Abstract
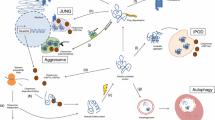
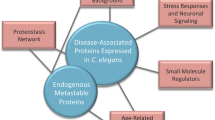
Proteome imbalance can lead to protein misfolding and aggregation which is associated with pathologies. Protein aggregation can also be an active, organized process and can be exploited by cells as a survival strategy. In adverse conditions, it is beneficial to deposit the proteins in a condensate rather degrading and resynthesizing. Membraneless organelles (MLOs) are biological condensates formed through liquid–liquid phase separation (LLPS), involving cellular components such as nucleic acids and proteins. LLPS is a regulated process, which when perturbed, can undergo a transition from a physiological liquid condensate to pathological solid-like protein aggregates. To understand how the MLO-associated proteins (MLO-APs) behave during aging, we performed a comparative meta-analysis with age-related proteome of C. elegans. We found that the MLO-APs are highly abundant throughout the lifespan in wild-type and long-lived daf-2 mutant animals. Interestingly, they are aggregating more in long-lived mutant animals compared to the age matched wild-type and short-lived daf-16 and hsf-1 mutant animals. GO term analysis revealed that the cell cycle and embryonic development are among the top enriched processes in addition to RNP components in aggregated proteome. Considering antagonistic pleotropic nature of these developmental genes and post mitotic status of C. elegans, we assume that these proteins phase transit during post development. As the organism ages, these MLO-APs either mature to become more insoluble or dissolve in uncontrolled manner. However, in the long-lived daf-2 mutant animals, the MLOs may attain protective states due to extended availability and association of molecular chaperones.






Similar content being viewed by others
References
Alberti S, Carra S (2018) Quality control of membraneless organelles. J Mol Biol 430:4711–4729. https://doi.org/10.1016/j.jmb.2018.05.013
Alberti S, Hyman AA (2016) Are aberrant phase transitions a driver of cellular aging? BioEssays 38:959–968. https://doi.org/10.1002/bies.201600042
Alberti S, Hyman AA (2021) Biomolecular condensates at the nexus of cellular stress, protein aggregation disease and ageing. Nat Rev Mol Cell Biol 22:196–213. https://doi.org/10.1038/s41580-020-00326-6
Alberti S, Dormann D (2019) Liquid-liquid phase separation in disease. Annu Rev Genet 53:171–194. https://doi.org/10.1146/annurev-genet-112618-043527
Alberti S (2017) Phase separation in biology. Curr Biol 27:R1097–R1102. https://doi.org/10.1016/j.cub.2017.08.069
Balch WE, Morimoto RI, Dillin A, Kelly JW (2008) Adapting proteostasis for disease intervention. Science 319:916–919. https://doi.org/10.1126/science.1141448
Banani SF, Lee HO, Hyman AA, Rosen MK (2017) Biomolecular condensates: organizers of cellular biochemistry. Nat Rev Mol Cell Biol 18:285–298. https://doi.org/10.1038/nrm.2017.7
Becker LA, Gitler AD (2018) A neurodegenerative-disease protein forms beneficial aggregates in healthy muscle. Nature 563:477–478. https://doi.org/10.1038/d41586-018-07141-2
Boczek EE, Fürsch J, Niedermeier ML, Jawerth L, Jahnel M, Ruer-Gruß M, Kammer KM, Heid P, Mediani L, Wang J, Yan X, Pozniakovski A, Poser I, Mateju D, Hubatsch L, Carra S, Alberti S, Hyman AA, Stengel F (2021) HspB8 prevents aberrant phase transitions of FUS by chaperoning its folded RNA-binding domain. Elife 10:e69377. https://doi.org/10.7554/eLife.69377
Boeynaems S, Alberti S, Fawzi NL, Mittag T, Polymenidou M, Rousseau F, Schymkowitz J, Shorter J, Wolozin B, Van Den Bosch L, Tompa P, Fuxreiter M (2018) Protein phase separation: a new phase in cell biology. Trends Cell Biol 28:420–435. https://doi.org/10.1016/j.tcb.2018.02.004
