Abstract
Background
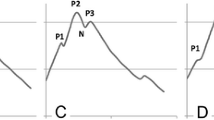
Intracranial pressure waveform morphology reflects compliance, which can be decreased by ventriculitis. We investigated whether morphologic analysis of intracranial pressure dynamics predicts the onset of ventriculitis.
Methods
Ventriculitis was defined as culture or Gram stain positive cerebrospinal fluid, warranting treatment. We developed a pipeline to automatically isolate segments of intracranial pressure waveforms from extraventricular catheters, extract dominant pulses, and obtain morphologically similar groupings. We used a previously validated clinician-supervised active learning paradigm to identify metaclusters of triphasic, single-peak, or artifactual peaks. Metacluster distributions were concatenated with temperature and routine blood laboratory values to create feature vectors. A L2-regularized logistic regression classifier was trained to distinguish patients with ventriculitis from matched controls, and the discriminative performance using area under receiver operating characteristic curve with bootstrapping cross-validation was reported.
Results
Fifty-eight patients were included for analysis. Twenty-seven patients with ventriculitis from two centers were identified. Thirty-one patients with catheters but without ventriculitis were selected as matched controls based on age, sex, and primary diagnosis. There were 1590 h of segmented data, including 396,130 dominant pulses in patients with ventriculitis and 557,435 pulses in patients without ventriculitis. There were significant differences in metacluster distribution comparing before culture-positivity versus during culture-positivity (p < 0.001) and after culture-positivity (p < 0.001). The classifier demonstrated good discrimination with median area under receiver operating characteristic 0.70 (interquartile range 0.55–0.80). There were 1.5 true alerts (ventriculitis detected) for every false alert.
Conclusions
Intracranial pressure waveform morphology analysis can classify ventriculitis without cerebrospinal fluid sampling.




Similar content being viewed by others
References
Rosenbaum BP, Vadera S, Kelly ML, Kshettry VR, Weil RJ. Ventriculostomy: frequency, length of stay and in-hospital mortality in the United States of America, 1988–2010. J Clin Neurosci. 2014;21(4):623–32.
Lewis A, Wahlster S, Karinja S, Czeisler BM, Kimberly WT, Lord AS. Ventriculostomy-related infections: the performance of different definitions for diagnosing infection. Br J Neurosurg. 2016;30(1):49–56.
Kirmani AR, Sarmast AH, Bhat AR. Role of external ventricular drainage in the management of intraventricular hemorrhage; its complications and management. Surg Neurol Int. 2015;6:188.
Lewis A, Irvine H, Ogilvy C, Kimberly WT. Predictors for delayed ventriculoperitoneal shunt placement after external ventricular drain removal in patients with subarachnoid hemorrhage. Br J Neurosurg. 2015;29(2):219–24.
Kuo LT, Lu HY, Tsai JC, Tu YK. Prediction of shunt dependency after intracerebral hemorrhage and intraventricular hemorrhage. Neurocrit Care. 2018;29(2):233–40.
Busl KM. Nosocomial infections in the neurointensive care unit. Neurol Clin. 2017;35(4):785–807.
Savin I, Ershova K, Kurdyumova N, et al. Healthcare-associated ventriculitis and meningitis in a neuro-ICU: incidence and risk factors selected by machine learning approach. J Crit Care. 2018;45:95–104.
Lozier AP, Sciacca RR, Romagnoli MF, Connolly ES Jr. Ventriculostomy-related infections: a critical review of the literature. Neurosurgery. 2008;62(Suppl 2):688–700.
Kitchen WJ, Singh N, Hulme S, Galea J, Patel HC, King AT. External ventricular drain infection: improved technique can reduce infection rates. Br J Neurosurg. 2011;25(5):632–5.
Lyke KE, Obasanjo OO, Williams MA, O’Brien M, Chotani R, Perl TM. Ventriculitis complicating use of intraventricular catheters in adult neurosurgical patients. Clin Infect Dis. 2001;33(12):2028–33.
Schade RP, Schinkel J, Roelandse FW, et al. Lack of value of routine analysis of cerebrospinal fluid for prediction and diagnosis of external drainage-related bacterial meningitis. J Neurosurg. 2006;104(1):101–8.
Sarnat HB. Ependymal reactions to injury. A review. J Neuropathol Exp Neurol. 1995;54(1):1–15.
Vardakis JC, Tully BJ, Ventikos Y. Multicompartmental poroelasticity as a platform for the integrative modeling of water transport in the brain. In: Computer models in biomechanics. Springer; 2013. p. 305–16.
