Abstract
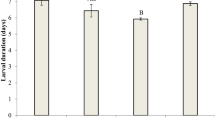
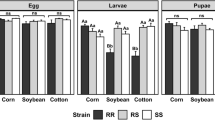
The oriental fruit fly, Bactrocera dorsalis (Hendel), is one of the most globally important insect pests that have evolved resistance to various groups of insecticides. To clarify the relationships between a novel and widely used anthranilic diamide insecticide (cyantraniliprole) and other regular insecticides used to effectively control B. dorsalis, its cross-resistance and fitness costs to various insecticides were determined. Bioassays with cyantraniliprole-resistant and cyantraniliprole-susceptible strains indicated moderate levels of cross-resistance to both of the spinosyn compounds, Spinosad (1.59-fold) and Spinetoram (1.54-fold), the organophosphate (Trichlorphon, 0.66-fold), and beta-cypermethrin (2.41-fold), one of the pyrethroids compounds. Most of the tested biological parameters of B. dorsalis were negatively affected by cyantraniliprole resistance. Compared with the susceptible strain, prolonged larval duration, higher pupa weight, longer timing of sexual maturation, were observed in the cyantraniliprole-resistant strain. Pupa duration, oviposition, and male remating inhibition on the fifth day were significantly lower for the cyantraniliprole-resistant strain than those of the susceptible strain. Moreover, there was no fitness costs associated with resistance to cyantraniliprole. Morderate levels of cross-resistance were observed between cyantraniliprole and four other selected insecticides (Spinosad, Spinetoram, Trichlorphon, and Beta-cypermethrin). However, the lack of fitness costs in the cyantraniliprole-resistant strain of B. dorsalis could promote the rapid development of resistance to cyantraniliprole. Thus, the results obtained here were useful for the application and resistance management of cyantraniliprole against B. dorsalis.
Similar content being viewed by others
References
Abbas, N., Khan, H., & Shad, S. (2015). Cross-resistance, stability, and fitness cost of resistance to imidacloprid in Musca domestica L.,(Diptera: Muscidae). Parasitology Research, 114, 247–255. https://doi.org/10.1007/s00436-014-4186-0.
Ahmad, M., & Iqbal Arif, M. (2009). Resistance of Pakistani field populations of spotted bollworm Earias vittella (Lepidoptera: Noctuidae) to pyrethroid, organophosphorus and new chemical insecticides. Pest Management Science: formerly Pesticide Science, 65, 433–439. https://doi.org/10.1002/ps.1702.
Alvi, A., Sayyed, A., Naeem, M., & Ali, M. (2012). Field evolved resistance in Helicoverpa armigera (Lepidoptera: Noctuidae) to Bacillus thuringiensis toxin Cry1Ac in Pakistan. PLoS One, 7, e47309. https://doi.org/10.1371/journal.pone.0047309.
Basit, M., Sayyed, A., Saeed, S., & Saleem, M. (2012). Lack of fitness costs associated with acetamiprid resistance in Bemisia tabaci (Hemiptera: Aleyrodidae). Journal of Economic Entomology, 105(4), 1401–1406. https://doi.org/10.1603/EC11196.
Bisset, J., Rodriguez, M., Soca, A., Pasteur, N., & Raymond, M. (1997). Cross-resistance to pyrethroid and organophosphorus insecticides in the southern house mosquito (Diptera: Culicidae) from Cuba. Journal of Medical Entomology, 34, 244–246. https://doi.org/10.1093/jmedent/34.2.244.
Bielza, P. (2008). Insecticide resistance management strategies against the western flower thrips, Frankliniella occidentalis. Pest Management Science, 64, 1131–1138. https://doi.org/10.1002/ps.1620.
Cao, G., Lu, Q., Zhang, L., Guo, F., Liang, G., Wu, K., et al. (2010). Toxicity of chlorantraniliprole to Cry1Ac-susceptible and resistant strains of Helicoverpa armigera. Pesticide Biochemistry Physiology, 98, 99–103. https://doi.org/10.1016/j.pestbp.2010.05.006.
Cai, P., Hong, J., Wang, C., Yang, Y., Zhang, Q., Ji, Q., & Chen, J. (2018). Radiation of Bactrocera dorsalis (Diptera: Tephritidae) eggs to improve the mass rearing of Diachasmimorpha longicaudata (Hymenoptera: Braconidae). Journal of Economic Entomology, 111(3), 1157–1164. https://doi.org/10.1093/jee/toy032.
Drew, R. (1987). Behavioural strategies of fruit flies of the genus Dacus (Diptera: Tephritidae) significant in mating and host-plant relationships. Bulletin of Entomological Research, 77, 73–81. https://doi.org/10.1017/S000748530001155X.
