Abstract
SiO2 is widely used in nanocomposites as reinforcement nanoparticle to enhance mechanical properties especially wear resistivity. Prior to use, surface modification with proper and sufficient coupling agent should be performed on it. Coupling agent concentration plays a key role in modification process.
Purpose
In this study, the influence of 3-(glycidoloxy propyl) trimethoxy silane (GPTMS) concentration on surface modification of SiO2 nanoparticles, is experimentally investigated.
Methods
The surface modification of nano-silica were performed by 30, 50, 80 and 110 wt.% of GPTMS in order to introduce the optimal GPTMS concentration to complete the process. Fourier Transformation Infrared Spectroscopy (FTIR), Field Emission Scanning Electron Microscopy (FESEM), Thermo Gravimetric Analysis (TGA) and X-Ray Diffraction (XRD) characterized the pure and surface modified samples; then, the results were compared to each other to achieve the aim of the research.
Results
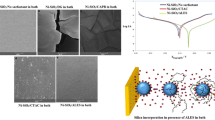
FTIR results confirmed the silanization proceed due to the silane absorption peak disappearing and shifting of the hydroxyl group bonds in to the amide bonds. This test showed that 30 wt.% GPTMS has not been sufficient for full functionalization of the NPs. According to FESEM images, it seems that the NPs were better modified by 80 wt.% GPTMS due to the least NPs aggregation and lack of coupling agent deposition on the NPs. Also, TGA illustrates that this sample has higher thermal stability because of lower weight loss (11.2%) in coupling agent decomposition temperature range: 130–380 °C. Furthermore, X-Ray Diffraction confirmed the FESEM and TGA results about the mentioned sample due to its highest crystallite size (increase 26.64% in crystallite size in comparison with the pure sample).
Conclusion
So, the 80 wt.% of GPTMS introduced as the optimal concentration for surface modification of SiO2 nanoparticles.
Similar content being viewed by others
Data Availability
Electronic supplementary materials contains FTIR, TGA and XRD original data
References
Zhou S-X, Wu L-M, Sun J, Shen W-D (2003) Effect of nanosilica on the properties of polyester-based polyurethane. J Appl Polym Sci 88:189–193. https://doi.org/10.1002/app.11624
Orfali WA (2015) Acoustic properties of polyurethane composition reinforced with carbon nanotubes and silicon oxide Nano-powder. Phys Procedia 70:699–702. https://doi.org/10.1016/j.phpro.2015.08.091
Mirzapour A, Asadollahi MH, Baghshaei S, Akbari M (2014) Effect of nanosilica on the microstructure, thermal properties and bending strength of nanosilica modified carbon fiber/phenolic nanocomposite. Compos Part A Appl Sci Manuf 63:159–167. https://doi.org/10.1016/j.compositesa.2014.04.009
Guo Y, Wang M, Zhang H, Liu G, Zhang L, Qu X (2008) The surface modification of nanosilica, preparation of nanosilica/acrylic core-shell composite latex, and its application in toughening PVC matrix. J Appl Polym Sci 107:2671–2680. https://doi.org/10.1002/app.27310
Petrovic ZS, Javni I, Waddon A, Banhegyi G (2000) Structure and properties of polyurethane-silica nanocomposites. J Appl Polym Sci 76:133–151. https://doi.org/10.1002/(SICI)1097-4628(20000411)76:2 <133::AID-APP3 >3.0.CO;2-K
Blees MH, Winkelman GB, Balkenende AR, den Toonder JMJ (2000) The effect of friction on scratch adhesion testing: application to a sol–gel coating on polypropylene. Thin Solid Films 359:1–13. https://doi.org/10.1016/S0040-6090(99)00729-4
Novak BM (1993) Hybrid nanocomposite materials?Between inorganic glasses and organic polymers. Adv Mater 5:422–433. https://doi.org/10.1002/adma.19930050603
Ching YC, Syamimie N (2013) Effect of Nanosilica filled polyurethane composite coating on polypropylene substrate. J Nanomater 2013:1–8. https://doi.org/10.1155/2013/567908
Wang F, Feng L, Ma H, Zhai Z, Liu Z (2019) Influence of nano-SiO2 on the bonding strength and wear resistance properties of polyurethane coating. Sci Eng Compos Mater 26:77–83. https://doi.org/10.1515/secm-2018-0078
Zhou S, Wu L, Sun J, Shen W (2002) The change of the properties of acrylic-based polyurethane via addition of nano-silica. Prog Org Coatings 45:33–42. https://doi.org/10.1016/S0300-9440(02)00085-1
