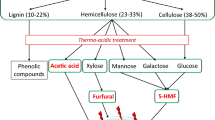
Abstract
Xylose is the second most prevalent sugar in lignocellulosic biomass after glucose. However, most of the microbial strains are unable to metabolize xylose efficiently leading to reduced ethanol yield and poor volumetric productivity. The aim of the study was to develop a co-culture strategy for efficient production of ethanol from mustard biomass. Present study covers enzymatic hydrolysis of alkali-treated mustard biomass using mixture of different cellulases. The highest reducing sugar (500.06 mg/g dry substrate) was obtained at a substrate loading of 15% (w/v), pH 5 within 48 h of incubation. Pentose sugar fermenting Fusarium incarnatum MTCC 10,292 was characterized for assimilation of different sugars and tolerance to ethanol and acetic acids. Optimization of simultaneous saccharification and fermentation process using co-culture of Fusarium incarnatum MTCC 10,292 and Saccharomyces cerevisiae MTCC 170 was carried out by using variable parameters. Within 5 days of incubation, the maximum ethanol production (3.29%, w/v) was recorded. Significant utilization of both hexose and pentose sugars for ethanol production is the major challenge for commercial lignocellulosic bioethanol production. This problem has been addressed in this study through co-culture strategy.


Similar content being viewed by others
Data availability
This submitted article contains all of the data generated or analyzed during this investigation.
References
Araújo K, Mahajan D, Kerr R, da-Silva M (2019) Global biofuels at the crossroads: an overview of technical, policy, and investment complexities: general. Oilseeds Focus 1:54–55. https://doi.org/10.3390/agriculture7040032
Isikgor FH, Becer CR (2015) Lignocellulosic biomass: a sustainable platform for the production of bio-based chemicals and polymers. Polym Chem 25:4497–4559. https://doi.org/10.1039/c5py00263j
Gupta R, Yadav G, Kumar G, Yadav A, Saini JK, Kuhad RC (2019) Second generation bioethanol production: the state of art. In: Srivastava N., Srivastava M., Mishra P., Upadhyay S., Ramteke P., Gupta V. (eds) Sustainable approaches for biofuels production technologies. biofuel and biorefinery technologies, vol 7. Springer, Cham. https://doi.org/10.1007/978-3-319-94797-6_8
Aditiya HB, Mahlia TM, Chong WT, Nur H, Sebayang AH (2016) Second generation bioethanol production: a critical review. Renew Sust Energ Rev 66:631–653. https://doi.org/10.1016/j.rser.2016.07.015
Unrean P, Srienc F (2010) Continuous production of ethanol from hexoses and pentoses using immobilized mixed cultures of Escherichia coli strains. J Biotechnol 2:215–223. https://doi.org/10.1016/j.jbiotec.2010.08.002
Sharma S, Sharma V, Kuila A (2018) Simultaneous saccharification and fermentation of corn husk by co-culture strategy. J Pet Environ Biotechnol 360:2. https://doi.org/10.4172/2157-7463.1000360
Farias D, Maugeri-Filho F (2019) Co-culture strategy for improved 2G bioethanol production using a mixture of sugarcane molasses and bagasse hydrolysate as substrate. Biochem Eng J 147:29–38. https://doi.org/10.1016/j.bej.2019.03.020
Karagöz P, Özkan M (2014) Ethanol production from wheat straw by Saccharomyces cerevisiae and Scheffersomyces stipitis co-culture in batch and continuous system. Bioresource Technol 158:286–293. https://doi.org/10.1016/j.biortech.2014.02.022
Agarwal M, Dinker A (2013) Application of consolidate enzymatic system of Fusarium and Saccharomyces to enhance the production of ethanol from spent grain. J Renew Sust Energ 5:053112. https://doi.org/10.1063/1.4821518
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D, Crocker D (2012) Determination of structural carbohydrates and lignin in biomass, Report NREL/TP-510-42618. National Renewable Energy Laboratory, Golden, CO, USA. https://www.nrel.gov/docs/gen/fy13/42618.pdf
Sharma S, Kuila A, Sharma V (2017) Enzymatic hydrolysis of thermochemically pretreated biomass using a mixture of cellulolytic enzymes produced from different fungal sources. Clean Technol Environ Policy 5:1577–1584. https://doi.org/10.1007/s10098-017-1346-9
