Abstract
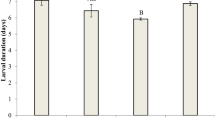
Insecticide resistance is usually associated with fitness costs. The magnitude of the fitness costs is affected by environmental and ecological factors. Here, we explored how host plants could affect fitness costs associated with insecticide resistance. Initially, spinetoram-resistant and susceptible strains of Spodoptera frugiperda were selected using a F2 screen from a population collected in São Desidério, Bahia State, Brazil. In addition to the RR and SS strains, fitness costs were also assessed for a heterozygous strain (RS). Life-history traits were evaluated to estimate population growth parameters of each strain feeding on corn, soybean, and cotton plants. The relative fitness of the RR strain was 1.06 higher compared to the SS strain on corn plants, while in soybean and cotton plants these values were 0.84 and 0.67 lower, respectively. The relative fitness of the RS strain was similar to the SS strain regardless of the host plant, suggesting a recessive fitness cost. No differences were found between the strains fed on corn plants. The larval development time was greater for the RR strain fed on soybean and cotton plants compared to the RS and SS strains. Low survival rate and fecundity of the RR strain were found when larvae fed on soybean and cotton plants. The results of this study showed that fitness costs of spinetoram resistance in S. frugiperda depend on the host plants that S. frugiperda larvae fed on. Such information can be used to design resistance management strategies considering the host plants of the agricultural landscape.



Similar content being viewed by others
References
Afzal MBS, Shad SA (2017) Spinosad resistance in an invasive cotton mealybug, Phenacoccus solenopsis: cross-resistance, stability and relative fitness. J Asia Pac Entomol 20:457–462. https://doi.org/10.1016/j.aspen.2017.03.002
Andow DA, Alstad DN (1998) F2 screen for rare resistance alleles. J Econ Entomol 91:572–578. https://doi.org/10.1093/jee/91.3.572
Awmack CS, Leather SR (2002) Host plant quality and fecundity in herbivorous insects. Annu Rev Entomol 47:817–844. https://doi.org/10.1146/annurev.ento.47.091201.145300
Baloch MN, Fan J, Haseeb M, Zhang R (2020) Mapping potential distribution of Spodoptera frugiperda (Lepidoptera: Noctuidae) in central Asia. InSects 11:172. https://doi.org/10.3390/insects11030172
Barros EM, Torres JB, Ruberson JR, Oliveira MD (2010) Development of Spodoptera frugiperda on different hosts and damage to reproductive structures in cotton. Entomol Exp Appl 137:237–245. https://doi.org/10.1111/j.1570-7458.2010.01058.x
Baxter SW, Chen M, Dawson A et al (2010) Mis-spliced transcripts of nicotinic acetylcholine receptor α6 are associated with field evolved spinosad resistance in Plutella xylostella (L.). PLoS Genet 6:e1000802. https://doi.org/10.1371/journal.pgen.1000802
Bird LJ, Akhurst RJ (2007) Effects of host plant species on fitness costs of Bt resistance in Helicoverpa armigera (Lepidoptera: Noctuidae). Biol Control 40:196–203. https://doi.org/10.1016/j.biocontrol.2006.11.004
Bird LJ, Drynan LJ, Walker PW (2020) Relative fitness and stability of resistance in a near-isogenic strain of indoxacarb resistant Helicoverpa armigera (Lepidoptera: Noctuidae). Pest Manag Sci 76:4077–4085. https://doi.org/10.1002/ps.5962
Bolzan A, Padovez FEO, Nascimento ARB et al (2019) Selection and characterization of the inheritance of resistance of Spodoptera frugiperda (Lepidoptera: Noctuidae) to chlorantraniliprole and cross-resistance to other diamide insecticides. Pest Manag Sci 75:2682–2689. https://doi.org/10.1002/ps.5376
Buainain AM, Lanna R, Navarro Z (2019) Agricultural development in Brazil: the rise of a global agro-food power. Routledge, London. https://doi.org/10.4324/9781351029742
