Abstract
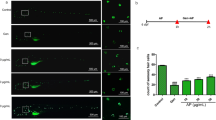
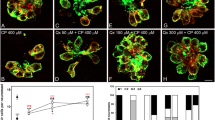
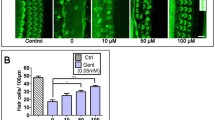
Aminoglycoside antibiotics, including gentamicin (GM), induce delayed ototoxic effects such as hearing loss after prolonged use, which results from the death of hair cells. However, the mechanisms underlying the ototoxicity of aminoglycosides warrant further investigation, and there are currently no effective drugs in the clinical setting. Herein, the therapeutic effect of the flavonoid compound rutin against the ototoxic effects of GM in zebrafish hair cells was investigated. Animals incubated with rutin (100–400 µmol/L) were protected against the pernicious effects of GM (200 µmol/L). We found that rutin improves hearing behavior in zebrafish, and rutin was effective in reducing the number of Tunel-positive cells in the neuromasts of the zebrafish lateral line and promoting cell proliferation after exposure to GM. Subsequently, rutin exerted a protective effect against GM-induced cell death in HEI-OC1 cells and could limit the production of cytosolic reactive oxygen species (ROS) and diminish the percentage of apoptotic cells. Additionally, the results of the proteomic analysis revealed that rutin could effectively inhibit the expression of necroptosis and apoptosis related genes. Meanwhile, molecular docking analysis revealed a high linking activity between the molecular docking of rutin and STAT1 proteins. The protection of zebrafish hair cells or HEI-OC1 cells from GM-induced ototoxicity by rutin was attenuated by the introduction of STAT1 activator. Finally, we demonstrated that rutin significantly improves the bacteriostatic effect of GM by in vitro experiments, emphasising its clinical application value. In summary, these results collectively unravel a novel therapeutic role for rutin as an otoprotective drug against the adverse effects of GM.







Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
References
Selimoglu E (2007) Aminoglycoside-induced ototoxicity. Curr Pharm Des 13(1):119–126. https://doi.org/10.2174/138161207779313731
Steyger PS (2021) Mechanisms of Ototoxicity and Otoprotection. Otolaryngol Clin North Am 54(6):1101–1115. https://doi.org/10.1016/j.otc.2021.08.007
Kros CJ, Steyger PS (2019) Aminoglycoside- and Cisplatin-Induced Ototoxicity: mechanisms and otoprotective strategies. Cold Spring Harb Perspect Med 9(11):a033548. https://doi.org/10.1101/cshperspect.a033548
Wargo KA, Edwards JD (2014) Aminoglycoside-induced nephrotoxicity. J Pharm Pract 27(6):573–577. https://doi.org/10.1177/0897190014546836
Lanvers-Kaminsky C, Ciarimboli G (2017) Pharmacogenetics of drug-induced ototoxicity caused by aminoglycosides and cisplatin. Pharmacogenomics 18(18):1683–1695. https://doi.org/10.2217/pgs-2017-0125
Steyger PS (2021) Mechanisms of Aminoglycoside- and Cisplatin-Induced Ototoxicity. Am J Audiol 30(3S):887–900. https://doi.org/10.1044/2021_AJA-21-00006
Zong Y, Chen F, Li S, Zhang H (2021) (-)-Epigallocatechin-3-gallate (EGCG) prevents aminoglycosides-induced ototoxicity via anti-oxidative and anti-apoptotic pathways. Int J Pediatr Otorhinolaryngol 150:110920. https://doi.org/10.1016/j.ijporl.2021.110920
Ghorbani A (2017) Mechanisms of antidiabetic effects of flavonoid rutin. Biomed Pharmacother 96:305–312. https://doi.org/10.1016/j.biopha.2017.10.001
Negahdari R, Bohlouli S, Sharifi S, Maleki Dizaj S, Rahbar Saadat Y, Khezri K, Jafari S, Ahmadian E et al (2021) Therapeutic benefits of rutin and its nanoformulations. Phytother Res 35(4):1719–1738. https://doi.org/10.1002/ptr.6904
Kamalakkannan N, Stanely Mainzen Prince P (2006) Rutin improves the antioxidant status in streptozotocin-induced diabetic rat tissues. Mol Cell Biochem 293(1–2):211–219. https://doi.org/10.1007/s11010-006-9244-1
Nafees S, Rashid S, Ali N, Hasan SK, Sultana S (2015) Rutin ameliorates cyclophosphamide induced oxidative stress and inflammation in Wistar rats: role of NFκB/MAPK pathway. Chem Biol Interact 231:98–107. https://doi.org/10.1016/j.cbi.2015.02.021
Zheng S, Liu C, Tang D, Zheng Z, Yan R, Wu C, Zuo N, Ma J et al (2022) The protective effect of rutin against the cisplatin-induced cochlear damage in vitro. Neurotoxicology 90:102–111. https://doi.org/10.1016/j.neuro.2022.03.005
