Abstract
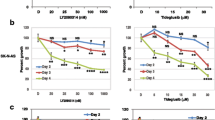
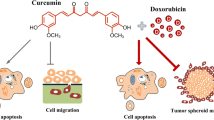
Neuroblastoma, a tumor of the sympathetic nervous system, is one of the most common tumors found in children. Most patients develop resistance to therapy and show poor prognosis, thus there is a need of novel therapeutic agents for the treatment of neuroblastoma. NVP-BEZ235 is a dual Phosphatidylinositol 3-kinase (PI3K)/mammalian target of rapamycin (mTOR) kinase inhibitor that induces apoptosis and suppresses the growth of cancer. Curcumin acts as an anticancer agent in certain cancers. This study investigated the synergetic effect of NVP-BEZ235 and curcumin against neuroblastoma SH-SY5Y cell line. In the current study, the synergic effect of NVP-BEZ235 and curcumin in SH-SY5Y was examined in terms of the cell growth by cell viability and colony forming assay, cell cycle and apoptotic cell death by flow cytometry and mRNA expression levels by quantitative Real Time Polymerase Chain Reaction (qRT-PCR). Curcumin, NVP-BEZ235 or a combination of both, showed cytotoxicity in a dose and time dependent manner in SH-SY5Y cells. 10 µM curcumin and 200 nM NVP-BEZ235 were chosen as combination therapy, as the combination index showed synergism. Colony forming assay showed decrease in cell growth in combination group. The cell cycle distribution for combination group demonstrated a decrease in G0/G1 phase at 48 h. Annexin V showed an anticancer effect in combination group when compared to control group. Moreover, qRT-PCR results showed a significant increase in caspase 3, caspase 7, Bax and p53 genes, while a decrease in Bcl-2 gene expression levels. These findings suggest that combination therapy of NVP-BEZ235 and curcumin may be a promising therapeutic candidate for treatment of neuroblastoma.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed in the present study are available from the corresponding author upon reasonable request.
References
Olsen RR, Otero JH, García-López J, Wallace K, Finkelstein D, Rehg JE, et al. MYCN induces neuroblastoma in primary neural crest cells. Oncogens. 2017;36:5075–82.
Cheung NKV, Dyer MA. Neuroblastoma: developmental biology, cancer genomics, and immunotherapy. Nat Rev Cancer. 2013;13:397.
Zafar A, Wang W, Liu G, Xian W, McKeon F, Zhou J, et al. Targeting the p53-MDM2 pathway for neuroblastoma therapy: rays of hope. Cancer Lett. 2021;496:16–29.
Wang Y, Wang L, Guan S, Cao W, Wang H, Chen Z, et al. Novel ALK inhibitor AZD3463 inhibits neuroblastoma growth by overcoming crizotinib resistance and inducing apoptosis. Sci Rep. 2016. https://doi.org/10.1038/srep19423.
Beck JT, Ismail A, Tolomeo C. Targeting the phosphatidylinositol 3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway: an emerging treatment strategy for squamous cell lung carcinoma. Cancer Treat Rev. 2014;40:980–9.
Altomare DA, Hui QW, Skele KL, De Rienzo A, Klein-Szanto AJ, Godwin AK, et al. AKT and mTOR phosphorylation is frequently detected in ovarian cancer and can be targeted to disrupt ovarian tumor cell growth. Oncogene. 2004;23:5853–7.
Mei H, Wang Y, Lin Z, Tong Q. The mTOR signaling pathway in pediatric neuroblastoma. Cancer Biol Ther. 2013;30:605–15.
Leung E, Kim JE, Rewcastle GW, Finlay GJ, Baguley BC. Comparison of the effects of the PI3K/mTOR inhibitors NVP-BEZ235 and GSK2126458 on tamoxifen-resistant breast cancer cells. Cancer Biol Ther. 2011;11:938–46.
Fatehi D, Soltani A, Ghatrehsamani M. SRT1720, a potential sensitizer for radiotherapy and cytotoxicity effects of NVB-BEZ235 in metastatic breast cancer cells. Pathol Res Pract. 2018;214:889–95.
Xie J, Wang X, Proud CG. mTOR inhibitors in cancer therapy. F1000Res. 2016;5:2078.
Sabatini DM. mTOR and cancer: insights into a complex relationship. Nat Rev Cancer. 2006;6:729–34.