Smillie A, Sobey I, Molnar Z. A hydro-elastic model of hydrocephalus. J Fluid Mech. 2004;539:417. https://doi.org/10.1017/S0022112005005707.
Bothwell SW, Janigro D, Patabendige A. Cerebrospinal fluid dynamics and intracranial pressure elevation in neurological diseases. Fluids Barriers CNS. 2019;16(1):9.
Megjhani M, Terilli K, Kaplan A, et al. Use of clustering to investigate changes in intracranial pressure waveform morphology in patients with ventriculitis. Acta Neurochir Suppl. 2021;131:59–62.
Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care—associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36(5):309–32.
Czeisler B, Choi HA, Guo K, et al. Comparison between institutionally-defined clinical criteria and CDC-criteria for the diagnosis of ventriculostomy-related infection (P02. 220). AAN Enterprises; 2012.
Strang G, Nguyen T. Wavelets and filter banks. SIAM; 1996.
Hu X, Xu P, Scalzo F, Vespa P, Bergsneider M. Morphological clustering and analysis of continuous intracranial pressure. IEEE Trans Biomed Eng. 2009;56(3):696–705.
Niennattrakul V, Ratanamahatana CA. On clustering multimedia time series data using k-means and dynamic time warping. IEEE; 2007. p. 733–8.
Keogh EJ, Pazzani MJ. An enhanced representation of time series which allows fast and accurate classification, clustering and relevance feedback. American Association for Artificial Intelligence Press; 1998. p. 239–41.
Megjhani M, Alkhachroum A, Terilli K, et al. An active learning framework for enhancing identification of non-artifactual intracranial pressure waveforms. Physiol Meas. 2019;40(1):015002.
Flierl MA, Rittirsch D, Weckbach S, et al. Disturbances of the hypothalamic-pituitary-adrenal axis and plasma electrolytes during experimental sepsis. Ann Intensive Care. 2011;1:53.
Csurka G, Bray C, Dance C, Fan L. Visual cetegoration with bags of keypoints. Workshop on statistical learning in computer vision. ECCV; 2004. p. 1–22.
Sivic J, Zisserman A. Video Google: a text retrieval approach to object matching in videos. Computer Vision, IEEE International Conference on 2003;3:1470.
Hanley JA, McNeil BJ. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology. 1983;148(3):839–43.
Pedregosa F, Varoquaux G, Gramfort A, et al. Scikit-learn: machine learning in Python. J Mach Learn Res. 2011;12:2825–30.
Bodilsen J, Dalager-Pedersen M, Schonheyder HC, Nielsen H. Time to antibiotic therapy and outcome in bacterial meningitis: a Danish population-based cohort study. BMC Infect Dis. 2016;16:392.
Koster-Rasmussen R, Korshin A, Meyer CN. Antibiotic treatment delay and outcome in acute bacterial meningitis. J Infect. 2008;57(6):449–54.
Proulx N, Frechette D, Toye B, Chan J, Kravcik S. Delays in the administration of antibiotics are associated with mortality from adult acute bacterial meningitis. QJM. 2005;98(4):291–8.
Lepur D, Barsic B. Community-acquired bacterial meningitis in adults: antibiotic timing in disease course and outcome. Infection. 2007;35(4):225–31.
Auburtin M, Wolff M, Charpentier J, et al. Detrimental role of delayed antibiotic administration and penicillin-nonsusceptible strains in adult intensive care unit patients with pneumococcal meningitis: the PNEUMOREA prospective multicenter study. Crit Care Med. 2006;34(11):2758–65.
Aronin SI, Peduzzi P, Quagliarello VJ. Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med. 1998;129(11):862–9.
Funding
This study was funded by the National Institutes of Health (Grant Number: R21NS113055 [SP]) and American Heart Association (Grant Number 20POST35210653 [MM]).
Author information
Authors and Affiliations
Contributions
Data collection (KT, LK, JC, JC, SM, NB, PH, DJR, SA, JC, ESC, and NM), Analysis (MM, SP, and KT), Writing (MM, SP, and KT), Editing (All). All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
NM reports funding from Accelerated Translational Incubator Pilot Grant through the University of Maryland Baltimore Institute of Clinical and Translational Research unrelated to this study.
Ethical Approval/Informed Consent
The study was approved by the institutional review boards at the respective centers.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Megjhani, M., Terilli, K., Kalasapudi, L. et al. Dynamic Intracranial Pressure Waveform Morphology Predicts Ventriculitis. Neurocrit Care 36, 404–411 (2022). https://doi.org/10.1007/s12028-021-01303-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-021-01303-3