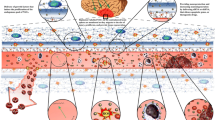
Abstract
Nanozymes are a new kind of material which has been applied since the beginning of this century, and its birth has promoted the development of chemistry, materials science, and biology. Nanozymes can be used as a substitute for natural enzyme and has a wide range of applications; therefore, it has attracted extensive attention from all sectors of the community, and the number of studies has constantly increasing. In this paper, we introduced the outstanding achievements in the field of nanozymes in recent years from the main function, the construction of nanozyme-based biosensors, and the treatment of ischemic stroke, and we also illustrated the internal mechanism and the catalytic principle. In the end, the obstacles and challenges in the future development of nanozymes were proposed.

Similar content being viewed by others
Abbreviations
- AchE:
-
acetylcholinesterase
- ACP:
-
acid phosphatase
- ALP:
-
alkaline phosphatase
- BBB:
-
blood-brain barrier
- C60 :
-
fullerene
- CAT:
-
catalase
- C-dots:
-
carbon nanodots
- CEA:
-
carcinoembryonic antigen
- ChI:
-
choline iodide
- CL:
-
chemiluminescence
- DA:
-
dopamine
- Eda:
-
edaravone
- GDYO:
-
graphdiyne oxide
- GO-COOH:
-
carboxyl-modified graphene oxide
- GOD:
-
glucose oxidase
- GOQD:
-
graphene oxide quantum dot
- GPx:
-
glutathione peroxidase
- GQD:
-
graphene quantum dots
- GSHOX:
-
glutathione oxidase
- H2O2 :
-
hydrogen peroxide
- HRP:
-
horseradish peroxidase
- MOF:
-
metal-organic framework
- NPs:
-
nanoparticles
- OTA:
-
ochratoxin A
- OXD:
-
oxidase
- oxTMB:
-
oxidation TMB
- pDA:
-
polydopamine
- POD:
-
peroxidase
- PTT:
-
photothermal therapy
- RONs:
-
reactive nitrogen species
- ROS:
-
reactive oxygen species
- SERS:
-
surface-enhanced Raman scattering
- SOD:
-
superoxide dismutase
- TA:
-
terephthalic acid
- TMB:
-
3,3’,5,5’-tetramethylbenzidine
- UDG:
-
uracil DNA glycosylase
References
Wu Y, Xu W, Jiao L, et al. Defect engineering in nanozymes. Mater Today. 2022;52:327–47. https://doi.org/10.1016/j.mattod.2021.10.032.
Zhang X, Wu D, Zhou X, et al. Recent progress in the construction of nanozyme-based biosensors and their applications to food safety assay. Trends Analyt Chem. 2019;121:115668. https://doi.org/10.1016/j.trac.2019.115668.
Gao L, Zhuang J, Nie L, et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat Nanotechnol. 2007;2:577–83. https://doi.org/10.1038/nnano.2007.260.
Wang GL, Jin LY, Dong YM, et al. Intrinsic enzyme mimicking activity of gold nanoclusters upon visible light triggering and its application for colorimetric trypsin detection. Biosens Bioelectron. 2015;64:523–9. https://doi.org/10.1016/j.bios.2014.09.071.
Kumawat M, Madhyastha H, Singh M, et al. Functional silver nanozymes regulate cell inflammatory cytokines expression in mouse macrophages. Colloids Surf A Physicochem Eng Asp. 2022;650. https://doi.org/10.1016/j.colsurfa.2022.129294.
Ma Z, Wu L, Han K, et al. Pt nanozyme for O2 self-sufficient, tumor-specific oxidative damage and drug resistance reversal. Nanoscale Horiz. 2019;4:1124–31. https://doi.org/10.1039/c9nh00088g.
Chang M, Hou Z, Wang M, et al. Single-atom pd nanozyme for ferroptosis-boosted mild-temperature photothermal therapy. Angew Chem Int Ed Engl. 2021;60:12971–9. https://doi.org/10.1002/anie.202101924.