Dang, X. L., Tian, J. H., Yi, H. Y., Wang, W. X., Zheng, M. I. N., Li, Y. F., et al. (2006). Inducing and isolation of antibacterial peptides from oriental fruit fly, Bactrocera dorsalis Hendel. Insect Sci., 13, 257–262.
Ejaz, M., & Ali Shad, S. (2017). Spirotetramat resistance selected in the phenacoccus solenopsis (homoptera: Pseudococcidae): Cross-resistance patterns, stability, and fitness costs analysis. Journal of Economic Entomology, 110(3), 1226–1234. https://doi.org/10.1093/jee/tox045.
Fang, C., Okuyama, T., Wu, W., Feng, H., & Hsu, J. (2011). Fitness costs of an insecticide resistance and their population dynamical consequences in the oriental fruit fly. Journal of Economic Entomology, 104, 2039–2045. https://doi.org/10.1603/EC11200.
Gomis-Cebolla, J., Wang, Y., Quan, Y., He, K., Walsh, T., James, B., et al. (2018). Analysis of cross-resistance to vip3 proteins in eight insect colonies, from four insect species, selected for resistance to Bacillus thuringiensis insecticidal proteins. Journal of Invertebrate Pathology, 155, 64–70.
Grávalos, C., Fernández, E., Belando, A., Moreno, I., Ros, C., & Bielza, P. (2015). Cross-resistance and baseline susceptibility of Mediterranean strains of Bemisia tabaci to cyantraniliprole. Pest Management Science, 71, 1030–1036. https://doi.org/10.1002/ps.3885.
Groeters, F., Tabashnik, B., Finson, N., & Johnson, M. (1993). Resistance to Bacillus thuringiensis affects mating success of the diamondback moth (Lepidoptera: Plutellidae). Journal of Economic Entomology, 86, 1035–1039. https://doi.org/10.1093/jee/86.4.1035.
Hsu, J., Feng, H., & Wu, W. (2004). Resistance and synergistic effects of insecticides in Bactrocera dorsalis (Diptera: Tephritidae) in Taiwan. Journal of Economic Entomology, 97, 1682–1688. https://doi.org/10.1603/0022-0493-97.5.1682.
Hsu, J. C., & Feng, H. T. (2006). Development of resistance to spinosad in oriental fruit fly (Diptera: Tephritidae) in laboratory selection and cross-resistance. Journal of Economic Entomology, 99, 931–936. https://doi.org/10.1093/jee/99.3.931.
Jin, T., Zeng, L., Lin, Y., Lu, Y., & Liang, G. (2011). Insecticide resistance of the oriental fruit fly, Bactrocera dorsalis (Hendel)(Diptera: Tephritidae), in mainland China. Pest Management Science, 67, 370–376. https://doi.org/10.1002/ps.2076.
Khan, H., & Akram, W. (2018). Trichlorfon and spinosad resistance survey and preliminary determination of the resistance mechanism in Pakistani field strains of Bactrocera dorsalis. Scientific Reports, 8, 11223. https://doi.org/10.1038/s41598-018-29622-0.
Kliot, A., & Ghanim, M. (2012). Fitness costs associated with insecticide resistance. Pest Management Science, 68, 1431–1437. https://doi.org/10.1002/ps.3395.
Lahm, G., Cordova, D., Barry, J., Andaloro, J., Annan, I., Marcon, P., Portillo, H., Stevenson, T., & Selby, T. (2011). Anthranilic diamide insecticides-Rynaxypyr® and Cyazypyr™.In: W. Kramer, U. Schirmer (Eds). Modern Crop Protection Compounds Second Revised and Enlarged Edition; Wiley-VCH, pp.1409-1425.
Liu, X., Ning, Y., Wang, H., & Wang, K. (2015). Cross-resistance, mode of inheritance, synergism, and fitness effects of cyantraniliprole resistance in Plutella xylostella. Entomologia Experimentalis et Applicata, 157, 271–278. https://doi.org/10.1111/eea.12361.
Rowland, M. (1991 Apr). Activity and mating competitiveness of gamma HCH/dieldrin resistant and susceptible male and virgin female Anopheles gambiae and An.stephensi mosquitoes, with assessment of an insecticide-rotation strategy. Medical and Veterinary Entomology, 5, 207–222. https://doi.org/10.1111/j.1365-2915.1991.tb00543.x.
Sang, S., Shu, B., Yi, X., Liu, J., Hu, M., & Zhong, G. (2016). Cross-resistance and baseline susceptibility of Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae) to cyantraniliprole in the south of China. Pest Management Science, 72, 922–928. https://doi.org/10.1002/ps.4068.
Sattelle, D., Cordova, D., & Cheek, T. (2008). Insect ryanodine receptors: Molecular targets for novel pest control chemicals. Invertebrate Neuroscience, 8, 107–119. https://doi.org/10.1007/s10158-008-0076-4.
