Abstract
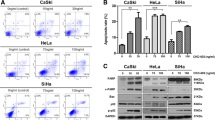
Combined chemotherapy is recommended strategy as a first-line treatment method in patients with cervical cancer. Ganetespib (STA-9090) is a second-generation heat shock protein 90 (Hsp90) inhibitor that blocks the ATPase function of Hsp90 and inhibits the proper folding of oncogenic client proteins. Venetoclax (ABT-199) is an orally bioavailable Bcl-2 (B-cell lymphoma 2) inhibitor that stimulates apoptotic signaling pathways in cancer cells. This study evaluated the anticancer effects of STA-9090 combined with Venetoclax in the human cervical cancer cell line (HeLa). The human cervical cancer cells were treated with STA-9090, Venetoclax, and Sta-9090 plus Venetoclax for 48 h, and cell viability was measured using the XTT assay. The alteration of the Hsp90 protein expression level and the chaperone activity of HSP90 were detected by ELISA and luciferase aggregation assay, respectively. For the apoptotic process, qRT-PCR was applied to study Bcl-2-associated X protein (Bax), B-cell lymphoma 2 (Bcl-2), Bcl-2-like protein 1 (Bcl-xL ), Cytochrome c (Cyt-c), Caspase3 (Cas-3), and Caspase7 (Cas-7) expression levels after drug treatments. Also, a colorimetric Cas-3 activity assay was performed to detect the induction of the apoptosis process. Our results demonstrated that 8 nM of STA-9090 combined with 4 µM of Venetoclax synergistically inhibited cervical cancer cell proliferation more than STA-9090 or Venetoclax alone after 48 h of treatment. STA-9090 and Venetoclax combination decreased the protein expression level of Hsp90 and significantly inhibited chaperone activity of Hsp90. This combination stimulated apoptosis in cervical cancer cells by down-regulating of anti-apoptotic markers while inducing pro-apoptotic markers. Also, the STA-9090-Venetoclax combination increased Cas-3 activity in Hela cells. Collectively, these findings pointed out that the STA-9090-Venetoclax combination exhibited more activity than the individual drugs to stimulate toxicity and apoptosis in cervical cancer cells based on HSP90 inhibition.






Similar content being viewed by others
Data availability
The data that support the findings of the current study are available from the corresponding author upon reasonable request.
References
Zhang S, Xu H, Zhang L, Qiao Y. Cervical cancer: Epidemiology, risk factors and screening. Chin J Cancer Res. 2020;32:720–8.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33.
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73:17–48.
Johnson CA, James D, Marzan A, Armaos M. Cervical Cancer: an overview of pathophysiology and management. Semin Oncol Nurs. 2019;35:166–74.
Bedell SL, Goldstein LS, Goldstein AR, Goldstein AT. Cervical Cancer screening: past, Present, and Future. Sex Med Rev. 2020;8:28–37.
Wipperman J, Neil T, Williams T. Cervical Cancer: evaluation and management. Am Fam Physician. 2018;97:449–54.
Pectasides D, Kamposioras K, Papaxoinis G, Pectasides E. Chemotherapy for recurrent cervical cancer. Cancer Treat Rev. 2008;4:603–13.
Eifel PJ. Concurrent chemotherapy and radiation therapy as the standard of care for cervical cancer. Nat Clin Pract Oncol. 2006;3:248–55.
Tzioras S, Pavlidis N, Paraskevaidis E, Ioannidis JP. Effects of different chemotherapy regimens on survival for advanced cervical cancer: systematic review and meta-analysis. Cancer Treat Rev. 2007;33:24–38.
Schopf FH, Biebl MM, Buchner J. The HSP90 chaperone machinery. Nat Rev Mol Cell Biol. 2017;18:345–60.
Gümus M, Ozgur A, Tutar L, Disli A, Koca I, Tutar Y, Design. Synthesis, and evaluation of heat shock protein 90 inhibitors in human breast Cancer and its metastasis. Curr Pharm Biotechnol. 2016;17:1231–45.