Rai M, Pandit R, Paralikar P, et al. Copper and copper nanoparticles: role in management of insect-pests and pathogenic microbes. Nanotechnol Rev. 2018;7:303–15. https://doi.org/10.1515/ntrev-2018-0031.
Yan BC, Cao J, Liu J, et al. Dietary Fe3O4 nanozymes prevent the injury of neurons and blood-brain barrier integrity from cerebral ischemic stroke. ACS Biomater Sci Eng. 2021;7:299–310. https://doi.org/10.1021/acsbiomaterials.0c01312.
Feng N, Liu Y, Dai X, et al. Advanced applications of cerium oxide based nanozymes in cancer. RSC Adv. 2022;12:1486–93. https://doi.org/10.1039/d1ra05407d.
Karim MN, Singh M, Weerathunge P, et al. Visible-light-triggered reactive-oxygen-species-mediated antibacterial activity of peroxidase-mimic CuO nanorods. ACS Appl Nano Mater. 2018;1:1694–704. https://doi.org/10.1021/acsanm.8b00153.
Cheng Y, Cheng C, Yao J, et al. Mn3O4 nanozyme for inflammatory bowel disease therapy. Adv Ther. 2021;4:2100081. https://doi.org/10.1002/adtp.202100081.
Ray C, Dutta S, Sarkar S, et al. Intrinsic peroxidase-like activity of mesoporous nickel oxide for selective cysteine sensing. J Mater Chem B. 2014;2:6097–105. https://doi.org/10.1039/c4tb00968a.
Ghosh S, Roy P, Karmodak N, et al. Nanoisozymes: crystal-facet-dependent enzyme-mimetic activity of V2O5 nanomaterials. Angew Chem Int Ed Engl. 2018;57:4510–5. https://doi.org/10.1002/anie.201800681.
Ma W, Xue Y, Guo S, et al. Graphdiyne oxide: a new carbon nanozyme. Chem Commun (Camb). 2020;56:5115–8. https://doi.org/10.1039/d0cc01840f.
Sun H, Zhao A, Gao N, et al. Deciphering a nanocarbon-based artificial peroxidase: chemical identification of the catalytically active and substrate-binding sites on graphene quantum dots. Angew Chem Int Ed Engl. 2015;54:7176–80. https://doi.org/10.1002/anie.201500626.
Ali SS, Hardt JI, Quick KL, et al. A biologically effective fullerene (C60) derivative with superoxide dismutase mimetic properties. Free Radic Biol Med. 2004;37:1191–202. https://doi.org/10.1016/j.freeradbiomed.2004.07.002.
Ren C, Hu X, Zhou Q. Graphene oxide quantum dots reduce oxidative stress and inhibit neurotoxicity in vitro and in vivo through catalase-like activity and metabolic regulation. Adv Sci. 2018;5:1700595. https://doi.org/10.1002/advs.201700595.
Song Y, Qu K, Zhao C, et al. Graphene oxide: intrinsic peroxidase catalytic activity and its application to glucose detection. Adv Mater. 2010;22:2206–10. https://doi.org/10.1002/adma.200903783.
Shi W, Wang Q, Long Y, et al. Carbon nanodots as peroxidase mimetics and their applications to glucose detection. Chem Commun (Camb). 2011;47:6695–7. https://doi.org/10.1039/c1cc11943e.
Xi J, Wei G, An L, et al. Copper/carbon hybrid nanozyme: tuning catalytic activity by the copper state for antibacterial therapy. Nano Lett. 2019;19:7645–54. https://doi.org/10.1021/acs.nanolett.9b02242.
Wei F, Cui X, Wang Z, et al. Recoverable peroxidase-like fe3o4@MoS2-Ag nanozyme with enhanced antibacterial ability. Chem Eng J. 2021;408:127240. https://doi.org/10.1016/j.cej.2020.127240.
Lu C, Liu X, Li Y, et al. Multifunctional janus hematite-silica nanoparticles: mimicking peroxidase-like activity and sensitive colorimetric detection of glucose. ACS Appl Mater Interfaces. 2015;7:15395–402. https://doi.org/10.1021/acsami.5b03423.