Chitnis AB, Nogare DD, Matsuda M (2012) Building the posterior lateral line system in zebrafish. Dev Neurobiol 72(3):234–255. https://doi.org/10.1002/dneu.20962
Park MK, Lee BD, Chae SW, Chi J, Kwon SK, Song JJ (2012) Protective effect of NecroX, a novel necroptosis inhibitor, on gentamicin-induced ototoxicity. Int J Pediatr Otorhinolaryngol 76(9):1265–1269. https://doi.org/10.1016/j.ijporl.2012.05.016
Tripathi S, Parmar D, Fathima S, Raval S, Singh G (2023) Coenzyme Q10, Biochanin A and Phloretin Attenuate Cr(VI)-Induced oxidative stress and DNA damage by stimulating Nrf2/HO-1 pathway in the experimental model. Biol Trace Elem Res 201(5):2427–2441. https://doi.org/10.1007/s12011-022-03358-5
Stolzer I, Schickedanz L, Chiriac MT, López-Posadas R, Grassl GA, Mattner J, Wirtz S, Winner B et al (2022) STAT1 coordinates intestinal epithelial cell death during gastrointestinal infection upstream of Caspase-8. Mucosal Immunol 15(1):130–142. https://doi.org/10.1038/s41385-021-00450-2
Gale JE, Marcotti W, Kennedy HJ, Kros CJ, Richardson GP (2001) FM1-43 dye behaves as a permeant blocker of the hair-cell mechanotransducer channel. J Neurosci 21(18):7013–7025. https://doi.org/10.1523/JNEUROSCI.21-18-07013.2001
Baek S, Tran NTT, Diaz DC, Tsai YY, Acedo JN, Lush ME, Piotrowski T (2022) Single-cell transcriptome analysis reveals three sequential phases of gene expression during zebrafish sensory hair cell regeneration. Dev Cell 57(6):799–819e6. https://doi.org/10.1016/j.devcel.2022.03.001
Nicolson T (2017) The genetics of hair-cell function in zebrafish. J Neurogenet 31(3):102–112. https://doi.org/10.1080/01677063.2017.1342246
Kniss JS, Jiang L, Piotrowski T (2016) Insights into sensory hair cell regeneration from the zebrafish lateral line. Curr Opin Genet Dev 40:32–40. https://doi.org/10.1016/j.gde.2016.05.012
Pittman JT, Lott CS (2014) Startle response memory and hippocampal changes in adult zebrafish pharmacologically-induced to exhibit anxiety/depression-like behaviors. Physiol Behav 17:123:174–179. https://doi.org/10.1016/j.physbeh.2013.10.023
Zheng Z, Tang D, Zhao L, Li W, Han J, Hu B, Nie G, He Y (2020) Liproxstatin-1 protects Hair Cell-Like HEI-OC1 cells and cochlear hair cells against Neomycin Ototoxicity. Oxid Med Cell Longev 2020:1782659. https://doi.org/10.1155/2020/1782659
Ivanov M, Novović K, Malešević M, Dinić M, Stojković D, Jovčić B, Soković M (2022) Polyphenols as Inhibitors of Antibiotic Resistant Bacteria-Mechanisms Underlying Rutin Interference with Bacterial Virulence. Pharmaceuticals (Basel) 15(3):385. https://doi.org/10.3390/ph15030385
Tabolacci E, Tringali G, Nobile V, Duca S, Pizzoferrato M, Bottoni P, Clementi ME (2023) Rutin protects fibroblasts from UVA Radiation through Stimulation of Nrf2 pathway. Antioxid (Basel) 12(4):820. https://doi.org/10.3390/antiox12040820
Shu Y, Li W, Huang M, Quan YZ, Scheffer D, Tian C, Tao Y, Liu X et al (2019) Renewed proliferation in adult mouse cochlea and regeneration of hair cells. Nat Commun 10(1):5530. https://doi.org/10.1038/s41467-019-13157-7
He ZH, Li M, Fang QJ, Liao FL, Zou SY, Wu X, Sun HY, Zhao XY et al (2021) FOXG1 promotes aging inner ear hair cell survival through activation of the autophagy pathway. Autophagy 17(12):4341–4362. https://doi.org/10.1080/15548627.2021.1916194
He Z, Guo L, Shu Y, Fang Q, Zhou H, Liu Y, Liu D, Lu L et al (2017) Autophagy protects auditory hair cells against neomycin-induced damage. Autophagy 13(11):1884–1904. https://doi.org/10.1080/15548627.2017.1359449
Ruhl D, Du TT, Wagner EL, Choi JH, Li S, Reed R, Kim K, Freeman M et al (2019) Necroptosis and apoptosis contribute to Cisplatin and Aminoglycoside Ototoxicity. J Neurosc 39(15):2951–2964. https://doi.org/10.1523/JNEUROSCI.1384-18.2019
Gitika B, Sai Ram M, Sharma SK, Ilavazhagan G, Banerjee PK (2006) Quercetin protects C6 glial cells from oxidative stress induced by tertiary-butylhydroperoxide. Free Radic Res 40(1):95–102. https://doi.org/10.1080/10715760500335447