Cho DC, Cohen MB, Panka DJ, Collins M, Ghebremichael M, Atkins MB, et al. The efficacy of the novel dual PI3-kinase/mTOR inhibitor NVP-BEZ235 compared with rapamycin in renal cell carcinoma. Clin Cancer Res. 2010;16:3628–38. https://doi.org/10.1158/1078-0432.CCR-09-3022.
Liu SL, Liu Z, Di Zhang L, Zhu HQ, Guo JH, Zhao M, et al. GSK3β-dependent cyclin D1 and cyclin E1 degradation is indispensable for NVP-BEZ235 induced G0/G1 arrest in neuroblastoma cells. Cell Cycle. 2017;16:2386–95.
Safdie FM, Dorff T, Quinn D, Fontana L, Wei M, Lee C, et al. Fasting and cancer treatment in humans: a case series report. Aging. 2009;1:988–1007.
Agrawal DK, Mishra PK. Curcumin and its analogues: potential anticancer agents. Med Res Rev. 2010;30:818–60. https://doi.org/10.1002/med.20188.
Giordano A, Tommonaro G. Curcumin and cancer. Nutrients. 2019;11:2376.
Shanmugam MK, Rane G, Kanchi MM, Arfuso F, Chinnathambi A, Zayed ME, et al. The multifaceted role of curcumin in cancer prevention and treatment. Molecules. 2015;20:2728–69.
Ye Z, Chen D, Zheng R, Chen H, Xu T, Wang C, et al. Curcumin induced G2/M cycle arrest in SK-N-SH neuroblastoma cells through the ROS-mediated p53 signaling pathway. J Food Biochem. 2021;45: e13888.
Picone P, Nuzzo D, Caruana L, Messina E, Scafidi V, Di Carlo M. Curcumin induces apoptosis in human neuroblastoma cells via inhibition of AKT and Foxo3a nuclear translocation. Free Radic Res. 2014;48:1397–408.
Pisano M, Pagnan G, Dettori MA, Cossu S, Caffa I, Sassu I, et al. Enhanced anti-tumor activity of a new curcumin-related compound against melanoma and neuroblastoma cells. Mol Cancer. 2010;9:1–12. https://doi.org/10.1186/1476-4598-9-137.
Namkaew J, Jaroonwitchawan T, Rujanapun N, Saelee J, Noisa P. Combined effects of curcumin and doxorubicin on cell death and cell migration of SH-SY5Y human neuroblastoma cells. In Vitro Cell Dev Biol Anim. 2018;54:629–39. https://doi.org/10.1007/s11626-018-0288-9.
Jara JA, Rojas D, Castro-Castillo V, Fuentes-Retamal S, Sandoval-Acuña C, Parra E, et al. Novel benzoate-lipophilic cations selectively induce cell death in human colorectal cancer cell lines. Toxicol in Vitro. 2020;65: 104814.
Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55.
Chou TC. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev. 2006;58:621–81.
Zhu H, Shi Y, Jiao X, Yang G, Wang R, Yuan Y. Synergistic antitumor effect of dual PI3K and mTOR inhibitor NVP-BEZ235 in combination with cisplatin on drug-resistant non-small cell lung cancer cell. Oncol Lett. 2020. https://doi.org/10.3892/ol.2020.12189.
Park JR, Eggert A, Caron H. Neuroblastoma: biology, prognosis, and treatment. Hematol Oncol Clin North Am. 2010;24:65–86.
Modak S, Cheung NKV. Neuroblastoma: therapeutic strategies for a clinical enigma. Cancer Treat Rev. 2010;24:307–17.
Fong A, Park JR. High-risk neuroblastoma: a therapy in evolution. Pediatr Hematol Oncol. 2009;26:539–48.
Fine HA, Keith G Dear ll B, Loefler lay S, Black PM, Canellos GP. Meta-analysis of radiation therapy with and without adjuvant chemotherapy for malignant gliomas in adults. Available from: https://onlinelibrary.wiley.com/terms-and-conditions
Balmaceda C, Peereboom D, Pannullo S, Cheung YKK, Fisher PG, Alavi J, et al. Multi-institutional phase II study of temozolomide administered twice daily in the treatment of recurrent high-grade gliomas. Cancer. 2008;112:1139–46.