Bohnert KA, Kenyon C (2017) A lysosomal switch triggers proteostasis renewal in the immortal C. elegans germ lineage. Nature 551:629–633. https://doi.org/10.1038/nature24620
Boronat S, Cabrera M, Hidalgo E (2021) Spatial sequestration of misfolded proteins as an active chaperone-mediated process during heat stress. Curr Genet 67:237–243. https://doi.org/10.1007/s00294-020-01135-2
Bose M, Mahamid J, Ephrussi A (2022) Liquid-to-solid phase transition of oskar RNP granules is essential for their function in the Drosophila germline. Cell 185:1–17. https://doi.org/10.1016/j.cell.2022.02.022
Bryantsev AL, Chechenova MB, Shelden EA (2007) Recruitment of phosphorylated small heat shock protein Hsp27 to nuclear speckles without stress. Exp Cell Res 313:195–209. https://doi.org/10.1016/j.yexcr.2006.10.004
Cereghetti G, Wilson-Zbinden C, Kissling VM, Diether M, Arm A, Yoo H, Piazza I, Saad S, Picotti P, Drummond DA, Sauer U, Dechant R, Peter M (2021) Reversible amyloids of pyruvate kinase couple cell metabolism and stress granule disassembly. Nat Cell Biol 23:1085–1094. https://doi.org/10.1038/s41556-021-00760-4
Chen B, Retzlaff M, Roos T, Frydman J (2011) Cellular strategies of protein quality control. Cold Spring Harb Perspect Biol 3:a004374. https://doi.org/10.1101/cshperspect.a004374
Chen D, Pan K, Palter JE, Kapahi P (2007) Longevity determined by developmental arrest genes in Caenorhabditis elegans. Aging Cell 64:525–533. https://doi.org/10.1111/j.1474-9726.2007.00305.x
Ciryam P, Tartaglia GG, Morimoto RI, Dobson CM, Vendruscolo M (2013) Widespread aggregation and neurodegenerative diseases are associated with supersaturated proteins. Cell Rep 3:781–790. https://doi.org/10.1016/j.celrep.2013.09.043
Ciryam P, Kundra R, Morimoto RI, Dobson CM, Vendruscolo M (2015) Supersaturation is a major driving force for protein aggregation in neurodegenerative diseases. Trends Pharmacol Sci 36:72–77. https://doi.org/10.1016/j.tips.2014.12.004
Ciryam P, Antalek M, Cid F, Tartaglia GG, Dobson CM, Guettsches AK, Eggers B, Vorgerd M, Marcus K, Kley RA, Morimoto RI, Vendruscolo M, Weihl CC (2019) A metastable subproteome underlies inclusion formation in muscle proteinopathies. Acta Neuropathol Commun 7:197. https://doi.org/10.1186/s40478-019-0853-9
Collier NC, Schlesinger MJ (1986) The dynamic state of heat shock proteins in chicken embryo fibroblasts. J Cell Biol 103:1495–1507. https://doi.org/10.1083/jcb.103.4.1495
Curran SP, Ruvkun G (2007) Lifespan regulation by evolutionarily conserved genes essential for viability. PLoS Genet 3:e56. https://doi.org/10.1371/journal.pgen.0030056
Darling AL, Liu Y, Oldfield CJ, Uversky VN (2018) Intrinsically disordered proteome of human membrane-less organelles. Proteomics 18:e1700193. https://doi.org/10.1002/pmic.201700193
David DC, Ollikainen N, Trinidad JC, Cary MP, Burlingame AL, Kenyon C (2010) Widespread protein aggregation as an inherent part of aging in C. elegans. PLoS Biol 8:e1000450. https://doi.org/10.1371/journal.pbio.1000450
Dottermusch M, Lakner T, Peyman T, Klein M, Walz G, Neumann-Haefelin E (2016) Cell cycle controls stress response and longevity in C. elegans. Aging (Albany NY) 8:2100–2126. https://doi.org/10.18632/aging.101052
Fassler JS, Skuodas S, Weeks DL, Phillips BT (2021) Protein aggregation and disaggregation in cells and development. J Mol Biol 433:167215. https://doi.org/10.1016/j.jmb.2021.167215
Franzmann TM, Alberti S (2019) Protein phase separation as a stress survival strategy. Cold Spring Harb Perspect Biol 11:a034058. https://doi.org/10.1101/cshperspect.a034058