Bahramnia H, Mohammadian Semnani H, Habibolahzadeh A, Abdoos H (2020) Epoxy/polyurethane nanocomposite coatings for anti-erosion/wear applications: a review. J Compos Mater 54:3189–3203. https://doi.org/10.1177/0021998320908299
Laruelle G, Parvole J, Francois J, Billon L (2004) Block copolymer grafted-silica particles: a core/double shell hybrid inorganic/organic material. Polymer (Guildf) 45:5013–5020. https://doi.org/10.1016/j.polymer.2004.05.030
von Werne T, Patten TE (2001) Atom transfer radical polymerization from nanoparticles: a tool for the preparation of well-defined hybrid nanostructures and for understanding the chemistry of controlled/“living” radical polymerizations from surfaces. J Am Chem Soc 123:7497–7505. https://doi.org/10.1021/ja010235q
Hsiue G-H, Kuo W-J, Huang Y-P, Jeng R-J (2000) Microstructural and morphological characteristics of PS–SiO2 nanocomposites. Polymer (Guildf) 41:2813–2825. https://doi.org/10.1016/S0032-3861(99)00478-4
Bialk M, Prucker O, Rühe J (2002) Grafting of polymers to solid surfaces by using immobilized methacrylates. Colloids Surfaces A Physicochem Eng Asp 198–200:543–549. https://doi.org/10.1016/S0927-7757(01)00958-X
Yoshinaga K, Shimada J, Nishida H, Komatsu M (1999) Effects of secondary polymer covalently attached to monodisperse, poly(maleic anhydride–styrene)-modified colloidal silica on Dispersibility in organic solvent. J Colloid Interface Sci 214:180–188. https://doi.org/10.1006/jcis.1999.6182
Wang Q, Xia H, Zhang C (2001) Preparation of polymer/inorganic nanoparticles composites through ultrasonic irradiation. J Appl Polym Sci 80:1478–1488. https://doi.org/10.1002/app.1239
Ghosh S, Goswami SK, Mathias LJ (2013) Surface modification of nano-silica with amides and imides for use in polyester nanocomposites. J Mater Chem A 1:6073. https://doi.org/10.1039/c3ta10381a
Nagano T, Nagano K, Nabeshi H, Yoshida T, Kamada H, Tsunoda SI, Gao JQ, Higashisaka K, Yoshioka Y, Tsutsumi Y (2017) Modifying the surface of silica nanoparticles with amino or carboxyl groups decreases their cytotoxicity to parenchymal hepatocytes. Biol Pharm Bull 40:726–728. https://doi.org/10.1248/bpb.b16-00917
Xu MH, Cao YY, Gao SG (2014) Surface modification of Nano-silica with Silane coupling agent. Key Eng Mater 636:23–27. https://doi.org/10.4028/www.scientific.net/KEM.636.23
Nikje MMA, Tehrani ZM (2010) Novel modified Nanosilica-based on synthesized Dipodal Silane and its effects on the physical properties of rigid polyurethane foams. Des Monomers Polym 13:249–260. https://doi.org/10.1163/138577210X12634696333631
Kumudinie C (2001) Polymer–ceramic nanocomposites: interfacial bonding agents. In: Encyclopedia of materials: science and technology, Second. Elsevier, pp. 7574–7577
Bahramnia H, Mohammadian Semnani H, Habibolahzadeh A, Abdoos H, Rezaei F (2021) The effect of 3-(triethoxy silyl) propyl amine concentration on surface modification of multiwall carbon nanotubes. Fullerenes, Nanotub Carbon Nanostructures 29:74–82. https://doi.org/10.1080/1536383X.2020.1813719
Chuang W, Geng-sheng J, Lei P, Bao-lin Z, Ke-zhi L, Jun-long W (2018) Influences of surface modification of nano-silica by silane coupling agents on the thermal and frictional properties of cyanate ester resin. Results Phys 9:886–896. https://doi.org/10.1016/j.rinp.2018.03.056
Rong Z, Zhao M, Wang Y (2020) Effects of modified Nano-SiO2 particles on properties of high-performance cement-based composites. Materials (Basel) 13:646. https://doi.org/10.3390/ma13030646
Bahramnia H, Semnani HM, Habibolahzadeh A, Abdoos H (2021) Epoxy/polyurethane hybrid nanocomposite coatings reinforced with MWCNTs and SiO2 nanoparticles: processing, mechanical properties and wear behavior. Surf Coatings Technol 415:127121. https://doi.org/10.1016/j.surfcoat.2021.127121
Marrone M, Montanari T, Busca G, Conzatti L, Costa G, Castellano M, Turturro A (2004) A Fourier transform infrared (FTIR) study of the reaction of Triethoxysilane (TES) and Bis[3-triethoxysilylpropyl]tetrasulfane (TESPT) with the surface of amorphous silica. J Phys Chem B 108:3563–3572. https://doi.org/10.1021/jp036148x
Derchi G, Manca E, Shayganpour A, Barone A, Diaspro A, Salerno M (2015) Combined characterization of the time response of impression materials via traditional and FTIR measurements. Materials (Basel) 8:2387–2399. https://doi.org/10.3390/ma8052387
