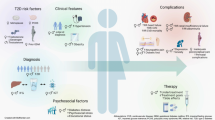
Abstract
The prevalence of adult obesity has risen markedly in recent decades. The endocrine system precisely regulates energy balance, fat abundance and fat deposition. Interestingly, white adipose tissue is an endocrine gland producing adipokines, which regulate whole-body physiology, including energy balance and reproduction. Endocrine disruptor chemicals (EDCs) include natural substances or chemicals that affect the endocrine system by multiple mechanisms and increase the risk of adverse health outcomes. Numerous studies have associated exposure to EDCs with obesity, classifying them as obesogens by their ability to activate different mechanisms, including the differentiation of adipocytes, increasing the storage of triglycerides, or elevating the number of adipocytes. Moreover, in recent years, not only industrial deception and obesity have intensified but also the problem of human infertility. Reproductive functions depend on hormone interactions, the balance of which may be disrupted by various EDCs or obesity. This review gives a brief summary of common EDCs linked with obesity, the mechanisms of their action, and the effect on adipokine levels, reproduction and connected disorders, such as polycystic ovarian syndrome, decrease in sperm motility, preeclampsia, intrauterine growth restriction in females and decrease of sperm motility in males.






Similar content being viewed by others
References
J.J. Heindel, B. Blumberg, M. Cave, R. Machtinger, A. Mantovani, M.A. Mendez, A. Nadal, P. Palanza, G. Panzica, R. Sargis, L.N. Vandenberg, F. Vom Saal, Metabolism disrupting chemicals and metabolic disorders. Reprod. Toxicol. 68, 3–33 (2017). https://doi.org/10.1016/j.reprotox.2016.10.001
A. Estienne, A. Bongrani, M. Reverchon, C. Ramé, P.H. Ducluzeau, P. Froment, J. Dupont, Involvement of novel adipokines, chemerin, visfatin, resistin and apelin in reproductive functions in normal and pathological conditions in humans and animal models. Int. J. Mol. Sci. 20(18), 4431 (2019). https://doi.org/10.3390/ijms20184431
J. Küblbeck, T. Vuorio, J. Niskanen, V. Fortino, A. Braeuning, K. Abass, A. Rautio, J. Hakkola, P. Honkakoski, A.L. Levonen, The EDCMET project: metabolic effects of endocrine disruptors. Int. J. Mol. Sci. 21(8), 3021 (2020). https://doi.org/10.3390/ijms21083021
C.R. Giviziez, E.G. Sanchez, M.S. Approbato, M.C. Maia, E.A. Fleury, R.S. Sasaki, Obesity and anovulatory infertility: a review. JBRA Assist. Reprod. 20(4), 240–245 (2016). https://doi.org/10.5935/1518-0557.20160046
P.D. Darbre, Endocrine disruptors and obesity. Curr. Obes. Rep. 6(1), 18–27 (2017). https://doi.org/10.1007/s13679-017-0240-4
F. Rancière, J. Botton, R. Slama, M.Z. Lacroix, L. Debrauwer, M.A. Charles, R. Roussel, B. Balkau, D.J. Maggliano, Exposure to bisphenol A and bisphenol S and incident type 2 diabetes: a case–cohort study in the French cohort DESIR. Environ. Health Perspect. 127, 107013 (2019). https://doi.org/10.1289/EHP5159
S.J. Blunden, R. Hill, Bis (tributyltin) oxide as a wood preservative: Its chemical nature in timber. Appl. Organomet. Chem. 2, 251–256 (1988). https://doi.org/10.1002/aoc.590020308
