Abstract
Background
Oct4 has critical role in maintaining pluripotency, proliferative potential, and self-renewal capacity in embryonic stem and germ cells. Although Oct4 has been shown to be upregulated in many cancers, its clinical significance in gallbladder carcinoma is poorly understood.
Methods
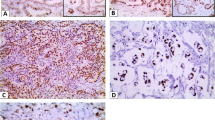
We studied the expression profile of Oct4 in 61 GBC and 30 chronic cholecystitis (as control) using real time RT-PCR, western blotting, and immunohistochemistry. The expression data was correlated with clinico-pathological parameters. The diagnostic utility was assessed through ROC curve, and prognostic value was analyzed by Kaplan–Meier method.
Results
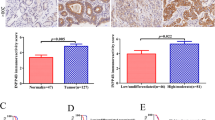
Oct4 was significantly upregulated at mRNA as well as protein levels. The higher mRNA expression shows significant association with late stage, late T stage, and higher grade of tumor. A significant positive correlation was also observed with stage, T stage, and tumor grade. Sum score analysis of protein expression shows positive correlation with stage and the presence or absence of gallstone in tumor samples. The ROC curve analysis revealed the moderate diagnostic potential of Oct4. Kaplan–Meier analysis showed that patients having higher expression of Oct4 were having low mean survival compared with the patients with lower Oct4 expression.
Conclusion
In conclusion, our data suggests that higher expression of Oct4 may serve as potential biological indicator for tumor aggressiveness and poor prognosis of GBC.






Similar content being viewed by others
Availability of Data and Materials
The materials and datasets analyzed in the present study are available from the first author on reasonable request.
References
Dutta U, Bush N, Kalsi D, Popli P, Kapoor VK. Epidemiology of gallbladder cancer in India. Chin Clin Oncol. 2019;8(4):33.
Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, et al. Cancer stem cells–perspectives on current status and future directions: AACR Workshop on cancer stem cells. Can Res. 2006;66(19):9339–44.
Sullivan JP, Minna JD, Shay JW. Evidence for self-renewing lung cancer stem cells and their implications in tumor initiation, progression, and targeted therapy. Cancer Metastasis Rev. 2010;29(1):61–72.
Scholer HR, Dressler GR, Balling R, Rohdewohld H, Gruss P. Oct-4: a germline-specific transcription factor mapping to the mouse t-complex. EMBO J. 1990;9(7):2185–95.
Niwa H, Miyazaki J, Smith AG. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat Genet. 2000;24(4):372–6.
Nichols J, Zevnik B, Anastassiadis K, Niwa H, Klewe-Nebenius D, Chambers I, et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 1998;95(3):379–91.
Singh SK, Clarke ID, Hide T, Dirks PB. Cancer stem cells in nervous system tumors. Oncogene. 2004;23(43):7267–73.
Yeom YI, Fuhrmann G, Ovitt CE, Brehm A, Ohbo K, Gross M, et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development. 1996;122(3):881–94.
Pan GJ, Chang ZY, Scholer HR, Pei D. Stem cell pluripotency and transcription factor Oct4. Cell Res. 2002;12(5–6):321–9.
Liu CG, Lu Y, Wang BB, Zhang YJ, Zhang RS, Lu Y, et al. Clinical implications of stem cell gene Oct-4 expression in breast cancer. Ann Surg. 2011;253(6):1165–71.
Chang CC, Shieh GS, Wu P, Lin CC, Shiau AL, Wu CL. Oct-3/4 expression reflects tumor progression and regulates motility of bladder cancer cells. Can Res. 2008;68(15):6281–91.
Monsef N, Soller M, Isaksson M, Abrahamsson PA, Panagopoulos I. The expression of pluripotency marker Oct 3/4 in prostate cancer and benign prostate hyperplasia. Prostate. 2009;69(9):909–16.
Chiou SH, Yu CC, Huang CY, Lin SC, Liu CJ, Tsai TH, et al. Positive correlations of Oct-4 and Nanog in oral cancer stem-like cells and high-grade oral squamous cell carcinoma. Clinical cancer research : an official journal of the American Association for Cancer Research. 2008;14(13):4085–95.
Quint K, Tonigold M, Di Fazio P, Montalbano R, Lingelbach S, Ruckert F, et al. Pancreatic cancer cells surviving gemcitabine treatment express markers of stem cell differentiation and epithelial-mesenchymal transition. Int J Oncol. 2012;41(6):2093–102.