Ganassi M, Mateju D, Bigi I, Mediani L, Poser I, Lee HO et al (2016) A surveillance function of the HSPB8-BAG3-HSP70 chaperone complex ensures stress granule integrity and dynamism. Mol Cell 63:796–810. https://doi.org/10.1016/j.molcel.2016.07.021
Gu J, Liu Z, Zhang S, Li Y, Xia W, Wang C, Xiang H, Liu Z, Tan L, Fang Y, Liu C, Li D (2020) Hsp40 proteins phase separate to chaperone the assembly and maintenance of membraneless organelles. Proc Natl Acad Sci U S A 117:31123–31133. https://doi.org/10.1073/pnas.2002437117
Gu J, Wang C, Hu R, Li Y, Zhang S, Sun Y, Wang Q, Li D, Fang Y, Liu C (2021) Hsp70 chaperones TDP-43 in dynamic, liquid-like phase and prevents it from amyloid aggregation. Cell Res 31:1024–1027. https://doi.org/10.1038/s41422-021-00526-5
Hartl FU (2016) Cellular homeostasis and aging. Annu Rev Biochem 85:1–4. https://doi.org/10.1146/annurev-biochem-011116-110806
Hipp MS, Park SH, Hartl FU (2014) Proteostasis impairment in protein-misfolding and -aggregation diseases. Trends Cell Biol 24:506–514. https://doi.org/10.1016/j.tcb.2014.05.003
Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, Bohr VA (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15:565–581. https://doi.org/10.1038/s41582-019-0244-7
Hsu AL, Murphy CT, Kenyon C (2003) Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300:1142–1145. https://doi.org/10.1126/science.1083701
Huiting W, Dekker SL, van der Lienden JCJ, Mergener R, Musskopf MK, Furtado GV, Gerrits E, Coit D, Oghbaie M, Di Stefano LH, Schepers H, van Waarde-Verhagen MAWH, Couzijn S, Barazzuol L, LaCava J, Kampinga HH, Bergink S (2022) Targeting DNA topoisomerases or checkpoint kinases results in an overload of chaperone systems, triggering aggregation of a metastable subproteome. Elife 11:e70726. https://doi.org/10.7554/eLife.70726
Klaips CL, Jayaraj GG, Hartl FU (2018) Pathways of cellular proteostasis in aging and disease. J Cell Biol 217:51–63. https://doi.org/10.1083/jcb.201709072
Labbadia J, Morimoto RI (2015) The biology of proteostasis in aging and disease. Annu Rev Biochem 84:435–464. https://doi.org/10.1146/annurev-biochem-060614-033955
Lechler MC, Crawford ED, Groh N, Widmaier K, Jung R, Kirstein J, Trinidad JC, Burlingame AL, David DC (2017) Reduced insulin/IGF-1 signaling restores the dynamic properties of key stress granule proteins during aging. Cell Rep 18:454–467. https://doi.org/10.1016/j.celrep.2016.12.033
Li D, Liu C (2022) Spatiotemporal dynamic regulation of membraneless organelles by chaperone networks. Trends in Cell Biol 32:1–3. https://doi.org/10.1016/j.tcb.2021.08.004
Li Y, Gu J, Wang C, Hu J, Zhang S, Liu C, Zhang S, Fang Y, Li D (2022) Hsp70 exhibits a liquid-liquid phase separation ability and chaperones condensed FUS against amyloid aggregation. iScience 25(6):104356. https://doi.org/10.1016/j.isci.2022.104356
Lu S, Hu J, Arogundade OA, Goginashvili A, Vazquez-Sanchez S, Diedrich JK, Gu J, Blum J, Oung S, Ye Q, Yu H, Ravits J, Liu C, Yates JR 3rd, Cleveland DW (2022) Heat shock chaperone HSPB1 regulates cytoplasmic TDP-43 phase separation and liquid-to-gel transition. Nat Cell Biol 2022. https://doi.org/10.1038/s41556-022-00988-8
Mateju D, Franzmann TM, Patel A, Kopach A, Boczek EE, Maharana S, Lee HO, Carra S, Hyman AA, Alberti S (2017) An aberrant phase transition of stress granules triggered by misfolded protein and prevented by chaperone function. EMBO J 36:1669–1687. https://doi.org/10.15252/embj.201695957
Mészáros B, Erdős G, Szabó B, Schád É, Tantos Á, Abukhairan R, Horváth T, Murvai N, Kovács OP, Kovács M, Tosatto SCE, Tompa P, Dosztányi Z, Pancsa R (2020) PhaSePro: the database of proteins driving liquid-liquid phase separation. Nucleic Acids Res 48:D360–D367. https://doi.org/10.1093/nar/gkz848