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 2:257–268. https://doi.org/10.1515/iupac.59.0006
Hall G, Reuter WM (2007) HPLC analysis for the monitoring of fermentation broth during ethanol production as a biofuel application brief, Perkin Elmer. http://www.perkinelmer.com/pdfs/downloads/ABR_EthanolAsBiofuelbyHPLCAppBrief.pdf
Liesche J, Marek M, Günther-Pomorski T (2015) Cell wall staining with trypan blue enables quantitative analysis of morphological changes in yeast cell. Front Microbiol 6:107. https://doi.org/10.3389/fmicb.2015.00107
Wanderley MCA, Soares ML, Gouveia ER (2014) Selection of inoculum size and Saccharomyces cerevisiae strain for ethanol production in simultaneous saccharification and fermentation (SSF) of sugarcane bagasse. Afr J Biotechnol 13:2762–2765. https://doi.org/10.5897/AJB2013.13179
Gao X, Kumar R, Singh S, Simmons BA, Balan V, Dale BE, Wyman CE (2014) Comparison of enzymatic reactivity of corn stover solids prepared by dilute acid, AFEXTM, and ionic liquid pretreatments. Biotechnol Biofuels 1:71. https://doi.org/10.1201/b18437-15
Toquero C, Bolado S (2014) Effect of four pretreatments on enzymatic hydrolysis and ethanol fermentation of wheat straw. Influence of inhibitors and washing. Bioresource Technol 157:68–76. https://doi.org/10.1016/j.biortech.2014.01.090
Zhang YH, Lynd LR (2004) Toward an aggregated understanding of enzymatic hydrolysis of cellulose: noncomplexed cellulase systems. Biotechnol Bioeng 7:797–824. https://doi.org/10.1002/bit.20282
Mathew AK, Chaney K, Crook M, Humphries AC (2011) Alkaline pre-treatment of oilseed rape straw for bioethanol production: evaluation of glucose yield and pre-treatment energy consumption. Bioresource Technol 11:6547–6553. https://doi.org/10.1016/j.biortech.2011.03.067
Ethaib S, Omar R, Mazlina MKS, Radiah ABD (2020) Evaluation of the interactive effect pretreatment parameters via three types of microwave-assisted pretreatment and enzymatic hydrolysis on sugar yield. Processes 7:787. https://doi.org/10.3390/pr8070787
Wen Z, Liao W, Chen S (2004) Hydrolysis of animal manure lignocellulosics for reducing sugar production. Bioresource Technol 1:31–39. https://doi.org/10.1016/s0960-8524(03)00166-4
Zhang Y, Yang J, Luo L, Wang E, Wang R, Liu L, Liu J, Yuan H (2020) Low-cost cellulase-hemicellulase mixture secreted by Trichoderma harzianum EM0925 with complete saccharification efficacy of lignocellulose. Int J Mol Sci 2:371. https://doi.org/10.3390/ijms21020371
Kumar V, Patel SK, Gupta RK, Otari SV, Gao H, Lee JK, Zhang L (2019) Enhanced saccharification and fermentation of rice straw by reducing the concentration of phenolic compounds using an immobilized enzyme cocktail. Biotechnol J 6:1800468. https://doi.org/10.1002/biot.201800468
Rech FR, Fontana RC, Rosa CA, Camassola M, Ayub MAZ, Dillon AJP (2019) Fermentation of hexoses and pentoses from sugarcane bagasse hydrolysates into ethanol by Spathaspora hagerdaliae. Bioprocess Biosyst Eng 42:83–92. https://doi.org/10.1007/s00449-018-2016-y
Martins GM, Bocchini-Martins DA, Bezzerra-Bussoli C, Pagnocca FC, Boscolo M, Monteiro DA, Silva R, Gomes E (2018) The isolation of pentose-assimilating yeasts and their xylose fermentation potential. Braz J Microbiol 49:162–168. https://doi.org/10.1016/j.bjm.2016.11.014
Sree NK, Sridhar M, Suresh K, Banat IM, Rao LV (2000) Isolation of thermotolerant, osmotolerant, flocculating Saccharomyces cerevisiae for ethanol production. Bioresource Technol 1:43–46. https://doi.org/10.1016/s0140-6701(00)90852-x
Xiros C, Vafiadi C, Paschos T, Christakopoulos P (2011) Toxicity tolerance of Fusarium oxysporum towards inhibitory compounds formed during pretreatment of lignocellulosic materials. J Chem Technol Biotechnol 86:223–230. https://doi.org/10.1002/jctb.2499
Ueno R, Urano N, Kimura S (2001) Characterization of thermotolerant, fermentative yeasts from hot spring drainage. Fish Sci 1:138–145. https://doi.org/10.1046/j.1444-2906.2001.00210.x