Özgür A, Tutar Y. Heat shock protein 90 inhibition in Cancer Drug Discovery: from Chemistry to Futural Clinical Applications. Anticancer Agents Med Chem. 2016;16:280–90.
Ren X, Li T, Zhang W, Yang X. Targeting heat-shock protein 90 in Cancer: an update on combination therapy. Cells. 2022;11:2556.
Costa TEMM, Raghavendra NM, Penido C. Natural heat shock protein 90 inhibitors in cancer and inflammation. Eur J Med Chem. 2020;189:112063.
Li L, Wang L, You QD, Xu XL. Heat shock protein 90 inhibitors: an Update on Achievements, Challenges, and future directions. J Med Chem. 2020;63(5):1798–822.
Koca İ, Gümüş M, Özgür A, Dişli A, Tutar Y. A Novel Approach to Inhibit Heat Shock Response as Anticancer Strategy by Coumarine Compounds Containing Thiazole Skeleton. Anticancer Agents Med Chem. 2015;15:916–30.
Karagöz GE, Rüdiger SG. Hsp90 interaction with clients. Trends Biochem Sci. 2015;40:117–25.
Sidera K, Patsavoudi E. HSP90 inhibitors: current development and potential in cancer therapy. Recent Pat Anticancer Drug Discov. 2014;9:1–20.
Neckers L, Workman P. Hsp90 molecular chaperone inhibitors: are we there yet? Clin Cancer Res. 2012;18(1):64–76.
Özgür A. Investigation of anticancer activities of STA-9090 (ganetespib) as a second generation HSP90 inhibitor in Saos-2 osteosarcoma cells. J Chemother. 2021;33:554–63.
Guan L, Zou Q, Liu Q, Lin Y, Chen S. HSP90 inhibitor Ganetespib (STA-9090) inhibits Tumor Growth in c-Myc-dependent esophageal squamous cell carcinoma. Onco Targets Ther. 2020;13:2997–3011.
Wang Y, Trepel JB, Neckers LM, Giaccone G. STA-9090, a small-molecule Hsp90 inhibitor for the potential treatment of cancer. Curr Opin Investig Drugs. 2010;11:1466–76.
Mihalyova J, Jelinek T, Growkova K, Hrdinka M, Simicek M, Hajek R. Venetoclax: a new wave in hematooncology. Exp Hematol. 2018;61:10–25.
Blair HA, Venetoclax. A review in previously untreated chronic lymphocytic leukaemia. Drugs. 2020;80:1973–80.
Juárez-Salcedo LM, Desai V, Dalia S. Venetoclax: evidence to date and clinical potential. Drugs Context. 2019;8:212574.
Lachowiez C, DiNardo CD, Konopleva M. Venetoclax in acute myeloid leukemia - current and future directions. Leuk Lymphoma. 2020;61:1313–22.
Huang RY, Pei L, Liu Q, Chen S, Dou H, Shu G, Yuan ZX, Lin J, Peng G, Zhang W, Fu H. Isobologram Analysis: a Comprehensive Review of Methodology and Current Research. Front Pharmacol. 2019;10:1222.
Özgür A, Tutar Y. Heat shock protein 90 inhibitors in oncology. Curr Proteom. 2014;11:2–16.
Jhaveri K, Taldone T, Modi S, Chiosis G. Advances in the clinical development of heat shock protein 90 (Hsp90) inhibitors in cancers. Biochim Biophys Acta. 2012;1823:742–55.
Xiao L, Lu X, Ruden DM. Effectiveness of hsp90 inhibitors as anti-cancer drugs. Mini Rev Med Chem. 2006;6:1137–43.
Subaiea G, Rizvi SMD, Yadav HKS, Al Hagbani T, Abdallah MH, Khafagy ES, Gangadharappa HV, Hussain T, Abu Lila AS. Ganetespib with Methotrexate Acts synergistically to impede NF-κB/p65 signaling in Human Lung Cancer A549 cells. Pharmaceuticals (Basel). 2023;16:230.