S. Takahashi, H. Mukai, S. Tanabe, K. Sakayama, T. Miyazaki, H. Masuno, Butyltin residues in livers of humans and wild terrestrial mammals and in plastic products. Environment 106, 213–218 (1999). https://doi.org/10.1016/S0269-7491(99)00068-8
L.E. Overgaard, K.M. Main, H. Frederiksen, S. Stender, P.B. Szecsi, H.C. Williams, J.P. Thyssen, Children with atopic dermatitis and frequent emollient use have increased urinary levels of low‐molecular‐weight phthalate metabolites and parabens. Allergy 72, 1768–1777 (2017). https://doi.org/10.1111/all.13157
J.P. Mixner, J. Meites, C.W. Turner, The stimulation and inhibition of milk secretion in goats with diethylstilbestrol. J. Dairy Sci. 27, 957–964 (1944). https://doi.org/10.3168/jds.S0022-0302(44)92670-6
A. Möller, Z. Xie, M. Cai, G. Zhong, P. Huang, M. Cai, R. Strum, J. He, R. Ebinghaus, Polybrominated diphenyl ethers vs alternate brominated flame retardants and dechloranes from East Asia to the Arctic. Environ. Sci. Technol. 45, 6793–6799 (2011). https://doi.org/10.1021/es201850n
M.A. Siddiqi, R.H. Laessig, K.D. Reed, Polybrominated diphenyl ethers (PBDEs): new pollutants–old diseases. Clin. Med. Res. 1, 281–290 (2003). https://doi.org/10.3121/cmr.1.4.281
M.K. Woźniak, E. Jaszczak, M. Wiergowski, Ż. Polkowska, J. Namieśnik, M. Biziuk, Meconium analysis as a promising diagnostic tool for monitoring fetal exposure to toxic substances: recent trends and perspectives. Trends Anal. Chem. 109, 124–141 (2018). https://doi.org/10.1016/j.trac.2018.09.02
A. Martinez, A.J. Al-Ahmad, Effects of glyphosate and aminomethylphosphonic acid on an isogeneic model of the human blood-brain barrier. Toxicol. Lett. 304, 39–49 (2019). https://doi.org/10.1016/j.toxlet.2018.12.013
F.D. Dudimah, S.O. Odman‐Ghazi, F. Hatcher, M.M. Whalen, Effect of tributyltin (TBT) on ATP levels in human natural killer (NK) cells: Relationship to TBT‐induced decreases in NK function. J. Appl. Toxicol. 27, 86–94 (2007). https://doi.org/10.1002/jat.1202
Z. Singh, J. Kaur, R. Kaur, S.S. Hundal, Toxic effects of organochlorine pesticides: a review. Am. J. BioSci. 4, 11 (2016). https://doi.org/10.11648/j.ajbio.s.2016040301.13
S. Richard, S. Moslemi, H. Sipahutar, N. Benachour, G.E. Seralini, Differential effects of glyphosate and roundup on human placental cells and aromatase. Environment 113, 716–720 (2005). https://doi.org/10.1289/ehp.7728
J.R. Palmer, L.A. Wise, E.E. Hatch, R. Troisi, L. Titus-Ernstoff, W. Strohsnitter, R. Kaufman, A.L. Herbst, M.H. Noller, R.N. Hoover, Prenatal diethylstilbestrol exposure and risk of breast cancer. Cancer Epidemiol 15, 1509–1514 (2006). https://doi.org/10.1158/1055-9965
G. Paolella, A.M. Romanelli, S. Martucciello, S. Sposito, M. Lepretti, C. Esposito, A. Capaldo, I. Caputo, The mechanism of cytotoxicity of 4‐nonylphenol in a human hepatic cell line involves ER‐stress, apoptosis, and mitochondrial dysfunction. J. Biochem Mol. Toxicol. 35, 22780 (2021). https://doi.org/10.1002/jbt.22780
S.S. Yalçın, E. Örün, S. Yalçın, O. Aykut, Organochlorine pesticide residues in breast milk and maternal psychopathologies and infant growth from suburban area of Ankara, Turkey. Int. J. Environ. 25, 364–372 (2015). https://doi.org/10.1080/09603123.2014.945515