Yin BB, Wu SJ, Zong HJ, Ma BJ, Cai D. Preliminary screening and identification of stem cell-like sphere clones in a gallbladder cancer cell line GBC-SD. J Zhejiang Univ Sci B. 2011;12(4):256–63.
Leonardo E, Valente G, Cappia S, Cianfriglia M, Arancia G, Meschini S, et al. Immunohistochemical evaluation of P-glycoprotein in human malignancies by monoclonal antibody MC57. Int J Cancer. 1994;57(6):841–6.
Okamoto K, Okazawa H, Okuda A, Sakai M, Muramatsu M, Hamada H. A novel octamer binding transcription factor is differentially expressed in mouse embryonic cells. Cell. 1990;60(3):461–72.
Gidekel S, Pizov G, Bergman Y, Pikarsky E. Oct-3/4 is a dose-dependent oncogenic fate determinant. Cancer Cell. 2003;4(5):361–70.
Yuan F, Zhou W, Zou C, Zhang Z, Hu H, Dai Z, et al. Expression of Oct4 in HCC and modulation to wnt/beta-catenin and TGF-beta signal pathways. Mol Cell Biochem. 2010;343(1–2):155–62.
Fatima N, Srivastava AN, Nigam J, Tandon N, Ahmad R, Kumar V. Clinicopathological correlation of cancer stem cell markers Oct-4 and CD133 expression as prognostic factor in malignant lesions of gallbladder: an immunohistochemical study. Indian J Pathol Microbiol. 2019;62(3):384–90 (Epub 2019/07/31).
Zou Q, Yang L, Yang Z, Huang J, Fu X. PSCA and Oct-4 expression in the benign and malignant lesions of gallbladder: implication for carcinogenesis, progression, and prognosis of gallbladder adenocarcinoma. Biomed Res Int. 2013;2013: 648420.
Zhang Y, Xu J, Xu Z, Wang Y, Wu S, Wu L, et al. Expression of vimentin and Oct-4 in gallbladder adenocarcinoma and their relationship with vasculogenic mimicry and their clinical significance. Int J Clin Exp Pathol. 2018;11(7):3618–27.
Santagata S, Ligon KL, Hornick JL. Embryonic stem cell transcription factor signatures in the diagnosis of primary and metastatic germ cell tumors. Am J Surg Pathol. 2007;31(6):836–45.
Zhang MX, Gan W, Jing CY, Zheng SS, Yi Y, Zhang J, et al. High expression of Oct4 and Nanog predict poor prognosis in intrahepatic cholangiocarcinoma patients after curative resection. J Cancer. 2019;10(5):1313–24.
Atlasi Y, Mowla SJ, Ziaee SA, Bahrami AR. OCT-4, an embryonic stem cell marker, is highly expressed in bladder cancer. Int J Cancer. 2007;120(7):1598–602.
Zhao FQ, Misra Y, Li DB, Wadsworth MP, Krag D, Weaver D, et al. Differential expression of Oct3/4 in human breast cancer and normal tissues. Int J Oncol. 2018;52(6):2069–78.
Yu B, Cai H, Xu Z, Xu T, Zou Q, Gu M. Expressions of stem cell transcription factors Nanog and Oct4 in renal cell carcinoma tissues and clinical significance. Artificial cells, nanomedicine, and biotechnology. 2016;44(8):1818–23.
Gwak JM, Kim M, Kim HJ, Jang MH, Park SY. Expression of embryonal stem cell transcription factors in breast cancer: Oct4 as an indicator for poor clinical outcome and tamoxifen resistance. Oncotarget. 2017;8(22):36305–18.
Hatefi N, Nouraee N, Parvin M, Ziaee SA, Mowla SJ. Evaluating the expression of oct4 as a prognostic tumor marker in bladder cancer. Iran J Basic Med Sci. 2012;15(6):1154–61.
Lu Y, Zhu H, Shan H, Lu J, Chang X, Li X, et al. Knockdown of Oct4 and Nanog expression inhibits the stemness of pancreatic cancer cells. Cancer Lett. 2013;340(1):113–23.
Monferrer E, Burgos-Panadero R, Blanquer-Maceiras M, Canete A, Navarro S, Noguera R. High Oct4 expression: implications in the pathogenesis of neuroblastic tumours. BMC Cancer. 2019;19(1):1.
