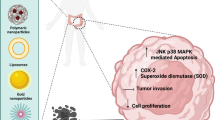
Abstract
Colorectal cancer (CRC) is the third broadly identified cancer in the world. The ineffectiveness of colorectal cancer treatment is redundantly reported. Natural bioactive compounds have gained popularity in reducing the drawback of conventional anti-cancer agents. Curcumin (Cur) and Artemisinin (Art) are materials of a natural source that have been utilized to treat numerous kinds of cancers. Although the benefits of bioactive materials, their utilization is limited because of poor solubility, bioavailability, and low dispersion rate in aqueous media. Nano delivery system such as niosome can improve the bioavailability and stability of bioactive compounds within the drug. In current work, we used Cur–Art co-loaded niosomal nanoparticles (Cur–Art NioNPs) as an anti-tumor factor versus colorectal cancer cell line. The synthesized formulations were characterized using dynamic light scattering, scanning electron microscopy, and FTIR. The proliferation ability of the cells and expression of apoptosis-associated gene were MTT assay and qRT-PCR, respectively. Cur–Art NioNPs exhibited well distributed with an encapsulation efficiency of 80.27% and 85.5% for Cur and Art. The NioNPs had good release and degradation properties, and had no negative effect on the survival and proliferation ability of SW480 cells. Importantly, nanoformulation form of Cur and Art significantly displayed higher toxicity effect against SW480 cells. Furthermore, Cur–Art NioNPs increased Bax, Fas, and p53 gene expressions and suppressed Bcl2, Rb, and Cyclin D 1 gene expressions. In summary, these results display the niosome NPs as a first report of nano-combinational application of the natural herbal substances with a one-step fabricated co-delivery system for effective colorectal cancer.






Similar content being viewed by others
Data availability
Not applicable.
References
Alagheband Y, et al. Design and fabrication of a dual-drug loaded nano-platform for synergistic anticancer and cytotoxicity effects on the expression of leptin in lung cancer treatment. J Drug Deliv Sci Technol. 2022;73:103389.
Eslami M, et al. Importance of probiotics in the prevention and treatment of colorectal cancer. J Cell Physiol. 2019;234(10):17127–43.
Eslami M, Peyghan AA. DNA nucleobase interaction with graphene like BC3 nano-sheet based on density functional theory calculations. Thin Solid Films. 2015;589:52–6.
Mogheri F, et al. Co-delivery of metformin and silibinin in dual-drug loaded nanoparticles synergistically improves chemotherapy in human non-small cell lung cancer A549 cells. J Drug Deliv Sci Technol. 2021;66:102752.
Pourgholi A, et al. Anticancer potential of silibinin loaded polymeric nanoparticles against breast cancer cells: insight into the apoptotic genes targets. Asian Pac J Cancer Prev: APJCP. 2021;22(8):2587.
Eslami M, et al. Importance of the microbiota inhibitory mechanism on the Warburg effect in colorectal cancer cells. J Gastrointest Cancer. 2020;51:738–47.
Putti TC, et al. Estrogen receptor-negative breast carcinomas: a review of morphology and immunophenotypical analysis. Mod Pathol. 2005;18(1):26–35.
Ataollahi M, et al. Breast cancer and associated factors: a review. J Med Life. 2015;8(4):6.
Zhou L, et al. NOTCH4 maintains quiescent mesenchymal-like breast cancer stem cells via transcriptionally activating SLUG and GAS1 in triple-negative breast cancer. Theranostics. 2020;10(5):2405.
Firouzai-Amandi A, et al. Development, characterization, and in vitro evaluation of cytotoxic activity of rutin loaded PCL-PEG nanoparticles against Skov3 ovarian cancer cell. Asian Pac J Cancer Prev. 2022;23(6):1951–7.
Eslami M, Moradi M, Moradi R. DFT investigation of hydrogen adsorption on the C3N nanotube. Vacuum. 2016;133:7–12.
Hassanpour A, et al. A DFT study on the electronic detection of mercaptopurine drug by boron carbide nanosheets. Comput Theor Chem. 2021;1198:113166.