Z. Zuo, S. Chen, T. Wu, J. Zhang, Y. Su, Y. Chen, C. Wang, Tributyltin causes obesity and hepatic steatosis in male mice. Environ. Toxicol. 26, 79–85 (2011). https://doi.org/10.1002/tox.20531
V. Delfosse, M. Grimaldi, J.L. Pons, A. Boulahtouf, A. le Maire, V. Cavailles, G. Labesse, W. Bourguet, P. Balaguer, Structural and mechanistic insights into bisphenols action provide guidelines for risk assessment and discovery of bisphenol A substitutes. Proc. Natl Acad. Sci. USA 109(37), 14930–14935 (2012). https://doi.org/10.1073/pnas.1203574109
M. Hoffmann, J. Gogola, M. Kotula-Balak, A. Ptak, Stimulation of ovarian cell proliferation by tetrabromobisphenol A but not tetrachlorobisphenol A through G protein-coupled receptor 30. Toxicol. Vitr. 45(1), 54–59 (2017). https://doi.org/10.1016/j.tiv.2017.08.009
J. Gogola, M. Hoffmann, A. Ptak, Persistent endocrine-disrupting chemicals found in human follicular fluid stimulate the proliferation of granulosa tumor spheroids via GPR30 and IGF1R but not via the classic estrogen receptors. Chemosphere 217, 100–110 (2019). https://doi.org/10.1016/j.chemosphere.2018.11.018
S. Ahmed, E. Valen, A. Sandelin, J. Matthews, Dioxin increases the interaction between aryl hydrocarbon receptor and estrogen receptor alpha at human promoters. Toxicol. Sci. 111(2), 254–266 (2009). https://doi.org/10.1093/toxsci/kfp144
J.P. Whitlock Jr, Induction of cytochrome P4501A1. Annu. Rev. Pharm. Toxicol. 39, 103–125 (1999). https://doi.org/10.1146/annurev.pharmtox.39.1.103
H. Inadera, A. Shimomura, Environmental chemical tributyltin augments adipocyte differentiation. Toxicol. Lett. 159(3), 226–234 (2005). https://doi.org/10.1016/j.toxlet.2005.05.015
P. Hu, X. Chen, R.J. Whitener, E.T. Boder, J.O. Jones, A. Porollo, J. Chen, L. Zhao, Effects of parabens on adipocyte differentiation. Toxicol. Sci. 1, 56–70 (2013). https://doi.org/10.1093/toxsci/kfs262
B. Desvergne, W. Wahli, Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr. Rev. 20(5), 649–688 (1999). https://doi.org/10.1210/edrv.20.5.0380
J. Wu, J. Oka, I. Tabata, M. Higuchi, T. Toda, N. Fuku, J. Ezaki, F. Sugiyama, S. Uchiyama, K. Yamada, Y. Ishimi, Effects of isoflavone and exercise on BMD and fat mass in postmenopausal Japanese women: a 1-year randomized placebo-controlled trial. J. Bone Min. Res. 21(5), 780–789 (2006). https://doi.org/10.1359/jbmr.060208
M. Fernández, M. Bianchi, V. Lux-Lantos, C. Libertun, Neonatal exposure to bisphenol a alters reproductive parameters and gonadotropin releasing hormone signaling in female rats. Environ. Health Perspect. 117, 757–762 (2009). https://doi.org/10.1289/ehp.0800267
H. Ullah, F. Ullah, O. Rehman, S. Jahan, T. Afsar, D. Al-Disi, A. Almajwal, S. Razak, Chronic exposure of bisphenol S (BPS) affect hypothalamic-pituitary-testicular activities in adult male rats: possible in estrogenic mode of action. Environ. Health Prev. Med. 26(1), 31 (2021). https://doi.org/10.1186/s12199-021-00954-0
E.R. Kadir, A. Imam, O.J. Olajide, M.S. Ajao, Alterations of Kiss 1 receptor, GnRH receptor and nuclear receptors of the hypothalamo-pituitary-ovarian axis following low dose bisphenol-A exposure in Wistar rats. Anat. Cell Biol. 54(2), 212–224 (2021). https://doi.org/10.5115/acb.20.215