Liu A, Cheng L, Du J, Peng Y, Allan RW, Wei L, et al. Diagnostic utility of novel stem cell markers SALL4, OCT4, NANOG, SOX2, UTF1, and TCL1 in primary mediastinal germ cell tumors. Am J Surg Pathol. 2010;34(5):697–706.
Dutta U. Gallbladder cancer: can newer insights improve the outcome? J Gastroenterol Hepatol. 2012;27(4):642–53.
Hyder O, Dodson RM, Sachs T, Weiss M, Mayo SC, Choti MA, et al. Impact of adjuvant external beam radiotherapy on survival in surgically resected gallbladder adenocarcinoma: a propensity score-matched Surveillance, Epidemiology, and End Results analysis. Surgery. 2014;155(1):85–93.
Villodre ES, Kipper FC, Pereira MB, Lenz G. Roles of OCT4 in tumorigenesis, cancer therapy resistance and prognosis. Cancer Treat Rev. 2016;51:1–9.
Shi CJ, Gao J, Wang M, Wang X, Tian R, Zhu F, et al. CD133(+) gallbladder carcinoma cells exhibit self-renewal ability and tumorigenicity. World J Gastroenterol. 2011;17(24):2965–71 (Epub 2011/07/08).
Koo BS, Lee SH, Kim JM, Huang S, Kim SH, Rho YS, et al. Oct4 is a critical regulator of stemness in head and neck squamous carcinoma cells. Oncogene. 2015;34(18):2317–24.
Wang XQ, Ongkeko WM, Chen L, Yang ZF, Lu P, Chen KK, et al. Octamer 4 (Oct4) mediates chemotherapeutic drug resistance in liver cancer cells through a potential Oct4-AKT-ATP-binding cassette G2 pathway. Hepatology. 2010;52(2):528–39.
Wen K, Fu Z, Wu X, Feng J, Chen W, Qian J. Oct-4 is required for an antiapoptotic behavior of chemoresistant colorectal cancer cells enriched for cancer stem cells: effects associated with STAT3/Survivin. Cancer Lett. 2013;333(1):56–65.
Golob-Schwarzl N, Wodlej C, Kleinegger F, Gogg-Kamerer M, Birkl-Toeglhofer AM, Petzold J, et al. Eukaryotic translation initiation factor 6 overexpression plays a major role in the translational control of gallbladder cancer. J Cancer Res Clin Oncol. 2019;145(11):2699–711 (Epub 2019/10/06).
Kessler SM, Lederer E, Laggai S, Golob-Schwarzl N, Hosseini K, Petzold J, et al. IMP2/IGF2BP2 expression, but not IMP1 and IMP3, predicts poor outcome in patients and high tumor growth rate in xenograft models of gallbladder cancer. Oncotarget. 2017;8(52):89736–45 (Epub 2017/11/23).
Acknowledgements
The authors acknowledge DST-PURSE, Banaras Hindu University; and UGC-UPE, Banaras Hindu University and IoE Faculty Incentive Grant to GN for financial assistance; Department of Science and Technology, Government of India for Junior and Senior research fellowships to DS; Department of Pathology, Institute of Medical Sciences; and Interdisciplinary School of Life Sciences and UGC-UPE, Banaras Hindu University for equipment facilities.
Author information
Authors and Affiliations
Contributions
GN and SS designed and supervised the study; DS carried out the experiments and did statistical analysis of the data; DB, MT, and MAA provided the tumor and normal biopsies along with the clinical information; AGK did the scoring of all IHC slides; manuscript preparation was done by DS and overall editing was done by GN.
Corresponding author
Ethics declarations
Ethical Approval and Consent
All procedures performed in studies involving human participants were in accordance with the 1964 Helsinki Declaration ethical standards and approved by the Institutional Ethical Committee of Institute of Science, Banaras Hindu University, Varanasi, India. The study was approved by the Institutional Ethical Committee of Institute of Science, Banaras Hindu University.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, D., Biswas, D., Tewari, M. et al. Clinical Significance of Overexpression of Oct4 in Advanced Stage Gallbladder Carcinoma. J Gastrointest Canc 54, 1231–1239 (2023). https://doi.org/10.1007/s12029-023-00913-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-023-00913-8