Zhuang Z, Zhang C, Yu Z, et al. Turn-on colorimetric detection of hydroquinone based on Au/CuO nanocomposite nanozyme. Mikrochim Acta. 2022;189:293. https://doi.org/10.1007/s00604-022-05384-5.
Huang Y, Liu Z, Liu C, et al. Self-assembly of multi-nanozymes to mimic an intracellular antioxidant defense system. Angew Chem Int Ed Engl. 2016;55:6646–50. https://doi.org/10.1002/anie.201600868.
Liu J, Hu X, Hou S, et al. Au@Pt core/shell nanorods with peroxidase- and ascorbate oxidase-like activities for improved detection of glucose. Sens Actuators B Chem. 2012;166-7:708–14. https://doi.org/10.1016/j.snb.2012.03.045.
Chen Q, Liang C, Zhang X, et al. High oxidase-mimic activity of Fe nanoparticles embedded in an N-rich porous carbon and their application for sensing of dopamine. Talanta. 2018;182:476–83. https://doi.org/10.1016/j.talanta.2018.02.032.
Ma Y, Tian Z, Zhai W, et al. Insights on catalytic mechanism of ceo2 as multiple nanozymes. Nano Res. 2022:1–15. https://doi.org/10.1007/s12274-022-4666-y.
Liao X, Xu Q, Sun H, et al. Plasmonic nanozymes: localized surface plasmonic resonance regulates reaction kinetics and antibacterial performance. J Phys Chem Lett. 2022;13:312–23. https://doi.org/10.1021/acs.jpclett.1c03804.
Rostami S, Mehdinia A, Jabbari A. Intrinsic peroxidase-like activity of graphene nanoribbons for label-free colorimetric detection of dopamine. Mater Sci Eng C Mater Biol Appl. 2020;114:111034. https://doi.org/10.1016/j.msec.2020.111034.
Chen K, Sun S, Wang J, et al. Catalytic nanozymes for central nervous system disease. Coord Chem Rev. 2021;432:213751. https://doi.org/10.1016/j.ccr.2020.213751.
Singh N. Antioxidant metal oxide nanozymes: role in cellular redox homeostasis and therapeutics. Pure Appl Chem. 2021;93:187–205. https://doi.org/10.1515/pac-2020-0802.
Xiong B, Xu R, Zhou R, et al. Preventing UV induced cell damage by scavenging reactive oxygen species with enzyme-mimic Au-Pt nanocomposites. Talanta. 2014;120:262–7. 16/j.talanta.2013.12.020
Cao Y, Liu J, Zou L, et al. Ratiometric fluorescence sensing of glutathione by using the oxidase-mimicking activity of MnO2 nanosheet. Anal Chim Acta. 2021;1145:46–51. https://doi.org/10.1016/j.aca.2020.12.019.
Huang Y, Ren J, Qu X. Nanozymes: classification, catalytic mechanisms, activity regulation, and applications. Chem Rev. 2019;119:4357–412. https://doi.org/10.1021/acs.chemrev.8b00672.
Naghavi M, Abajobir AA, Abbafati C, et al. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2017;390:1151–210. https://doi.org/10.1016/s0140-6736(17)32152-9.
Vanova V, Mitrevska K, Milosavljevic V, et al. Peptide-based electrochemical biosensors utilized for protein detection. Biosens Bioelectron. 2021;180:113087. https://doi.org/10.1016/j.bios.2021.113087.
Ashrafi AM, Bytesnikova Z, Barek J, et al. A critical comparison of natural enzymes and nanozymes in biosensing and bioassays. Biosens Bioelectron. 2021;192:113494. https://doi.org/10.1016/j.bios.2021.113494.
Xue Q, Li X, Peng Y, et al. Polyethylenimine-stabilized silver nanoclusters act as an oxidoreductase mimic for colorimetric determination of chromium(vi). Mikrochim Acta. 2020;187:263. https://doi.org/10.1007/s00604-020-04232-8.