Burtet LM, Bernardi O, Melo AA et al (2017) Managing fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), with Bt maize and insecticides in southern Brazil. Pest Manag Sci 73:2569–2577. https://doi.org/10.1002/ps.4660
CABI (2021) Spodoptera frugiperda. In: Invasive species compendium. CAB International, Wallingford. https://www.cabi.org/isc/fallarmyworm. Accessed 15 Jan 2021
Carrière Y, Ellers-Kirk C, Biggs R et al (2004) Effects of gossypol on fitness costs associated with resistance to Bt cotton in pink bollworm. J Econ Entomol 97:1710–1718. https://doi.org/10.1603/0022-0493-97.5.1710
Carrière Y, Yelich AJ, Degain BA et al (2019) Gossypol in cottonseed increases the fitness cost of resistance to Bt cotton in pink bollworm. Crop Prot 126:104914. https://doi.org/10.1016/j.cropro.2019.104914
Carvalho RA, Omoto C, Field LM et al (2013) Investigating the molecular mechanisms of organophosphate and pyrethroid resistance in the fall armyworm Spodoptera frugiperda. PLoS ONE 8(4):e62268. https://doi.org/10.1371/journal.pone.0062268
Chen X, Head GP, Price P et al (2019) Fitness costs of Vip3A resistance in Spodoptera frugiperda on different hosts. Pest Manag Sci 75:1074–1080. https://doi.org/10.1002/ps.5218
Crouse GD, Sparks TC, Schoonover J et al (2001) Recent advances in the chemistry of spinosyns. Pest Manag Sci 57:177–185. https://doi.org/10.1002/1526-4998(200102)57:2%3c177::AID-PS281%3e3.0.CO;2-Z
Diez-Rodríguez GI, Omoto C (2001) Herança da resistência de Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) a lambda-cialotrina. Neotrop Entomol 30:311–316. https://doi.org/10.1590/S1519-566X2001000200016
Dripps JE, Boucher RE, Chloridis A et al (2011) The spinosyn insecticides. In: Lopez O, Fernandez-Bolanos JG (eds) Green trends in insect control. Royal Society of Chemistry, Cambridge, pp 163–212. https://doi.org/10.1039/BK9781849731492-00163
Feyereisen R (2012) Insect CYP genes and P450 enzymes. In: Gilbert LI (ed) Insect molecular biology and biochemistry. Elsevier, Amsterdam, pp 236–316. https://doi.org/10.1016/B978-0-12-384747-8.10008-X
Freeman JC, Smith LB, Silva JJ et al (2021) Fitness studies of insecticide resistant strains: Lessons learned and future directions. Pest Manag Sci 77:3847–3856. https://doi.org/10.1002/ps.6306
Fu B, Li Q, Qiu H et al (2018) Resistance development, stability, cross-resistance potential, biological fitness and biochemical mechanisms of spinetoram resistance in Thrips hawaiiensis (Thysanoptera: Thripidae). Pest Manag Sci 74:1564–1574. https://doi.org/10.1002/ps.4887
Garlet CG, da Gubiani PS, Palharini RB et al (2021a) Field-evolved resistance to chlorpyrifos by Spodoptera frugiperda (Lepidoptera: Noctuidae): Inheritance mode, cross-resistance patterns, and synergism. Pest Manag Sci 77:5367–5374. https://doi.org/10.1002/ps.6576
Garlet CG, Moreira RP, da Gubiani PS et al (2021b) Fitness cost of chlorpyrifos resistance in Spodoptera frugiperda (Lepidoptera: Noctuidae) on different host plants. Environ Entomol 50:898–908. https://doi.org/10.1093/ee/nvab046
Gassmann AJ, Carrière Y, Tabashnik BE (2009) Fitness costs of insect resistance to Bacillus thuringiensis. Annu Rev Entomol 54:147–163. https://doi.org/10.1146/annurev.ento.54.110807.090518
Goergen G, Kumar PL, Sankung SB et al (2016) First report of outbreaks of the fall armyworm Spodoptera frugiperda (JE Smith)(Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS ONE 11:e0165632. https://doi.org/10.1371/journal.pone.0165632
Guillem-Amat A, Ureña E, López-Errasquín E et al (2020) Functional characterization and fitness cost of spinosad-resistant alleles in Ceratitis capitata. J Pest Sci 93:1043–1058. https://doi.org/10.1007/s10340-020-01205-x
Heidel-Fischer HM, Vogel H (2015) Molecular mechanisms of insect adaptation to plant secondary compounds. Curr Opin Insect Sci 8:8–14. https://doi.org/10.1016/j.cois.2015.02.004