C. Tang, J. Zhang, P. Liu, Y. Zhou, Q. Hu, Y. Zhong, X. Wang, L. Chen, Chronic exposure to low dose of bisphenol A causes follicular atresia by inhibiting kisspeptin neurons in anteroventral periventricular nucleus in female mice. Neurotoxicology 79, 164–176 (2020). https://doi.org/10.1016/j.neuro.2020.04.011
J. Zhang, P. Sun, F. Yang, T. Kong, R. Zhang, Tributyltin disrupts feeding and energy metabolism in the goldfish (Carassius auratus). Chemosphere 152, 221–228 (2016). https://doi.org/10.1016/j.chemosphere.2016.02.127
H. Fu, F. Gao, X. Wang, P. Tan, S. Qiu, B. Shi, A. Shan, Effects of glyphosate-based herbicide-contaminated diets on reproductive organ toxicity and hypothalamic-pituitary-ovarian axis hormones in weaned piglets. Environ. Pollut. 272, 115596 (2021). https://doi.org/10.1016/j.envpol.2020.115596
X. Ren, R. Li, J. Liu, K. Huang, S. Wu, Y. Li, C. Li, Effects of glyphosate on the ovarian function of pregnant mice, the secretion of hormones and the sex ratio of their fetuses. Environ. Pollut. 243, 833–841 (2018). https://doi.org/10.1016/j.envpol.2018.09.049
J. Jurewicz, M. Radwan, B. Wielgomas, A. Karwacka, A. Klimowska, P. Kałużny, P. Radwan, W. Hanke, Parameters of ovarian reserve in relation to urinary concentrations of parabens. Environ. Health 19, 26 (2020). https://doi.org/10.1186/s12940-020-00580-3
J. Peretz, J.A. Flaws, Bisphenol A down-regulates rate-limiting Cyp11a1 to acutely inhibit steroidogenesis in cultured mouse antral follicles. Toxicol. Appl Pharm. 271, 249–256 (2013). https://doi.org/10.1016/j.taap.2013.04.028
J. Shi, C. Liu, M. Chen, J. Yan, C. Wang, Z. Zuo, C. He, The interference effects of bisphenol A on the synthesis of steroid hormones in human ovarian granulosa cells. Environ. Toxicol. 36(4), 665–674 (2021). https://doi.org/10.1002/tox.23070
M. Lin, R. Hua, J. Ma, Y. Zhou, P. Li, X. Xu, Z. Yu, S. Quan, Bisphenol A promotes autophagy in ovarian granulosa cells by inducing AMPK/mTOR/ULK1 signalling pathway. Environ. Int. 147, 106298 (2021). https://doi.org/10.1016/j.envint.2020.106298
H. Lee, S. Lim, S. Yun, A. Yoon, G. Park, H. Yang, Tributyltin increases the expression of apoptosis- and adipogenesis-related genes in rat ovaries. Clin. Exp. Reprod. Med. 39, 15–21 (2012). https://doi.org/10.5653/cerm.2012.39.1.15
M. Saitoh, T. Yanase, H. Morinaga, M. Tanabe, Y.M. Mu, Y. Nishi, M. Nomura, T. Okabe, K. Goto, R. Takayanagi, H. Nawata, Tributyltin or triphenyltin inhibits aromatase activity in the human granulosa-like tumor ce ll line KGN. Biochem. Biophys. Res. Commun. 289, 198–204 (2001). https://doi.org/10.1006/bbrc.2001.5952
F. Zhang, P. Yang, L. Qin, J. Zhang, Adverse stimulation of 4-nonylphenol in abnormal reproductive organs of female chickens. Oncotarget 8(66), 110029–110038 (2017). https://doi.org/10.18632/oncotarget.21858
Y.L. Low, A.M. Dunning, M. Dowsett, E. Folkerd, D. Doody, J. Taylor, A. Bhaniani, R. Luben, K.T. Khaw, N.J. Wareham, S.A. Bingham, Phytoestrogen exposure is associated with circulating sex hormone levels in postmenopausal women and interact with ESR1 and NR1I2 gene variants. Cancer Epidemiol. Biomark. Prev. 16, 1009–1016 (2007). https://doi.org/10.1158/1055-9965.EPI-06-0899