Scott, I., Tolman, J., & MacArthur, D. (2015). Insecticide resistance and cross-resistance development in Colorado potato beetle Leptinotarsa decemlineata say (Coleoptera: Chrysomelidae) populations in Canada 2008-2011. Pest Management Science, 71, 712–721. https://doi.org/10.1603/EC13294.
Shelly, T., Edu, J., & McInnis, D. (2010). Pre-release consumption of methyl eugenol increases the mating competitiveness of sterile males of the oriental fruit fly, Bactrocera dorsalis, in large field enclosures. Journal of Insect Science, 10, 8–16. https://doi.org/10.1673/031.010.0801.
Shi, J., Zhang, L., Mi, J., & Gao, X. (2020). Role transformation of fecundity and viability: The leading cause of fitness costs associated with beta-cypermethrin resistance in musca domestica.PLoS One,15(1) doi: https://doi.org/10.1371/journal.pone.0228268.
Sun, J., Liang, P., & Gao, X. (2012). Cross-resistance patterns and fitness in fufenozide-resistant diamondback moth, Plutella xylostella (Lepidoptera: Plutellidae). Pest Management Science, 68, 285–289. https://doi.org/10.1002/ps.2258.
Tan, K., & Nishida, R. (1996). Sex pheromone and mating competition after methyl eugenol consumption in the Bactrocera dorsalis complex. Fruit fly pests: A world assessment of their biology and management. St. Lucie Press, Delray Beach, FL, 147-153.
Tamhane, A. (1979). A comparison of procedures for multiple comparisons of means with unequal variances. Journal of the American Statistical Association, 74, 471–480.
Teixeira, L., & Andaloro, J. (2013). Diamide insecticides: Global efforts to address insect resistance stewardship challenges. Pesticide Biochemistry Physiology, 106, 76–78. https://doi.org/10.1016/j.pestbp.2013.01.010.
Vontas, J., Hernández-Crespo, P., Margaritopoulos, J. T., Ortego, F., Feng, H. T., Mathiopoulos, K. D., & Hsu, J. C. (2011). Insecticide resistance in tephritid flies. Pesticide Biochemistry & Physiology, 100(3), 199–205. https://doi.org/10.1016/j.pestbp.2011.04.004.
Wang, D., Qiu, X., Wang, H., Qiao, K., & Wang, K. (2010). Reduced fitness associated with spinosad resistance in Helicoverpa armigera. Phytoparasitica, 38, 103–110. https://doi.org/10.1007/s12600-009-0077-9.
Wei, Q. B., Lei, Z. R., Nauen, R., Cai, D. C., & Gao, Y. L. (2015a). Abamectin resistance in strains of vegetable leafminer, Liriomyza sativae (Diptera: Agromyzidae) is linked to elevated glutathione S-transferase activity. Insect Sci., 22, 243–250. https://doi.org/10.1111/1744-7917.12080.
Wee, S. L., & Tan, K. H. (2000). Sexual maturity and intraspecific mating success of two sibling species of the Bactrocera dorsalis complex. Entomologia Experimentalis et Applicata, 94(2), 133–139. https://doi.org/10.1046/j.1570-7458.2000.00613.x.
Wei, D., Feng, Y., Wei, D., Yuan, G., Dou, W., & Wang, J. (2015b). Female remating inhibition and fitness of Bactrocera dorsalis (Diptera: Tephritidae) associated with male accessory glands. The Florida Entomologist, 98(1), 52–58. https://doi.org/10.1653/024.098.0110.
Zhang, R., He, S., & Chen, J. (2014). Monitoring of Bactrocera dorsalis (Diptera: Tephritidae) resistance to cyantraniliprole in the south of China. Journal of Economic Entomology, 107, 1233–1238. https://doi.org/10.1603/EC13294.
Zhang, R., Jang, E., He, S., & Chen, J. (2015). Lethal and sublethal effects of cyantraniliprole on Bactrocera dorsalis (Hendel) (Diptera: Tephritidae). Pest Management Science, 71, 250–256. https://doi.org/10.1002/ps.3791.
Zhao, J., Collins, H. L., & Shelton, A. M. (2010). Testing insecticide resistance management strategies: Mosaic versus rotations.Pest. Management Science, 66(10), 1101–1105. https://doi.org/10.1002/ps.1985.
Acknowledgments
This work is funded by the National Key Research and Development Program of China (2017YFD0202000); the National Natural Science Foundation of China (31701755); the China Agriculture Research System (CARS-26); and the President’s Fund of Guangdong Academy of Agricultural Sciences (201703).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ruimin, Z., Shiyu, H., Jiwu, Z. et al. Cross-resistance and lack of fitness costs occurred in the cyantraniliprole-resistant oriental fruit fly. Phytoparasitica 49, 703–712 (2021). https://doi.org/10.1007/s12600-021-00894-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-021-00894-z