Liu H, Lv L, Yang K. Chemotherapy targeting cancer stem cells. Am J Cancer Res. 2015;5(3):880.
Hosseini A, Ghorbani A. Cancer therapy with phytochemicals: evidence from clinical studies. Avicenna J Phytomed. 2015;5(2):84–97.
Greenwell M, Rahman P. Medicinal plants: their use in anticancer treatment. Int J Pharm Sci Res. 2015;6(10):4103–12.
Du G-J, et al. Epigallocatechin gallate (EGCG) is the most effective cancer chemopreventive polyphenol in green tea. Nutrients. 2012;4(11):1679–91.
Salmani Javan E, et al. Development of a magnetic nanostructure for co-delivery of metformin and silibinin on growth of lung cancer cells: possible action through leptin gene and its receptor regulation. Asian Pac J Cancer Prev. 2022;23(2):519–27.
Rasouli S, et al. Synergistic anticancer effects of electrospun nanofiber-mediated codelivery of curcumin and chrysin: possible application in prevention of breast cancer local recurrence. J Drug Deliv Sci Technol. 2020;55:101402.
Javan N, et al. Synergistic antiproliferative effects of co-nanoencapsulated curcumin and chrysin on mda-mb-231 breast cancer cells through upregulating mir-132 and mir-502c. Nutr Cancer. 2019;71(7):1201–13.
Azar LK, et al. Design and development of nanostructured co delivery of artemisinin and chrysin for targeting hTERT gene expression in breast cancer cell line: possible clinical application in cancer treatment. Asian Pac J Cancer Prev: APJCP. 2022;23(3):919.
Amirsaadat S, et al. Potential anti-proliferative effect of nano-formulated curcumin through modulating micro RNA-132, cyclin D1, and hTERT genes expression in breast cancer cell lines. J Cluster Sci. 2023. https://doi.org/10.1007/s10876-023-02404-z.
Mofarrah M, et al. Fabricating ZSM-5 zeolite/polycaprolactone nano-fibers co-loaded with dexamethasone and ascorbic acid: potential application in osteogenic differentiation of human adipose-derived stem cells. J Drug Deliv Sci Technol. 2023;79:103999.
Amirazad H, Dadashpour M, Zarghami N. Application of decellularized bone matrix as a bioscaffold in bone tissue engineering. J Biol Eng. 2022;16(1):1–18.
Dadashpour M, et al. Increased pro-apoptotic and anti-proliferative activities of simvastatin encapsulated PCL-PEG nanoparticles on human breast cancer adenocarcinoma cells. J Cluster Sci. 2023;34(1):211–22.
Bharali DJ, Mousa SA. Emerging nanomedicines for early cancer detection and improved treatment: current perspective and future promise. Pharmacol Ther. 2010;128(2):324–35.
Tarahomi M, et al. Niosomes nanoparticles as a novel approach in drug delivery enhances anticancer properties of chrysin in human ovarian carcinoma cells (SKOV3): an in vitro study. Med Oncol. 2023;40(3):87.
Ibrahim A, Khalil IA, El-Sherbiny IM. Development and evaluation of core-shell nanocarrier system for enhancing the cytotoxicity of doxorubicin/metformin combination against breast cancer cell line. J Pharm Sci. 2022;111(9):2581–91.
Lu M, et al. miR-338-3p regulates the proliferation, apoptosis and migration of SW480 cells by targeting MACC1. Exp Ther Med. 2019;17(4):2807–14.
Chang E, et al. Isolation of high-quality RNA from Platycladus orientalis and other Cupressaceae plants. Electron J Biotechnol. 2016;23:21–7.
Worldwide, M.I. (2011)Dynamic light scattering, common terms defined. Inform white paper. Malwern Instruments Limited. 2011: 1-6
Mudalige T, et al. Characterization of nanomaterials: tools and challenges. In: Nanomaterials for food applications. Elsevier; 2019. p. 313–53.
Manmode AS, Sakarkar DM, Mahajan NM. Nanoparticles-tremendous therapeutic potential: a review. Int J PharmTech Res. 2009;1(4):1020–7.