Zhou X, Wang M, Wang M, et al. Nanozyme-based detection of alkaline phosphatase. ACS Appl Nano Mater. 2021;4:7888–96. https://doi.org/10.1021/acsanm.1c01220.
Ma Q, Qiao J, Liu Y, et al. Colorimetric monitoring of serum dopamine with promotion activity of gold nanocluster-based nanozymes. Analyst. 2021;146:6615–20. https://doi.org/10.1039/d1an01511g.
Liu Q, Tian J, Liu J, et al. Modular assembly of tumor-penetrating and oligomeric nanozyme based on intrinsically self-assembling protein nanocages. Adv Mater. 2021;33:e2103128. https://doi.org/10.1002/adma.202103128.
Boguniewicz-Zablocka J, Klosok-Bazan I, Naddeo V. Water quality and resource management in the dairy industry. Environ Sci Pollut Res Int. 2019;26:1208–16. https://doi.org/10.1007/s11356-017-0608-8.
Durán N, Esposito E. Potential applications of oxidative enzymes and phenoloxidase-like compounds in wastewater and soil treatment: a review. Appl Catal B. 2000;28:83–99. https://doi.org/10.1016/s0926-3373(00)00168-5.
Klibanov AM, Morris ED. Horseradish peroxidase for the removal of carcinogenic aromatic amines from water. Enzyme Microb. Technol. 1981;3:119–22. https://doi.org/10.1016/0141-0229(81)90069-7.
Wang J, Huang R, Qi W, et al. Construction of a bioinspired laccase-mimicking nanozyme for the degradation and detection of phenolic pollutants. Appl Catal B. 2019;254:452–62. https://doi.org/10.1016/j.apcatb.2019.05.012.
Wang FM, Zhang Y, Liu ZW, et al. Mesoporous encapsulated nanozyme decontaminating two kinds of wastewater and avoiding secondary pollution. Nanoscale. 2020;12:14465–71. https://doi.org/10.1039/d0nr03217d.
Luo Q, Li J, Wang W, et al. Transition metal engineering of molybdenum disulfide nanozyme for biomimicking anti-biofouling in seawater. ACS Appl Mater Interfaces. 2022;14:14218–25. https://doi.org/10.1021/acsami.2c00172.
Fu T, Xu C, Guo R, et al. Zeolitic imidazolate framework-90 nanoparticles as nanozymes to mimic organophosphorus hydrolase. ACS Appl Nano Mater. 2021;4:3345–50. https://doi.org/10.1021/acsanm.1c00540.
Peng M, Zhao Z, Liang Z. Biodegradation of ochratoxin A and ochratoxin B by brevundimonas naejangsanensis isolated from soil. Food Control. 2022;133. https://doi.org/10.1016/j.foodcont.2021.108611.
Tian F, Zhou J, Jiao B, et al. A nanozyme-based cascade colorimetric aptasensor for amplified detection of ochratoxin a. Nanoscale. 2019;11:9547–55. https://doi.org/10.1039/c9nr02872b.
Wang H, Li P, Yu D, et al. Unraveling the enzymatic activity of oxygenated carbon nanotubes and their application in the treatment of bacterial infections. Nano Lett. 2018;18:3344–51. https://doi.org/10.1021/acs.nanolett.7b05095.
Yu G, Cheng Y, Duan Z. Research progress of polymers/inorganic nanocomposite electrical insulating materials. Molecules. 2022;27(22):7867. https://doi.org/10.3390/molecules27227867.
Holzinger M, Le Goff A, Cosnier S. Synergetic effects of combined nanomaterials for biosensing applications. Sensors (Basel). 2017;17(5):1010. https://doi.org/10.3390/s17051010.
Liang X, Li N, Zhang R, et al. Carbon-based sers biosensor: from substrate design to sensing and bioapplication. NPG Asia Mater. 2021:13. https://doi.org/10.1038/s41427-020-00278-5.