Zha Y, Muilwijk B, Coulier L, Punt PJ (2012) Inhibitory compounds in lignocellulosic biomass hydrolysates during hydrolysate fermentation processes. J Bioprocess Biotech 1:112–122. https://doi.org/10.4172/2155-9821.1000112
Fakruddin M, Quayum MA, Ahmed MM, Naiyyam C (2012) Analysis of key factors affecting ethanol production by Saccharomyces cerevisiae IFST-072011. Biotechnology 4:248–252. https://doi.org/10.3923/biotech.2012.248.252
Thanonkeo P, Chamnipa N, Thanonkeo S (2014) Isolation and selection of thermo tolerant yeast for ethanol production at high temperature. In: The 26th Annual Meeting of the Thai Society of Biotechnology and International Conference, pp 65–69
Boudjema K, Fazouane-naimi F, Hellal A (2016) Isolation, identification of yeast strains producing bioethanol and improvement of bioethanol production on cheese whey. Turkish J Biochem 41:157–166. https://doi.org/10.1515/tjb-2016-0026
Ekunsanmi TJ, Odunfa SA (1990) Ethanol tolerance, sugar tolerance and invertase activities of some yeast strains isolated from steep water of fermenting cassava tubers. J Appl Bacteriol 5:672–675. https://doi.org/10.1111/j.1365-2672.1990.tb01561.x
Logothetis S, Walker G, Nerantzis ET (2007) Effect of salt hyperosmotic stress on yeast cell viability. Proceedings for Natural Sciences of Matica Srpska Novi Sad 113:271–284. https://doi.org/10.2298/zmspn0713271l
Zhao XQ, Bai FW (2009) Mechanisms of yeast stress tolerance and its manipulation for efficient fuel ethanol production. J Biotechnol 1:23–30. https://doi.org/10.1016/j.jbiotec.2009.05.001
Sutticha N, Thammasittirong T, Thammasittirong T, Malee S (2013) Improvement of ethanol production by ethanol-tolerant Saccharomyces cerevisiae UVNR56. Springer plus 1:583. https://doi.org/10.1186/2193-1801-2-583
Moremi ME, Jansen-Van-Rensburg EL, La-Grange DC (2020) The improvement of bioethanol production by pentose-fermenting yeasts isolated from herbal preparations, the gut of dung beetles, and marula wine. Int J Microbiol 2020:1–13. https://doi.org/10.1155/2020/5670936
Balakumar S, Arasaratnam V (2012) Osmo-, thermo-and ethanol-tolerances of Saccharomyces cerevisiae S1. Braz J Microbiol 1:157–166. https://doi.org/10.1590/s1517-83822012000100017
Morales P, Gentina JC, Aroca GE, Mussatto SI (2017) Development of an acetic acid tolerant Spathaspora passalidarum strain through evolutionary engineering with resistance to inhibitors compounds of autohydrolysate of Eucalyptus globulus. Ind Crop Prod 106:5–11. https://doi.org/10.1016/j.indcrop.2016.12.023
Naseeruddin S, Desai S, Rao LV (2021) Co-culture of Saccharomyces cerevisiae (VS3) and Pichia stipitis (NCIM 3498) for bioethanol production using concentrated Prosopis juliflora acid hydrolysate. 3 Biotech 11:1–12. https://doi.org/10.1007/s13205-020-02595-6
Mishra A, Ghosh S (2020) Saccharification of kans grass biomass by a novel fractional hydrolysis method followed by co-culture fermentation for bioethanol production. Renew Energ 146:750–759. https://doi.org/10.1016/j.renene.2019.07.016
Chohan NA, Aruwajoye GS, Sewsynker-Sukai Y, Gueguim Kana EB (2020) Valorisation of potato peel wastes for bioethanol production using simultaneous saccharification and fermentation: Process optimization and kinetic assessment. Renew Energ 146:1031–1040. https://doi.org/10.1016/j.renene.2019.07.042
Acknowledgements
The authors thank Ranjeet kaur for the critical reading of this manuscript.
Funding
This work was funded by the Department of Biotechnology (DBT), New Delhi (Grant No. BT/PR31154/PBD/26/762/2019).
Author information
Authors and Affiliations
Contributions
AK conceived the project. SP and Ritika performed all the work related to present study. AK, Eduardo DP, AARD, MAR, RKB, and AK planned the experiments. AK, AK, EDP, AARD, MAR, and RKB wrote the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ritika, Pant, S., Komesu, A. et al. Simultaneous fermentation of glucose and xylose by using co-culture of S. cerevisiae and a potential robust pentose fermenting fungi (Fusarium incarnatum). Biomass Conv. Bioref. 13, 8089–8100 (2023). https://doi.org/10.1007/s13399-021-01801-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-01801-6