M. Yamamoto, M. Shirai, K. Sugita, N. Nagai, Y. Miura, R. Mogi, K. Yamamoto, A. Tamura, K. Arishima, Effects of maternal exposure to diethylstilbestrol on the development of the reproductive system and thyroid function in male and female rat offspring. J. Toxicol. Sci. 28, 385–394 (2003). https://doi.org/10.2131/jts.28.385
A. Karpeta, A. Rak-Mardyła, J. Jerzak, E.L. Gregoraszczuk, Congener-specific action of PBDEs on steroid secretion, CYP17, 17β-HSD and CYP19 activity and protein expression in porcine ovarian follicles. Toxicol. Lett. 206, 258–263 (2011). https://doi.org/10.1016/j.toxlet.2011.08.005
K.S. Kechagias, A. Semertzidou, A. Athanasiou, M. Paraskevaidi, M. Kyrgiou, Bisphenol-A and polycystic ovary syndrome: a review of the literature. Rev. Environ. Health 35(4), 323–331 (2020). https://doi.org/10.1515/reveh-2020-0032
L. Akın, M. Kendirci, F. Narin, S. Kurtoğlu, N. Hatipoğlu, F. Elmalı, Endocrine disruptors and polycystic ovary syndrome: phthalates. J. Clin. Res. Pediatr. Endocrinol. 12(4), 393–400 (2020). https://doi.org/10.4274/jcrpe.galenos.2020.2020.0037
X.Y. Shi, Z. Wang, L. Liu, L.M. Feng, N. Li, S. Liu, H. Gao, Low concentrations of bisphenol A promote human ovarian cancer cell proliferation and glycolysis-based metabolism through the estrogen receptor-α pathway. Chemosphere 185, 361–367 (2017). https://doi.org/10.1016/j.chemosphere.2017.07.027
M. Huang, X. Li, S. Jia, S. Liu, L. Fu, X. Jiang, M. Yang, Bisphenol AF induces apoptosis via estrogen receptor beta (ERβ) and ROS-ASK1-JNK MAPK pathway in human granulosa cell line KGN. Environ. Pollut. 270, 116051 (2021). https://doi.org/10.1016/j.envpol.2020.116051
C. Pivonello, G. Muscogiuri, A. Nardone, F. Garifalos, D.P. Provvisiero, N. Verde, C. de Angelis, A. Conforti, M. Piscopo, R.S. Auriemma, A. Colao, R. Pivonello, A. Bisphenol, an emerging threat to female fertility. Reprod. Biol. Endocrinol. 18, 22 (2020). https://doi.org/10.1186/s12958-019-0558-8
O. Kishta, A. Adeeko, D. Li, T. Luu, J.R. Brawer, C. Morales, L. Hermo, B. Robaire, B.F. Hales, J. Barthelemy, D.G. Cyr, J.M. Trasler, In utero exposure to tributyltin chloride differentially alters male and female fetal gonad morphology and gene expression profiles in the Sprague-Dawley rat. Reprod. Toxicol. 23, 1–11 (2007). https://doi.org/10.1016/j.reprotox.2006.08.014
V. Rouiller-Fabre, R. Habert, G. Livera, Effects of endocrine disruptors on the human fetal testis. Ann. Endocrinol. 75, 54–57 (2014). https://doi.org/10.1016/j.ando.2014.03.010
Y.B. Wetherill, B.T. Akingbemi, J. Kanno, J.A. McLachlan, A. Nadal, C. Sonnenschein, C.S. Watson, R.T. Zoeller, S.M. Belcher, In vitro molecular mechanisms of bisphenol A action. Reprod. Toxicol. 24, 178–198 (2007). https://doi.org/10.1016/j.reprotox.2007.05.010
Y. Gong, J. Wu, Y. Huang, S. Shen, X. Han, Nonylphenol induces apoptosis in rat testicular Sertoli cells via endoplasmic reticulum stress. Toxicol. Lett. 186, 84–95 (2009). https://doi.org/10.1016/j.toxlet.2009.01.010
C. McKinnell, N. Atanassova, K. Williams, J.S. Fisher, M. Walker, K.J. Turner, T.K. Saunders, R.M. Sharpe, Suppression of androgen action and the induction of gross abnormalities of the reproductive tract in male rats treated neonatally with diethylstilbestrol. J. Androl. 22, 323–338 (2001)