Zhao Y, Li L, Ma R, et al. A competitive colorimetric aptasensor transduced by hybridization chain reaction-facilitated catalysis of aunps nanozyme for highly sensitive detection of saxitoxin. Anal Chim Acta. 2021;1173:338710. https://doi.org/10.1016/j.aca.2021.338710.
Xue L, Jin N, Guo R, et al. Microfluidic colorimetric biosensors based on MnO2 nanozymes and convergence-divergence spiral micromixers for rapid and sensitive detection of salmonella. ACS Sens. 2021;6:2883–92. https://doi.org/10.1021/acssensors.1c00292.
Chen M, Sun L, Ding Y, et al. N,n′-di-carboxymethyl perylene diimide functionalized magnetic nanocomposites with enhanced peroxidase-like activity for colorimetric sensing of H2O2 and glucose. New J Chem. 2017;41:5853–62. https://doi.org/10.1039/c7nj00292k.
Lu D, Li J, Wu Z, et al. High-activity daisy-like zeolitic imidazolate framework-67/reduced grapheme oxide-based colorimetric biosensor for sensitive detection of hydrogen peroxide. J Colloid Interface Sci. 2022;608:3069–78. https://doi.org/10.1016/j.jcis.2021.11.034.
Song C, Liu H, Zhang L, et al. Fes nanoparticles embedded in 2D carbon nanosheets as novel nanozymes with peroxidase-like activity for colorimetric and fluorescence assay of H2O2 and antioxidant capacity. Sens Actuators B Chem. 2022;353. https://doi.org/10.1016/j.snb.2021.131131.
Qu H, Fan C, Chen M, et al. Recent advances of fluorescent biosensors based on cyclic signal amplification technology in biomedical detection. J Nanobiotechnology. 2021;19:403. https://doi.org/10.1186/s12951-021-01149-z.
Guo J, Wu S, Wang Y, et al. A label-free fluorescence biosensor based on a bifunctional MIL-101(Fe) nanozyme for sensitive detection of choline and acetylcholine at nanomolar level. Sens Actuators B Chem. 2020:312. https://doi.org/10.1016/j.snb.2020.128021.
Zou W, Liu Y, Li R, et al. Ingenious multifunctional MnO2 quantum dot nanozymes with superior catechol oxidase-like activity for highly selective sensing of redox-active dopamine based on an interfacial passivation strategy. ACS Sustain Chem Eng. 2022;10:10057–67. https://doi.org/10.1021/acssuschemeng.2c02981.
Li S, Hu X, Chen Q, et al. Introducing bifunctional metal-organic frameworks to the construction of a novel ratiometric fluorescence sensor for screening acid phosphatase activity. Biosens Bioelectron. 2019;137:133–9. https://doi.org/10.1016/j.bios.2019.05.010.
Khoshfetrat SM, Fasihi K, Moradnia F, et al. A label-free multicolor colorimetric and fluorescence dual mode biosensing of HIV-1 DNA based on the bifunctional NiFe2O4@UiO-66 nanozyme. Anal Chim Acta. 2023;1252:341073. https://doi.org/10.1016/j.aca.2023.341073.
Mao X, Lu Y, Zhang X, et al. Beta-cyclodextrin functionalization of metal-organic framework MOF-235 with excellent chemiluminescence activity for sensitive glucose biosensing. Talanta. 2018;188:161–7. https://doi.org/10.1016/j.talanta.2018.05.077.
Zhang X, Li G, Wu D, et al. Recent progress in the design fabrication of metal-organic frameworks-based nanozymes and their applications to sensing and cancer therapy. Biosens Bioelectron. 2019;137:178–98. https://doi.org/10.1016/j.bios.2019.04.061.
Halawa MI, Xia Q, Li BS. An ultrasensitive chemiluminescent biosensor for tracing glutathione in human serum using BSA@AuNCs as a peroxidase-mimetic nanozyme on a luminol/artesunate system. J Mater Chem B. 2021;9:8038–47. https://doi.org/10.1039/d1tb01343b.
Li J, Cao Y, Hinman SS, et al. Efficient label-free chemiluminescent immunosensor based on dual functional cupric oxide nanorods as peroxidase mimics. Biosens Bioelectron. 2018;100:304–11. https://doi.org/10.1016/j.bios.2017.09.011.