Singh G, Thaker R, Sharma A, Parmar D (2021) Therapeutic effects of biochanin A, phloretin, and epigallocatechin-3-gallate in reducing oxidative stress in arsenic-intoxicated mice. Environ Sci Pollut Res Int 28(16):20517–20536. https://doi.org/10.1007/s11356-020-11740-w
Tripathi S, Fhatima S, Parmar D, Singh DP, Mishra S, Mishra R, Singh G (2022) Therapeutic effects of CoenzymeQ10, biochanin A and Phloretin against arsenic and chromium induced oxidative stress in mouse (Mus musculus) brain. 3 Biotech 12(5):116. https://doi.org/10.1007/s13205-022-03171-w
Kim HS, Lee MS (2007) STAT1 as a key modulator of cell death. Cell Signal 19(3):454–465. https://doi.org/10.1016/j.cellsig.2006.09.003
Wei TT, Zhang MY, Zheng XH, Xie TH, Wang W, Zou J, Li Y, Li HY et al (2022) Interferon-γ induces retinal pigment epithelial cell ferroptosis by a JAK1-2/STAT1/SLC7A11 signaling pathway in Age-related Macular Degeneration. FEBS J 289(7):1968–1983. https://doi.org/10.1111/febs.16272
Murata H, Yasui Y, Oiso K, Ochi T, Tomonobu N, Yamamoto KI, Kinoshita R, Sakaguchi M (2023) STAT1/3 signaling suppresses axon degeneration and neuronal cell death through regulation of NAD+-biosynthetic and consuming enzymes. Cell Signal 108:110717. https://doi.org/10.1016/j.cellsig.2023.110717
Schmitt NC, Rubel EW, Nathanson NM (2009) Cisplatin-induced hair cell death requires STAT1 and is attenuated by epigallocatechin gallate. J Neurosci 29(12):3843–3851. https://doi.org/10.1523/JNEUROSCI.5842-08.2009
Zhang QX, He XJ, Wong HC (2016) Kindt KS (2016) Functional calcium imaging in zebrafish lateral-line hair cells. Methods Cell Biol. 133:229–52. https://doi.org/10.1016/bs.mcb.2015.12.002
Barta CL, Liu H, Chen L, Giffen KP, Li Y, Kramer KL, Beisel KW, He DZ (2018) RNA-seq transcriptomic analysis of adult zebrafish inner ear hair cells. Sci Data 5:180005. https://doi.org/10.1038/sdata.2018.5
Hung GY, Wu CL, Chou YL, Chien CT, Horng JL, Lin LY (2019) Cisplatin exposure impairs ionocytes and hair cells in the skin of zebrafish embryos. Aquat Toxicol 209:168–177. https://doi.org/10.1016/j.aquatox.2019.02.006
Li M, Liu J, Liu D, Duan X, Zhang Q, Wang D, Zheng Q, Bai X et al (2021) Naringin attenuates cisplatin- and aminoglycoside-induced hair cell injury in the zebrafish lateral line via multiple pathways. J Cell Mol Med 25(2):975–989. https://doi.org/10.1111/jcmm.16158
Li M, Zhao J, Tang Q, Zhang Q, Wang Y, Zhang J, Hao Y, Bai X et al (2021) Lamivudine improves cognitive decline in SAMP8 mice: integrating in vivo pharmacological evaluation and network pharmacology. J Cell Mol Med 25(17):8490–8503. https://doi.org/10.1111/jcmm.16811
Li M, Zhang J, Chen W, Liu S, Liu X, Ning Y, Cao Y, Zhao Y (2023) Supraphysiologic doses of 17β-estradiol aggravate depression-like behaviors in ovariectomized mice possibly via regulating microglial responses and brain glycerophospholipid metabolism. J Neuroinflammation 20(1):204. https://doi.org/10.1186/s12974-023-02889-5
Acknowledgements
This research was funded by the Qingdao Medical and Health Research guidance Project (2022-WJZD107) and the National Natural Science Foundation of China (Nos. 82000979).
Funding
This research was funded by the Qingdao Medical and Health Research guidance Project (2022-WJZD107) and the National Natural Science Foundation of China (Nos. 82000979).
Author information
Authors and Affiliations
Contributions
HY and RY designed the experiments; HY performed the animal experiments; TZ, JL, and DW performed the experiments; KG, HY and WZ analyzed the data, reviewed the data, advised the study, and interpreted the analyzed data; HY and TX wrote the manuscript. The manuscript has been revised and approved by all authors.
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the Animal Ethics Committee (No.20,230,506) of Shandong Provincial Hospital (Jinan, China).
Consent to Participate
Not applicable.
Consent for publication
All authors have read and approve the manuscript and consent to its publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, H., Zong, T., Liu, J. et al. Rutin Attenuates Gentamycin-induced Hair Cell Injury in the Zebrafish Lateral Line via Suppressing STAT1. Mol Neurobiol (2024). https://doi.org/10.1007/s12035-024-04179-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-024-04179-4