Lai CH, Park KS, Lee DH, Alberobello AT, Raffeld M, Pierobon M, Pin E, Petricoin Iii EF, Wang Y, Giaccone G. HSP-90 inhibitor ganetespib is synergistic with doxorubicin in small cell lung cancer. Oncogene. 2014;33:4867–76.
Proia DA, Sang J, He S, Smith DL, Sequeira M, Zhang C, Liu Y, Ye S, Zhou D, Blackman RK, Foley KP, Koya K, Wada Y. Synergistic activity of the Hsp90 inhibitor ganetespib with taxanes in non-small cell lung cancer models. Invest New Drugs. 2012;30:2201–9.
Solit DB, Rosen N. Hsp90: a novel target for cancer therapy. Curr Top Med Chem. 2006;6:1205–14.
Jego G, Hazoumé A, Seigneuric R, Garrido C. Targeting heat shock proteins in cancer. Cancer Lett. 2013;332:275–85.
Shimamura T, Perera SA, Foley KP, Sang J, Rodig SJ, Inoue T, Chen L, Li D, Carretero J, Li YC, Sinha P, Carey CD, Borgman CL, Jimenez JP, Meyerson M, Ying W, Barsoum J, Wong KK, Shapiro GI. Ganetespib (STA-9090), a nongeldanamycin HSP90 inhibitor, has potent antitumor activity in in vitro and in vivo models of non-small cell lung cancer. Clin Cancer Res. 2012;18:4973–85.
Kızıl HE, Caglayan C, Darendelioğlu E, Ayna A, Gür C, Kandemir FM, Küçükler S. Morin ameliorates methotrexate-induced hepatotoxicity via targeting Nrf2/HO-1 and Bax/Bcl2/Caspase-3 signaling pathways. Mol Biol Rep. 2023;50:3479–88.
Emre Kızıl H, Gür C, Ayna A, Darendelioğlu E, Küçükler S, Sağ S. Contribution of oxidative stress, apoptosis, endoplasmic reticulum stress and autophagy pathways to the Ameliorative Effects of Hesperidin in NaF-Induced Testicular toxicity. Chem Biodivers. 2023;20:e202200982.
Jan R, Chaudhry GE. Understanding apoptosis and apoptotic pathways targeted Cancer therapeutics. Adv Pharm Bull. 2019;9:205–18.
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35:495–516.
D’Arcy MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43:582–92.
Xu X, Lai Y, Hua ZC. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci Rep. 2019;39:BSR20180992.
Mori M, Hitora T, Nakamura O, Yamagami Y, Horie R, Nishimura H, Yamamoto T. Hsp90 inhibitor induces autophagy and apoptosis in osteosarcoma cells. Int J Oncol. 2015;46:47–54.
Özgür A, Kara A, Gökşen Tosun N, Tekin Ş, Gökçe İ. Debio-0932, a second generation oral Hsp90 inhibitor, induces apoptosis in MCF-7 and MDA-MB-231 cell lines. Mol Biol Rep. 2021;48:3439–49.
Wang X, Chen M, Zhou J, Zhang X. HSP27, 70 and 90, anti-apoptotic proteins, in clinical cancer therapy (review). Int J Oncol. 2014;45:18–30.
Li ZN, Luo Y. HSP90 inhibitors and cancer: prospects for use in targeted therapies (review). Oncol Rep. 2023;49:6.
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Dilay Karademir and Aykut Özgür. The first draft of the manuscript was written by Aykut Özgür and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
Consent for publication
All authors agree with the publication of this manuscript in this journal.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karademir, D., Özgür, A. The effects of STA-9090 (Ganetespib) and venetoclax (ABT-199) combination on apoptotic pathways in human cervical cancer cells. Med Oncol 40, 234 (2023). https://doi.org/10.1007/s12032-023-02107-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02107-5