Hsu J-C, Feng H-T, Wu W-J et al (2012) Truncated transcripts of nicotinic acetylcholine subunit gene Bdα6 are associated with spinosad resistance in Bactrocera dorsalis. Insect Biochem Mol Biol 42:806–815. https://doi.org/10.1016/j.ibmb.2012.07.010
Jakka SRK, Knight VR, Jurat-Fuentes JL (2014) Fitness costs associated with field-evolved resistance to Bt maize in Spodoptera frugiperda (Lepidoptera: Noctuidae). J Econ Entomol 107:342–351. https://doi.org/10.1603/EC13326
Janmaat AF, Myers JH (2005) The cost of resistance to Bacillus thuringiensis varies with the host plant of Trichoplusia ni. Proc R Soc B Biol Sci 272:1031–1038. https://doi.org/10.1098/rspb.2004.3040
Kasten P Jr, Precetti A, Parra JRP (1978) Dados biologicos comparativos de Spodoptera frugiperda (JE Smith, 1797) em duas dietas artificiais e substrato natural. Rev Agric 53:68–78. https://doi.org/10.5216/pat.v13i1.2442
Kennedy GG, Storer NP (2000) Life systems of polyphagous arthropod pests in temporally unstable cropping systems. Annu Rev Entomol 45:467–493. https://doi.org/10.1146/annurev.ento.45.1.467
Kliot A, Ghanim M (2012) Fitness costs associated with insecticide resistance. Pest Manag Sci 68:1431–1437. https://doi.org/10.1002/ps.3395
Li X, Schuler MA, Berenbaum MR (2007a) Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu Rev Entomol 52:231–253. https://doi.org/10.1146/annurev.ento.51.110104.151104
Li ZM, Liu SS, Liu YQ, Ye GY (2007b) Temperature-related fitness costs of resistance to spinosad in the diamondback moth, Plutella xylostella (Lepidoptera: Plutelidae). Bull Entomol Res 97:627–635. https://doi.org/10.1017/S0007485307005366
Lira EC, Bolzan A, Nascimento ARB et al (2020) Resistance of Spodoptera frugiperda (Lepidoptera: Noctuidae) to spinetoram: inheritance and cross-resistance to spinosad. Pest Manag Sci 76:2674–2680. https://doi.org/10.1002/ps.5812
Lu K, Song Y, Zeng R (2020) The role of cytochrome P450-mediated detoxification in insect adaptation to xenobiotics. Curr Opin Insect Sci 73:103–107. https://doi.org/10.1016/j.cois.2020.11.004
Maia AHN, Luiz AJB, Campanhola C (2000) Statistical inference on associated fertility life table parameters using jackknife technique: computational aspects. J Econ Entomol 93:511–518. https://doi.org/10.1603/0022-0493-93.2.511
Maia AHN, Pazianotto RAA, Luiz AJB et al (2014) Inference on arthropod demographic parameters: computational advances using R. J Econ Entomol 107:432–439. https://doi.org/10.1603/EC13222
Montezano DG, Specht A, Sosa-Gómez DR et al (2018) Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. African Entomol 26:286–301. https://doi.org/10.4001/003.026.0286
Moral RA, Hinde J, Demétrio CGB (2017) Half-normal plots and overdispersed models in R: The hnp Package. J Stat Software 81:1–23. https://doi.org/10.18637/jss.v081.i10
Mota-Sanchez D, Wise JC (2021) The arthropod pesticide resistance database. Michigan state university. http://www.pesticideresistance.org Accessed 16 Nov 2021
Muraro DS, de Oliveira Abbade Neto D, Kanno RH et al (2021) Inheritance patterns, cross-resistance and synergism in Spodoptera frugiperda (Lepidoptera: Noctuidae) resistant to emamectin benzoate. Pest Manag Sci 77:5049–5057. https://doi.org/10.1002/ps.6545
Nascimento ARB, Farias JR, Bernardi D et al (2016) Genetic basis of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to the chitin synthesis inhibitor lufenuron. Pest Manag Sci 72:810–815. https://doi.org/10.1002/ps.4057
Okuma DM, Bernardi D, Horikoshi RJ et al (2018) Inheritance and fitness costs of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to spinosad in Brazil. Pest Manag Sci 74:1441–1448. https://doi.org/10.1002/ps.4829
