1719–1730. https://doi.org/10.2136/sssaj2005.0383
Hu Z, Ma X, Li L (2015) Optimal conditions for the catalytic and non-catalytic pyrolysis of water hyacinth. Energy Convers Manag 94:337–344. https://doi.org/10.1016/j.enconman.2015.01.087
Nagel K, Hoilett NO, Mottaleb MA, Meziani MJ, Wistrom J, Bellamy M (2019) Physicochemical characteristics of biochars derived from corn, hardwood, miscanthus, and horse manure biomasses. Commun Soil Sci Plant Anal 50:987–1002. https://doi.org/10.1080/00103624.2019.1594881
Batista E, Shultz J, Matos TTS, Fornari MR, Ferreira TM, Szpoganicz B, de Freitas RA, Mangrich AS (2018) Effect of surface and porosity of biochar on water holding capacity aiming indirectly at preservation of the Amazon biome. Sci Rep 8:10677. https://doi.org/10.1038/s41598-018-28794-z
Mangrich A, Cardoso E, Doumer M, Romão L, Vidal M, Rigol A, Novotny E (2015) Improving the water holding capacity of soils of Northeast Brazil by biochar augmentation. In: Ahuja S, de Andrade JB, Dionysiou DD, Hristovski KD, Loganathan BG (eds) Water challenges and solutions on a global scale, ACS Publications, Washington DC, pp 339–354. https://doi.org/10.1021/bk-2015-1206.ch016A
Acknowledgements
Professor Shinjiro Sato’s lab members from the Faculty of Science and Engineering at Soka University, Japan, who helped with the biochar analysis, are highly acknowledged by the authors. The Ethiopian Lake Tana and Other Water Bodies Authority is dully acknowledged for granting access to the water hyacinth at Lake Tana.
Funding
This research was supported by Plankton Ecoengineering for Environment and Economic Transformations project, which was funded by MEXT/Japan, and the Project for Eco-Engineering for Agricultural Revitalization toward Improvement of Human Nutrition/EARTH: Water Hyacinth to Energy and Agricultural Crops funded by the Japan Science and Technology Agency/JST and Japan International Cooperation Agency/JICA under Japan Science and Technology Research Partnership for Sustainable Development/SATREPS (grant number—JPMJSA2005).
Author information
Authors and Affiliations
Contributions
Ashenafei Gezahegn: conceptualization; methodology; formal analysis and investigation; writing—original draft preparation. Yihenew G. Selassie: conceptualization; methodology; supervision; writing—review and editing. Getachew Agegnehu: conceptualization; methodology; supervision; writing—review and editing. Solomon Addisu: conceptualization; methodology; supervision; funding acquisition; resources; writing—review and editing. Fekremariam Asargew Mihretie: conceptualization; methodology; writing—review and editing. Yudai Kohira: methodology; formal analysis and investigation; writing—review and editing. Shinjiro Sato: conceptualization; methodology; funding acquisition; formal analysis and investigation; resources; supervision; writing—review and editing.
Corresponding author
Ethics declarations
Ethical
This declaration is not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gezahegn, A., G. Selassie, Y., Agegnehu, G. et al. Pyrolysis temperature changes the physicochemical characteristics of water hyacinth-based biochar as a potential soil amendment. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05338-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05338-2