Hassani N, et al. The effect of dual bioactive compounds artemisinin and metformin co-loaded in PLGA-PEG nano-particles on breast cancer cell lines: potential apoptotic and anti-proliferative action. Appl Biochem Biotechnol. 2022;194(10):4930–45.
Lotfi-Attari J, et al. Co-delivery of curcumin and chrysin by polymeric nanoparticles inhibit synergistically growth and hTERT gene expression in human colorectal cancer cells. Nutr Cancer. 2017;69(8):1290–9.
Amoabediny G, et al. Overview of preparation methods of polymeric and lipid-based (niosome, solid lipid, liposome) nanoparticles: a comprehensive review. Int J Polym Mater Polym Biomater. 2018;67(6):383–400.
Ameeduzzafar, et al. Colloidal drug delivery system: amplify the ocular delivery. Drug Deliv. 2016;23(3):700–16.
Yasamineh S, et al. A state-of-the-art review on the recent advances of niosomes as a targeted drug delivery system. Int J Pharm. 2022;624:121878.
Jafari-Gharabaghlou D, et al. Potentiation of folate-functionalized PLGA-PEG nanoparticles loaded with metformin for the treatment of breast cancer: possible clinical application. Mol Biol Rep. 2023;50:1–11.
Adlravan E, et al. Potential activity of free and PLGA/PEG nanoencapsulated Nasturtium officinale extract in inducing cytotoxicity and apoptosis in human lung carcinoma A549 cells. J Drug Deliv Sci Technol. 2021;61:102256.
Momekova DB, Gugleva VE, Petrov PD. Nanoarchitectonics of multifunctional niosomes for advanced drug delivery. ACS Omega. 2021;6(49):33265–73.
Ge X, et al. Advances of non-ionic surfactant vesicles (niosomes) and their application in drug delivery. Pharmaceutics. 2019;11(2):55.
Abtahi NA, et al. Multifunctional stimuli-responsive niosomal nanoparticles for co-delivery and co-administration of gene and bioactive compound: In vitro and in vivo studies. Chem Eng J. 2022;429:132090.
Marques MS, et al. Nanotechnology for the treatment of paediatric diseases: a review. J Drug Deliv Sci Technol. 2022;75:103628.
Joy R, George J, John F. Brief outlook on polymeric nanoparticles, micelles, niosomes, hydrogels and liposomes: preparative methods and action. ChemistrySelect. 2022;7(6):e202104045.
Alemi A, et al. Paclitaxel and curcumin coadministration in novel cationic PEGylated niosomal formulations exhibit enhanced synergistic antitumor efficacy. J Nanobiotechnol. 2018;16:1–20.
El-Far SW, et al. Targeting colorectal cancer cells with niosomes systems loaded with two anticancer drugs models; comparative in vitro and anticancer studies. Pharmaceuticals. 2022;15(7):816.
Senapati D, et al. Promising approaches of small interfering RNAs (siRNAs) mediated cancer gene therapy. Gene. 2019;719:144071.
Nasr M. Development of an optimized hyaluronic acid-based lipidic nanoemulsion co-encapsulating two polyphenols for nose to brain delivery. Drug Deliv. 2016;23(4):1444–52.
Acknowledgements
Authors would like to thank the Department of Medical Biotechnology, Faculty of Medicine, Semnan University of Medical Sciences for financial supporting this project (Grant No: 2904).
Funding
There is no funding.
Author information
Authors and Affiliations
Contributions
AFA and EJ: Investigation, Methodology, Data curation, and Preparation of original draft. ME and MR: Reviewing and Editing; MD, YY, and BN: Supervision, Conceptualization, Funding acquisition, Reviewing, and Editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
The ethical approval for this paper was obtained from Semnan University of Medical Sciences (IR.SEMUMS.REC.1400.242).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Firouzi Amandi, A., Jokar, E., Eslami, M. et al. Enhanced anti-cancer effect of artemisinin- and curcumin-loaded niosomal nanoparticles against human colon cancer cells. Med Oncol 40, 170 (2023). https://doi.org/10.1007/s12032-023-02032-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02032-7