Montali L, Calabretta MM, Lopreside A, et al. Multienzyme chemiluminescent foldable biosensor for on-site detection of acetylcholinesterase inhibitors. Biosens Bioelectron. 2020;162:112232. https://doi.org/10.1016/j.bios.2020.112232.
Lin Y, Wu L, Huang Y, et al. Positional assembly of hemin and gold nanoparticles in graphene–mesoporous silica nanohybrids for tandem catalysis. Chem Sci. 2015;6:1272–6. https://doi.org/10.1039/c4sc02714k.
Khoshfetrat SM, Khoshsafar H, Afkhami A, et al. Enhanced visual wireless electrochemiluminescence immunosensing of prostatespecific antigen based on the luminol loaded into MIL-53(Fe)-NH2 accelerator and hydrogen evolution reaction mediation. Anal Chem. 2019;91:6383–90. https://doi.org/10.1021/acs.analchem.9b01506.
Lv W, Ye J, Yuan Z, et al. Recent advances in electrochemiluminescence-based simultaneous detection of multiple targets. TrAC Trends Anal Chem. 2020;123:115767. https://doi.org/10.1016/j.trac.2019.115767.
Khoshfetrat SM, Hashemi P, Afkhami A, et al. Cascade electrochemiluminescence-based integrated graphitic carbon nitride-encapsulated metal-organic framework nanozyme for prostate-specific antigen biosensing. Sens Actuators B Chem. 2021;348:130658. https://doi.org/10.1016/j.snb.2021.130658.
Bizzotto D, Burgess IJ, Doneux T, et al. Beyond simple cartoons: challenges in characterizing electrochemical biosensor interfaces. ACS Sens. 2018;3:5–12. https://doi.org/10.1021/acssensors.7b00840.
Khanmohammadi A, Aghaie A, Vahedi E, et al. Electrochemical biosensors for the detection of lung cancer biomarkers: a review. Talanta. 2020;206:120251. https://doi.org/10.1016/j.talanta.2019.120251.
Uniyal S, Sharma RK. Technological advancement in electrochemical biosensor based detection of organophosphate pesticide chlorpyrifos in the environment: a review of status and prospects. Biosens Bioelectron. 2018;116:37–50. https://doi.org/10.1016/j.bios.2018.05.039.
Li X, Li X, Li D, et al. Electrochemical biosensor for ultrasensitive exosomal miRNA analysis by cascade primer exchange reaction and MOF@Pt@MOF nanozyme. Biosens Bioelectron. 2020;168:112554. https://doi.org/10.1016/j.bios.2020.112554.
Li Y, Zhang C, He Y, et al. A generic and non-enzymatic electrochemical biosensor integrated molecular beacon-like catalyzed hairpin assembly circuit with MOF@Au@G-triplex/hemin nanozyme for ultrasensitive detection of miR-721. Biosens Bioelectron. 2022;203:114051. https://doi.org/10.1016/j.bios.2022.114051.
Liu T, Li Z, Chen M, et al. Sensitive electrochemical biosensor for uracil-DNA glycosylase detection based on self-linkable hollow Mn/Ni layered doubled hydroxides as oxidase-like nanozyme for cascade signal amplification. Biosens Bioelectron. 2021;194:113607. https://doi.org/10.1016/j.bios.2021.113607.
Yu R, Xue J, Wang Y, et al. Novel Ti3C2Tx MXene nanozyme with manageable catalytic activity and application to electrochemical biosensor. J Nanobiotechnology. 2022;20:119. https://doi.org/10.1186/s12951-022-01317-9.
Matsuhisa N, Chen X, Bao Z, et al. Materials and structural designs of stretchable conductors. Chem Soc Rev. 2019;48:2946–66. https://doi.org/10.1039/c8cs00814k.
