Abstract
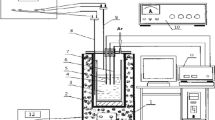
The electrochemical reduction of Zr4+ (complex) ions in NaCl-KCl-K2ZrF6 molten salt on Pt electrode was investigated using cyclic voltammetry and square wave voltammetry at 1023 K. Two cathodic reduction peaks related to Zr4+/Zr2+ and Zr2+/Zr steps were observed in the cyclic voltammograms. The result was also confirmed by square wave voltammetry. The diffusion coefficient of Zr4+ (complex) ions at 1023 K in NaCl-KCl-K2ZrF6 melt, measured by cyclic voltammetry, is about 4.22×10−6 cm2/s. The characterization of the deposits obtained by potentiostatic electrolysis at different potentials was investigated by XRD, and the results were well consistent with the electrochemical reduction mechanism of Zr4+ (complex) ions
Similar content being viewed by others
References
Polyakova L.P. and Stangrit P.T., Cathodic processes at electrolysis of chloride and chloride-fluoride melts of zirconium, Electrochim. Acta, 1982, 27(11): 1641.
Chen G.S., Okido M., and Oki T., Electrochemical studies of zirconium and hafnium in alkali chloride and alkali fluoride-chloride molten salts, J. Appl. Electrochem., 1990, 20(1):77.
Basile F., Chassaing E., and Lorthioir G., Electrochemical reduction of ZrCl4 in molten NaCl, CsCl and KCl-LiCl and chemical reactions coupled to the electrodeposition of zirconium, J. Appl. Electrochem., 1981, 11(5): 645.
Sakamura Y., Zirconium behavior in molten LiCl-KCl eutectic, J. Electrochem. Soc., 2004, 151(3): C187.
Swaroop B. and Flengas S.N., Thermodynamic and electrochemical properties of zirconium chlorides in alkali chloride melts, Can. J. Chem., 1966, 44(2): 199.
Ghosh S., Vandarkuzhali S., Venkatesh P., Seenivasan G., Subramanian T., Reddy B.P., and Nagarajan K., Electrochemical studies on the redox behaviour of zirconium in molten LiCl-KCl eutectic, J. Electroanal. Chem., 2009, 627(1–2):15.
Chen Z., Zhang M.L., Han W., Li S.J., Wang J., Yan Y.D., and Hou Z.Y., Electrochemical reduction of Zr(IV) in the LiCl-KCl molten salt, Rare Met. Mater. Eng., 2009, 38(3):456.
Qiao Z.Y., Zhao G.W., Zhang J.Y., and Li G.X., A study of some electrochemical properties of molten salt without containing titanium, J. Beijing Univ. Iron Steel Technol., 1980(3): 88.
Bard A.J. and Faulkner L.R., Electrochemical Methods Fundamentals and Applications, 2nd ed., John Wiley & Sons, New York, 2000: 157.
Reddy B.P., Vandarkuzhali S., Subramanian T., and Venkatesh P., Electrochemical studies on the redox mechanism of uranium chloride in molten LiCl-KCl eutectic, Electrochim. Acta, 2004, 49: 2471.
Nicholson R.S. and Shain I., Theory of stationary electrode polarography: single scan and cyclic methods applied to reversible, irreversible, and kinetic systems, Anal. Chem., 1964, 36(4): 706.
Pletcher D., Greff R., Peat R., Peter L.M., and Robinson J., Instrumental Methods in Electrochemistry, Woodhead Publishing Ltd., Cambridge, 2001: 266.
Osteryoung J.G. and Osteryoung R.A., Square wave voltammetry, Anal. Chem., 1985, 57: 101.
Chamelot P., Lafage B., and Taxil P., Using square-wave voltammetry to monitor molten alkaline fluoride baths for electrodeposition of niobium, Electrochim. Acta, 1997, 43(5–6): 607.
Zachowski E.J., Wojciechowski M., and Osteryoung J., The analytical application of square-wave voltammetry, Anal. Chim. Acta, 1986, 183: 47.
Biggs T. and Taylor S.S., The hardening of platinum alloys for potential jewellery application, Platinum Met. Rev., 2005, 49: 14.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Y., Xu, Z., Chen, S. et al. Electrochemical behavior of zirconium in molten NaCl-KCl-K2ZrF6 system. Rare Metals 30, 8–13 (2011). https://doi.org/10.1007/s12598-011-0187-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-011-0187-7