Oliveira RN, Mancini MC, de Oliveira FCS et al (2016) FTIR analysis and quantification of phenols and flavonoids of five commercially available plants extracts used in wound healing. Matéria (Rio Janeiro) 21:767–779. https://doi.org/10.1590/S1517-707620160003.0072
Jung MR, Horgen FD, Orski SV, Rodriguez C. V, Beers KL, Balazs GH, Jones TT, Work TM, Brignac KC, Royer SJ, Hyrenbach KD, Jensen BA, Lynch JM (2018) Validation of ATR FT-IR to identify polymers of plastic marine debris, including those ingested by marine organisms. Mar Pollut Bull 127:704–716. https://doi.org/10.1016/j.marpolbul.2017.12.061
Robaina NF, de Paula CER, Brum DM, de la Guardia M, Garrigues S, Cassella RJ (2013) Novel approach for the determination of azithromycin in pharmaceutical formulations by Fourier transform infrared spectroscopy in film-through transmission mode. Microchem J 110:301–307. https://doi.org/10.1016/j.microc.2013.04.015
Turnes Palomino G, Otero Areán C, Geobaldo F, Ricchiardi G, Bordiga S, Zecchina A (1997) FTIR study of CO adsorbed at low temperature on zeolite L evidence for an ordered distribution of aluminium atoms. J Chem Soc Faraday Trans 93:189–191. https://doi.org/10.1039/a605523k
Shahrokh Abadi MH, Delbari A, Fakoor Z, Baedi J (2015) Effects of annealing temperature on infrared spectra of SiO2 extracted from Rice husk. J Ceram Sci Technol 06:41–46. https://doi.org/10.4416/JCST2014-00028
Yang W-G, Ha J-H, Kim S-G, Chae W-S (2016) Spectroscopic determination of alkyl resorcinol concentration in hydroxyapatite composite. J Anal Sci Technol 7:9. https://doi.org/10.1186/s40543-016-0089-2
Sanches NB, Cassu SN, Dutra R de CL (2015) TG/FT-IR characterization of additives typically employed in EPDM formulations. Polímeros 25:247–255. https://doi.org/10.1590/0104-1428.1819
Nandiyanto ABD, Oktiani R, Ragadhita R (2019) How to read and interpret FTIR spectroscope of organic material. Indones J Sci Technol 4:97. https://doi.org/10.17509/ijost.v4i1.15806
Sayed M, Kamal El-Dean AM, Ahmed M, Hassanien R (2019) Design, synthesis, and characterization of novel pyrimidines bearing indole as antimicrobial agents. J Chin Chem Soc 66:218–225. https://doi.org/10.1002/jccs.201800115
Zhang H, Chen B, Banfield JF (2010) Particle size and pH effects on nanoparticle dissolution. J Phys Chem C 114:14876–14884. https://doi.org/10.1021/jp1060842
Biswas RK, Khan P, Mukherjee S, Mukhopadhyay AK, Ghosh J, Muraleedharan K (2018) Study of short range structure of amorphous Silica from PDF using ag radiation in laboratory XRD system, RAMAN and NEXAFS. J Non-Cryst Solids 488:1–9. https://doi.org/10.1016/j.jnoncrysol.2018.02.037
Qiu F, Xu H, Wang Y, Xu J, Yang D (2012) Preparation, characterization and properties of UV-curable waterborne polyurethane acrylate/SiO2 coating. J Coatings Technol Res 9:503–514. https://doi.org/10.1007/s11998-012-9397-7
Wu D, Qiu F, Xu H, Zhang J, Yang D (2011) Preparation, characterization, and properties of environmentally friendly waterborne poly(urethane acrylate)/silica hybrids. J Appl Polym Sci 119:1683–1695. https://doi.org/10.1002/app.32846
de Ferri L, Lottici PP, Lorenzi A, Montenero A, Salvioli-Mariani E (2011) Study of silica nanoparticles – polysiloxane hydrophobic treatments for stone-based monument protection. J Cult Herit 12:356–363. https://doi.org/10.1016/j.culher.2011.02.006
Acknowledgments
The authors would like acknowledge from Ministry of Science, Research and Technology of Islamic Republic of Iran for supporting this work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Bahramnia H. designed and performed experiments, investigated the results and wrote manuscript draft.
Mohammadian Semnani H. supervised the entire research and supported financially the experiments.
Habibolahzadeh A. conceptualized the research.
Abdoos H. performed data curation, reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
Authors declared no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bahramnia, H., Semnani, H.M., Habibolahzadeh, A. et al. The Effect of 3-(Glycidoloxy Propyl) Trimethoxy Silane Concentration on Surface Modification of SiO2 Nanoparticles. Silicon 14, 4969–4977 (2022). https://doi.org/10.1007/s12633-021-01237-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-021-01237-7