Wu Y, Chen JY, He WM. Surface-enhanced Raman spectroscopy biosensor based on silver nanoparticles@metal-organic frameworks with peroxidase-mimicking activities for ultrasensitive monitoring of blood cholesterol. Sens Actuators B Chem. 2022;365. https://doi.org/10.1016/j.snb.2022.131939.
Huang S, Wu C, Wang Y, et al. Ag/TiO2 nanocomposites as a novel SERS substrate for construction of sensitive biosensor. Sens Actuators B Chem. 2021;339:129843. https://doi.org/10.1016/j.snb.2021.129843.
Tang R, Xia X, Zhang X, et al. Synergistic function of au NPs/GeO2 nanozymes with enhanced peroxidase-like activity and SERS effect to detect choline iodide. Spectrochim Acta A Mol Biomol Spectrosc. 2022;266:120467. https://doi.org/10.1016/j.saa.2021.120467.
Tan Y, Jiang H, Wang B, et al. MoS2-based composite nanozymes with superior peroxidase-like activity for ultrasensitive SERS detection of glucose. New J Chem. 2021;45:19593–604. https://doi.org/10.1039/d1nj02451e.
Banerjee A, McCullough LD. Sex-specific immune responses in stroke. Stroke. 2022;53(5):1449–59. https://doi.org/10.1161/STROKEAHA.122.036945.
Jurcau A, Ardelean AI. Oxidative stress in ischemia/reperfusion injuries following acute ischemic stroke. Biomedicines. 2022;10(3):574. https://doi.org/10.3390/biomedicines10030574.
Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524–51. https://doi.org/10.1016/j.redox.2015.08.020.
Lei XG, Zhu JH, Cheng WH, et al. Paradoxical roles of antioxidant enzymes: basic mechanisms and health implications. Physiol Rev. 2016;96:307–64. https://doi.org/10.1152/physrev.00010.2014.
De Bock M, Van Haver V, Vandenbroucke RE, et al. Into rather unexplored terrain-transcellular transport across the blood-brain barrier. Glia. 2016;64:1097–123. https://doi.org/10.1002/glia.22960.
Hu H, Huang H, Xia L, et al. Engineering vanadium carbide MXene as multienzyme mimetics for efficient in vivo ischemic stroke treatment. Chem Eng J. 2022;440:135810. https://doi.org/10.1016/j.cej.2022.135810.
Feng L, Dou C, Xia Y, et al. Neutrophil-like cell-membrane-coated nanozyme therapy for ischemic brain damage and long-term neurological functional recovery. ACS Nano. 2021;15:2263–80. https://doi.org/10.1021/acsnano.0c07973.
Fabian RH, Derry PJ, Rea HC, et al. Efficacy of novel carbon nanoparticle antioxidant therapy in a severe model of reversible middle cerebral artery stroke in acutely hyperglycemic rats. Front Neurol. 2018;9:199. https://doi.org/10.3389/fneur.2018.00199.
Jiang Y, Kang Y, Liu J, et al. Nanomaterials alleviating redox stress in neurological diseases: mechanisms and applications. J Nanobiotechnology. 2022;20(265):1–27. https://doi.org/10.1186/s12951-022-01434-5.
Liu Y, Ai K, Ji X, et al. Comprehensive insights into the multi-antioxidative mechanisms of melanin nanoparticles and their application to protect brain from injury in ischemic stroke. J Am Chem Soc. 2017;139:856–62. https://doi.org/10.1021/jacs.6b11013.
Mittal M, Siddiqui MR, Tran K, et al. Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal. 2014;20:1126–67. https://doi.org/10.1089/ars.2012.5149.
Dirnagl U, Iadecola C, Moskowitz MA. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci. 1999;22:391–7. https://doi.org/10.1016/s0166-2236(99)01401-0.
Liu Y, Wang X, Li X, et al. A co-doped Fe3O4 nanozyme shows enhanced reactive oxygen and nitrogen species scavenging activity and ameliorates the deleterious effects of ischemic stroke. ACS Appl Mater Interfaces. 2021;13:46213–24. https://doi.org/10.1021/acsami.1c06449.