Miller SB, Mogk A, Bukau B (2015) Spatially organized aggregation of misfolded proteins as cellular stress defense strategy. J Mol Biol 427:1564–1574. https://doi.org/10.1016/j.jmb.2015.02.006
Ning W, Guo Y, Lin S, Mei B, Wu Y, Jiang P, Tan X, Zhang W, Chen G, Peng D, Chu L, Xue Y (2020) DrLLPS: a data resource of liquid-liquid phase separation in eukaryotes. Nucleic Acids Res 48:D288–D295. https://doi.org/10.1093/nar/gkz1027
Ong JY, Torres JZ (2020) Phase separation in cell division. Mol Cell 80:9–20. https://doi.org/10.1016/j.molcel.2020.08.007
Orti F, Navarro AM, Rabinovich A, Wodak SJ, Marino-Buslje C (2021) Insight into membraneless organelles and their associated proteins: drivers, clients and regulators. Comput Struct Biotechnol J 19:3964–3977. https://doi.org/10.1016/j.csbj.2021.06.042
Pu YZ, Wan QL, Ding AJ, Luo HR, Wu GS (2017) Quantitative proteomics analysis of Caenorhabditis elegans upon germ cell loss. J Proteomics 156:85–93. https://doi.org/10.1016/j.jprot.2017.01.011
Putnam A, Cassani M, Smith J, Seydoux G (2019) A gel phase promotes condensation of liquid P granules in Caenorhabditis elegans embryos. Nat Struc Mol Biol 26:220–226. https://doi.org/10.1038/s41594-019-0193-2
Riback JA, Katanski CD, Kear-Scott JL, Pilipenko EV, Rojek AE, Sosnick TR, Drummond DA (2017) Stress-triggered phase separation is an adaptive, evolutionarily tuned response. Cell 168:1028–1040. https://doi.org/10.1016/j.cell.2017.02.027
Ross CA, Poirier MA (2004) Protein aggregation and neurodegenerative disease. Nat Med 10:S10-17. https://doi.org/10.1038/nm1066
Saad S, Cereghetti G, Feng Y, Picotti P, Peter M, Dechant R (2017) Reversible protein aggregation is a protective mechanism to ensure cell cycle restart after stress. Nat Cell Biol 19:1202–1213. https://doi.org/10.1038/ncb3600
Shin Y, Brangwynne CP (2017) Liquid phase condensation in cell physiology and disease. Science 357:eaaf4382. https://doi.org/10.1126/science.aaf4382
Skuodas S, Clemons A, Hayes M, Goll A, Zora B, Weeks DL, Phillips BT, Fassler JS (2020) The ABCF gene family facilitates disaggregation during animal development. Mol Biol Cell 31:1324–1345. https://doi.org/10.1091/mbc.E19-08-0443
Soto C, Pritzkow S (2018) Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat Neurosci 21:1332–1340. https://doi.org/10.1038/s41593-018-0235-9
Turner BE, Basecke SM, Bazan GC, Dodge ES, Haire CM, Heussman DJ, Johnson CL, Mukai CK, Naccarati AM, Norton SJ, Sato JR, Talavera CO, Wade MV, Hillers KJ (2015) Proteomic identification of germline proteins in Caenorhabditis elegans. Worm 4:e1008903. https://doi.org/10.1080/21624054.2015.1008903
Tyanova S, Temu T, Sinitcyn P, Carlson A, Hein MY, Geiger T, Mann M, Cox J (2016) The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat Methods 13:731–740. https://doi.org/10.1038/nmeth.3901
Ungelenk S, Moayed F, Ho CT, Grousl T, Scharf A, Mashaghi A, Tans S, Mayer MP, Mogk A, Bukau B (2016) Small heat shock proteins sequester misfolding proteins in near-native conformation for cellular protection and efficient refolding. Nat Commun 7:13673. https://doi.org/10.1038/ncomms13673
Updike DL, Knutson AK, Egelhofer TA, Campbell AC, Strome S (2014) Germ-granule components prevent somatic development in the C. elegans germline. Curr Biol 24:970–975. https://doi.org/10.1016/j.cub.2014.03.015
Uversky VN (2021) Recent developments in the field of intrinsically disordered proteins: intrinsic disorder-based emergence in cellular biology in light of the physiological and pathological liquid-liquid phase transitions. Annu Rev Biophys 50:135–156. https://doi.org/10.1146/annurev-biophys-062920-063704