Polivka J, Janku F. Molecular targets for cancer therapy in the PI3K/AKT/mTOR pathway. Pharmacol Ther. 2014;142:164–75.
Engelman JA. Targeting PI3K signalling in cancer: opportunities, challenges and limitations. Nat Rev Cancer. 2009;9:550–62.
O’Reilly KE, Rojo F, She QB, Solit D, Mills GB, Smith D, et al. mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res. 2006;66:1500–8.
Chanthery YH, Gustafson WC, Itsara M, Persson A, Hackett CS, Grimmer M, et al. Paracrine signaling through MYCN enhances tumor-vascular interactions in neuroblastoma. Sci Transl Med. 2012;4:1153.
Westhoff MA, Faham N, Marx D, Nonnenmacher L, Jennewein C, Enzenmüller S, et al. Sequential dosing in chemosensitization: targeting the PI3K/Akt/mTOR pathway in neuroblastoma. PLoS ONE. 2013;8:e83128.
Serra V, Markman B, Scaltriti M, Eichhorn PJA, Valero V, Guzman M, et al. NVP-BEZ235, a dual PI3K/mTOR inhibitor, prevents PI3K signaling and inhibits the growth of cancer cells with activating PI3K mutations. Cancer Res. 2008;68:8022–30.
Stewart ZA, Westfall MD, Pietenpol JA. Cell-cycle dysregulation and anticancer therapy. Trends Pharmacol Sci. 2003;24:139–45.
Nunez R. DNA measurement and cell cycle analysis by flow cytometry. Curr Issues Mol Biol. 2001;3:67–70.
Masamha CP, Benbrook DM. Cyclin D1 degradation is sufficient to induce G1 cell cycle arrest despite constitutive expression of cyclin E2 in ovarian cancer cells. Cancer Res. 2009;69:6565–72.
Seo BR, Min KJ, Cho IJ, Kim SC, Kwon TK. Curcumin significantly enhances dual PI3K/Akt and mTOR inhibitor NVP-BEZ235-induced apoptosis in human renal carcinoma Caki cells through down-regulation of p53-dependent Bcl-2 expression and inhibition of Mcl-1 protein stability. PLoS ONE. 2014;9:e95588.
Mirza-Aghazadeh-Attari M, Ekrami EM, Aghdas SAM, Mihanfar A, Hallaj S, Yousefi B, et al. Targeting PI3K/Akt/mTOR signaling pathway by polyphenols: implication for cancer therapy. Life Sci. 2020;255:117481.
Sukumari-Ramesh S, Bentley JN, Laird MD, Singh N, Vender JR, Dhandapani KM. Dietary phytochemicals induce p53- and caspase-independent cell death in human neuroblastoma cells. Int J Dev Neurosci. 2011;29:701–10.
Kagawa S, Fujiwara T, Hizuta A, Yasuda T, Zhang W-W, Roth JA, et al. p53 expression overcomes p21 WAF1/CIP1-mediated G1 arrest and induces apoptosis in human cancer cells. Oncogene. 1997. https://doi.org/10.1038/sj.onc.1201362.
Acknowledgements
SH-SY5Y and HUVEC cell lines were a kind gift from Prof. F. Sahin at Yeditepe University. In vitro cell culture experiments were conducted in Department of Molecular Biology and Genetics, at Istanbul Sabahattin Zaim University.
Funding
This work was supported by the Scientific Research Project (BAP) of Istanbul Sabahattin Zaim University with the grant number BAP:1000-102.
Author information
Authors and Affiliations
Contributions
FC Conceptualization, methodology, investigation of in vitro cell culture studies and writing- original draft. SK Methodology, investigation of cell viability and cell cycle studies, and writing- original draft. HA Resources, supervised data analysis, reviewing and revising the manuscript. ZBB Design the study, guided the conduct of studies, funding acquisition, writing and revising the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Ethical approval
The authors confirm that no ethical approval is required in the study.
Consent to participate
Not applicable.
Consent for publication
All authors read and approved for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Çetin, F., Kosba, S., Abdik, H. et al. Synergistic anti-proliferative and apoptotic effect of NVP-BEZ235 and curcumin on human SH-SY5Y neuroblastoma cells. Med Oncol 41, 11 (2024). https://doi.org/10.1007/s12032-023-02239-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02239-8