Perry T, McKenzie JA, Batterham P (2007) A Dα6 knockout strain of Drosophila melanogaster confers a high level of resistance to spinosad. Insect Biochem Mol Biol 37:184–188. https://doi.org/10.1016/j.ibmb.2006.11.009
Peruca RD, Coelho RG, da Silva GG et al (2018) Impacts of soybean-induced defenses on Spodoptera frugiperda (Lepidoptera: Noctuidae) development. Arthropod Plant Interact 12:257–266. https://doi.org/10.1007/s11829-017-9565-x
R Core Team (2020) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. https://www.R-project.org/
Raymond B, Sayyed AH, Wright DJ (2007) Host plant and population determine the fitness costs of resistance to Bacillus thuringiensis. Biol Lett 3:83–86. https://doi.org/10.1098/rsbl.2006.0560
Raymond B, Wright DJ, Bonsall MB (2011) Effects of host plant and genetic background on the fitness costs of resistance to Bacillus thuringiensis. Heredity (edinb) 106:281–288. https://doi.org/10.1038/hdy.2010.65
Rehan A, Freed S (2014) Selection, mechanism, cross resistance and stability of spinosad resistance in Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae). Crop Prot 56:10–15. https://doi.org/10.1016/j.cropro.2013.10.013
Rehan A, Freed S (2015) Lethal and sub-lethal effects of spinosad on the life-history traits of army worm, Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae), and its fitness cost of resistance. Entomol Res 45:247–253. https://doi.org/10.1111/1748-5967.12117
Roush RT, Daly JC (1990) The role of population genetics in resistance research and management BT-pesticide resistance in arthropods. In: Roush RT, Tabashnik BE (eds) Pesticide resistance in arthropods. Springer US, Boston, MA, pp 97–152. https://doi.org/10.1007/978-1-4684-6429-0_5
Roush RT, McKenzie JA (1987) Ecological genetics of insecticide and acaricide resistance. Annu Rev Entomol 32:361–380. https://doi.org/10.1146/annurev.en.32.010187.002045
Salgado VL, Sparks TC (2010) The spinosyns: chemistry, biochemistry, mode of action, and resistance. In: Gilbert LI, Gill SS (eds) Insect control: biological and synthetic agents. Academic Press, New York, pp 207–243
Santos-Amaya OF, Tavares CS, Rodrigues JVC, Oliveira EE, Guedes RNC, Pereira EJG (2022) Strong Fitness costs of fall armyworm resistance to dual-gene Bt maize are magnified on less-suitable host-crop cultivars. Agronomy 12:682. https://doi.org/10.3390/agronomy12030682
Schuler MA (2011) P450s in plant–insect interactions. Biochim Biophys Acta (BBA)-Proteins Proteomics 1814:36–45. https://doi.org/10.1016/j.bbapap.2010.09.012
Sial AA, Brunner JF, Garczynski SF (2011) Biochemical characterization of chlorantraniliprole and spinetoram resistance in laboratory-selected obliquebanded leafroller, Choristoneura rosaceana (Harris) (Lepidoptera: Tortricidae). Pestic Biochem Physiol 99:274–279. https://doi.org/10.1016/j.pestbp.2011.01.006
Silva WM, Berger M, Bass C et al (2016) Mutation (G275E) of the nicotinic acetylcholine receptor α6 subunit is associated with high levels of resistance to spinosyns in Tuta absoluta (Meyrick)(Lepidoptera: Gelechiidae). Pestic Biochem Physiol 131:1–8. https://doi.org/10.1016/j.pestbp.2016.02.006
Silva DM, de Bueno AF, Andrade K et al (2017) Biology and nutrition of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on different food sources. Sci Agric 74:18–31. https://doi.org/10.1590/1678-992x-2015-0160
Sivakoff FS, Rosenheim JA, Dutilleul P, Carrière Y (2013) Influence of the surrounding landscape on crop colonization by a polyphagous insect pest. Entomol Exp Appl 149:11–21. https://doi.org/10.1111/eea.12101
Sparks TC, Crouse GD, Durst G (2001) Natural products as insecticides: the biology, biochemistry and quantitative structure–activity relationships of spinosyns and spinosoids. Pest Manag Sci 57:896–905. https://doi.org/10.1002/ps.358