Zhang K, Tu M, Gao W, et al. Hollow Prussian blue nanozymes drive neuroprotection against ischemic stroke via attenuating oxidative stress, counteracting inflammation, and suppressing cell apoptosis. Nano Lett. 2019;19:2812–23. https://doi.org/10.1021/acs.nanolett.8b04729.
Tian R, Ma H, Ye W, et al. Se-containing MOF coated dual-Fe-atom nanozymes with multi-enzyme cascade activities protect against cerebral ischemic reperfusion injury. Adv Funct Mater. 2022;32:2204025. https://doi.org/10.1002/adfm.202204025.
Xi J, Zhang R, Wang L, et al. A nanozyme-based artificial peroxisome ameliorates hyperuricemia and ischemic stroke. Adv Funct Mater. 2020;31:2007130. https://doi.org/10.1002/adfm.202007130.
Lin AM, Fang SF, Lin SZ, et al. Local carboxyfullerene protects cortical infarction in rat brain. Neurosci Res. 2000;43(4):317–21. https://doi.org/10.1016/s0168-0102(02)00056-1.
Zhao Q, Du W, Zhou L, et al. Transferrin-enabled blood-brain barrier crossing manganese-based nanozyme for rebalancing the reactive oxygen species level in ischemic stroke. Pharmaceutics. 2022;14:1122. https://doi.org/10.3390/pharmaceutics14061122.
Jiang Y, Brynskikh AM, S-Manickam D, et al. Sod1 nanozyme salvages ischemic brain by locally protecting cerebral vasculature. J Control Release. 2015;213:36–44. https://doi.org/10.1016/j.jconrel.2015.06.021.
Manickam DS, Brynskikh AM, Kopanic JL, et al. Well-defined cross-linked antioxidant nanozymes for treatment of ischemic brain injury. J Control Release. 2012;162:636–45. https://doi.org/10.1016/j.jconrel.2012.07.044.
Wang Z, Zhang R, Yan X, et al. Structure and activity of nanozymes: Inspirations for de novo design of nanozymes. Mater Today. 2020;41:81–119. https://doi.org/10.1016/j.mattod.2020.08.020.
Zhang Z, Liu B, Liu J. Molecular imprinting for substrate selectivity and enhanced activity of enzyme mimics. Small. 2017;13. https://doi.org/10.1002/smll.201602730.
Naveen Prasad S, Bansal V, Ramanathan R. Detection of pesticides using nanozymes: trends, challenges and outlook. Trends Analyt Chem. 2021;144:116429. https://doi.org/10.1016/j.trac.2021.116429.
Khoshfetrat SM, Dorraji PS, Fotouhi L, et al. Enhanced electrochemiluminescence biosensing of gene-specific methylation in thyroid cancer patients’ plasma-based integrated graphitic carbon nitride-encapsulated metal-organic framework nanozyme optimized by central composite design. Sens Actuators B Chem. 2022;364:131895. https://doi.org/10.1016/j.snb.2022.131895.
Khoshfetrat SM, Seyed Dorraji P, Shayan M, et al. Smartphone-based electrochemiluminescence for visual simultaneous detection of RASSF1A and SLC5A8 tumor suppressor gene methylation in thyroid cancer patient plasma. Anal Chem. 2022;94(22):8005–13. https://doi.org/10.1021/acs.analchem.2c01132.
Acknowledgements
This work was supported in part by grants of National Natural Science Foundation of China (No.81870938), Natural Science Foundation of Shandong Province (No.ZR2019ZD32, ZR2021QH160, ZR2022QH120, ZR2021MH025), Fund of Taishan Scholar Project, Fund of Teaching Promotion Programme, and Fund of Academic Promotion Program of Shandong First Medical University & Shandong Academy of Medical Sciences (No.2019QL016).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declared that they have no conflicts of interest to this work.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Han, Q., Wang, C., Liu, J. et al. Application of Nanozymes and its Progress in the Treatment of Ischemic Stroke. Transl. Stroke Res. (2023). https://doi.org/10.1007/s12975-023-01182-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12975-023-01182-0