van den IJssel PR, Overkamp P, Bloemendal H, de Jong WW (1998) Phosphorylation of alphaB-crystallin and HSP27 is induced by similar stressors in HeLa cells. Biochem Biophys Res Commun 247:518–523. https://doi.org/10.1006/bbrc.1998.8699
Vecchi G, Sormanni P, Mannini B, Vandelli A, Tartaglia GG, Dobson CM, Hartl FU, Vendruscolo M (2020) Proteome-wide observation of the phenomenon of life on the edge of solubility. Proc Natl Acad Sci U S A 117:1015–1020. https://doi.org/10.1073/pnas.1910444117
Venz R, Pekec T, Katic I, Ciosk R, Ewald CY (2021) End-of-life targeted degradation of DAF-2 insulin/IGF-1 receptor promotes longevity free from growth-related pathologies. eLife 10:e71335. https://doi.org/10.7554/eLife.71335
Verma K, Verma M, Chaphalkar A, Chakraborty K (2021) Recent advances in understanding the role of proteostasis. Fac Rev 10:72. https://doi.org/10.12703/r/10-72
Vos MJ, Kanon B, Kampinga HH (2009) HSPB7 is a SC35 speckle resident small heat shock protein. Biochim. Biophys. Acta (BBA). Mol Cell Res 1793:1343–1353. https://doi.org/10.1016/j.bbamcr.2009.05.005
Wallace EW, Kear-Scott JL, Pilipenko EV, Schwartz MH, Laskowski PR, Rojek AE, Katanski CD, Riback JA, Dion MF, Franks AM, Airoldi EM, Pan T, Budnik BA, Drummond DA (2015) Reversible, specific, active aggregates of endogenous proteins assemble upon heat stress. Cell 162:1286–1298. https://doi.org/10.1016/j.cell.2015.08.041
Walther DM, Kasturi P, Zheng M, Pinkert S, Vecchi G, Ciryam P, Morimoto RI, Dobson CM, Vendruscolo M, Mann M, Hartl FU (2015) Widespread proteome remodeling and aggregation in aging C. elegans. Cell 161:919–932. https://doi.org/10.1016/j.cell.2015.03.032
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York. https://ggplot2.tidyverse.org
Wiegand T, Hyman AA (2020) Drops and fibers - how biomolecular condensates and cytoskeletal filaments influence each other. Emerg Top Life Sci 4:247–261. https://doi.org/10.1042/ETLS20190174
Xue B, Dunbrack RL, Williams RW, Dunker AK, Uversky VN (2010) PONDR-FIT: a meta-predictor of intrinsically disordered amino acids. Biochim Biophys Acta 1804:996–1010. https://doi.org/10.1016/j.bbapap.2010.01.011
Yoo H, Bard JAM, Pilipenko EV, Drummond DA (2022) Chaperones directly and efficiently disperse stress-triggered biomolecular condensates. Mol Cell 82:741–755. https://doi.org/10.1016/j.molcel.2022.01.005
You K, Huang Q, Yu C, Shen B, Sevilla C, Shi M, Hermjakob H, Chen Y, Li T (2020) PhaSepDB: a database of liquid-liquid phase separation related proteins. Nucleic Acids Res 48:D354–D359. https://doi.org/10.1093/nar/gkz847
Zhou Y, Zhou B, Pache L, Chang M, Khodabakhshi AH, Tanaseichuk O, Benner C, Chanda SK (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Comm 10:1523. https://doi.org/10.1038/s41467-019-09234-6
Acknowledgements
The authors acknowledge BioX Centre, SBB at IIT Mandi for providing facilities.
Funding
This work was supported by SERB-CRG (Science and Engineering Research Board Core Research Grant, CRG/2021/007177 to P.K) and IIT Mandi seed grant (IITM/SG/PKS/71 to P.K). P.M acknowledges Ministry of Education and IIT Mandi for HTRA fellowship and P.P acknowledges DST for INSPIRE fellowship.
Author information
Authors and Affiliations
Contributions
P.M and P.K conceived the project. P.M performed analysis and made draft figures. P.P used R package to make figures. P.M, P.P, and P.K analyzed the data and performed the figure visualizations. P.M and P.K wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mukherjee, P., Panda, P. & Kasturi, P. A comparative meta-analysis of membraneless organelle-associated proteins with age related proteome of C. elegans. Cell Stress and Chaperones 27, 619–631 (2022). https://doi.org/10.1007/s12192-022-01299-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-022-01299-5