Sparks TC, Dripps JE, Watson GB, Paroonagian D (2012) Resistance and cross-resistance to the spinosyns–A review and analysis. Pestic Biochem Physiol 102:1–10. https://doi.org/10.1016/j.pestbp.2011.11.004
Vandenhole M, Dermauw W, Van Leeuwen T (2020) Short term transcriptional responses of P450s to phytochemicals in insects and mites. Curr Opin Insect Sci 43:117–127. https://doi.org/10.1016/j.cois.2020.12.002
Venables B, Ripley B (2002) Modern applied statistics with S. Springer, New York. https://doi.org/10.1007/978-0-387-21706-2
Wan Y, Yuan G, He B et al (2018) Foccα6, a truncated nAChR subunit, positively correlates with spinosad resistance in the western flower thrips, Frankliniella occidentalis (Pergande). Insect Biochem Mol Biol 99:1–10. https://doi.org/10.1016/j.ibmb.2018.05.002
Wang D, Qiu X, Ren X et al (2009) Resistance selection and biochemical characterization of spinosad resistance in Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae). Pestic Biochem Physiol 95:90–94. https://doi.org/10.1016/j.pestbp.2009.07.003
Wang D, Qiu X, Wang H et al (2010) Reduced fitness associated with spinosad resistance in Helicoverpa armigera. Phytoparasitica 38:103–110. https://doi.org/10.1007/s12600-009-0077-9
Wang R, Tetreau G, Wang P (2016) Effect of crop plants on fitness costs associated with resistance to Bacillus thuringiensis toxins Cry1Ac and Cry2Ab in cabbage loopers. Sci Rep 6:20959. https://doi.org/10.1038/srep20959
Wang J, Ma H, Zuo Y et al (2020a) CRISPR-mediated gene knockout reveals nicotinic acetylcholine receptor (nAChR) subunit α6 as a target of spinosyns in Helicoverpa armigera. Pest Manag Sci 76:2925–2931. https://doi.org/10.1002/ps.5889
Wang X, Ma Y, Wang F et al (2020b) Disruption of nicotinic acetylcholine receptor α6 mediated by CRISPR/Cas9 confers resistance to spinosyns in Plutella xylostella. Pest Manag Sci 76:1618–1625. https://doi.org/10.1002/ps.5689
Williams JL, Ellers-Kirk C, Orth RG et al (2011) Fitness cost of resistance to Bt cotton linked with increased gossypol content in pink bollworm larvae. PLoS ONE 6:e21863. https://doi.org/10.1371/journal.pone.0021863
Wyss CF, Young HP, Shukla J, Roe RM (2003) Biology and genetics of a laboratory strain of the tobacco budworm, Heliothis virescens (Lepidoptera: Noctuidae), highly resistant to spinosad. Crop Prot 22:307–314. https://doi.org/10.1016/S0261-2194(02)00153-9
Zimmer CT, Garrood WT, Puinean AM et al (2016) A CRISPR/Cas9 mediated point mutation in the alpha 6 subunit of the nicotinic acetylcholine receptor confers resistance to spinosad in Drosophila melanogaster. Insect Biochem Mol Biol 73:62–69. https://doi.org/10.1016/j.ibmb.2016.04.007
Zuo Y, Xue Y, Lu W et al (2020) Functional validation of nicotinic acetylcholine receptor (nAChR) α6 as a target of spinosyns in Spodoptera exigua utilizing the CRISPR/Cas9 system. Pest Manag Sci 76:2415–2422. https://doi.org/10.1002/ps.5782
Acknowledgements
We thank Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for Granting a PhD scholarship to the first author (Grant #2019/06217-8) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for Granting a research fellowship to CO (Grant #314160/2020-5)
Funding
This work was supported by São Paulo Research Foundation (FAPESP) (Grant #2019/06217-8).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human or animal rights
“This article does not contain any studies with human participants or animals performed by any of the authors.”
Additional information
Communicated by Gaelle Le Goff.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kanno, R.H., Guidolin, A.S., Padovez, F.E.O. et al. Fitness costs associated with spinetoram resistance in Spodoptera frugiperda is driven by host plants. J Pest Sci 96, 1625–1635 (2023). https://doi.org/10.1007/s10340-023-01614-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-023-01614-8