G. Yuan, Y. Liu, G. Liu, L. Wei, Y. Wen, S. Huang, Y. Guo, F. Zou, J. Cheng, Associations between semen phytoestrogens concentrations and semen quality in Chinese men. Environ. Int 129, 136–144 (2019). https://doi.org/10.1016/j.envint.2019.04.076
R.S. Tavares, F.C. Martins, P.J. Oliveira, J. Ramalho-Santos, F.P. Peixoto, Parabens in male infertility-is there a mitochondrial connection? Reprod. Toxicol. 27, 1–7 (2009). https://doi.org/10.1016/j.reprotox.2008.10.002
M. Astiz, G.E. Hurtado de Catalfo, M.N. García, S.M. Galletti, A.L. Errecalde, M.J. de Alaniz, C.A. Marra, Pesticide-induced decrease in rat testicular steroidogenesis is differentially prevented by lipoate and tocopherol. Ecotoxicol. Environ. Saf. 91, 129–138 (2013). https://doi.org/10.1016/j.ecoenv.2013.01.022
D.K. Li, Z. Zhou, M. Miao, Y. He, J. Wang, J. Ferber, L.J. Herrinton, E. Gao, W. Yuan, Urine bisphenol-A (BPA) level in relation to semen quality. Fertil. Steril. 95, 625–630 (2011). https://doi.org/10.1016/j.fertnstert.2010.09.026
N. Abdelouahab, Y. Ainmelk, L. Takser, Polybrominated diphenyl ethers and sperm quality. Reprod. Toxicol. 31, 546–550 (2011). https://doi.org/10.1016/j.reprotox.2011.02.005
V.B. Mutwedu, A.W. Nyongesa, P.C. Azine, D.K. Chiregereza, V.H. Ngoumtsop, Y. Mugumaarhahama, R. Ayagirwe, Growth performance and reproductive function impairment of glyphosate-based herbicide in male guinea pig (Cavia porcellus). Vet. Med. Sci. 7(3), 1047–1055 (2021). https://doi.org/10.1002/vms3.443
S. Tait, R. Tassinari, F. Maranghi, A. Mantovani, Bisphenol A affects placental layers morphology and angiogenesis during early pregnancy phase in mice. J. Appl. Toxicol. 35(11), 1278–1291 (2015). https://doi.org/10.1002/jat.3176
M.A. Elmetwally, A.A. Halawa, Y.Y. Lenis, W. Tang, G. Wu, F.W. Bazer, Effects of BPA on expression of apoptotic genes and migration of ovine trophectoderm (oTr1) cells during the peri-implantation period of pregnancy. Reprod. Toxicol. 83, 73–79 (2019). https://doi.org/10.1016/j.reprotox.2018.12.001
M. Ponniah, E.E. Billett, L.A. De Girolamo, Bisphenol A increases BeWo trophoblast survival in stress-induced paradigms through regulation of oxidative stress and apoptosis. Chem. Res. Toxicol. 28(9), 1693–1703 (2015). https://doi.org/10.1021/acs.chemrestox.5b00093
P. Wei, D. Ru, X. Li, D. Shi, M. Zhang, Q. Xu, H. Zhou, S. Wen, Exposure to environmental bisphenol A inhibits HTR-8/SVneo cell migration and invasion. J. Biomed. Res. 34(5), 369–378 (2020). https://doi.org/10.7555/JBR.34.20200013
J. Troisi, C. Mikelson, S. Richards, S. Symes, D. Adair, F. Zullo, M. Guida, Placental concentrations of bisphenol A and birth weight from births in the Southeastern U.S. Placenta 35(11), 947–952 (2014). https://doi.org/10.1016/j.placenta.2014.08.091
F. Leclerc, M.F. Dubois, A. Aris, Maternal, placental and fetal exposure to bisphenol A in women with and without preeclampsia. Hypertens. Preg. 33(3), 341–348 (2014). https://doi.org/10.3109/10641955.2014.892607
W. Huo, W. Xia, Y. Wan, B. Zhang, A. Zhou, Y. Zhang, K. Huang, Y. Zhu, C. Wu, Y. Peng, M. Jiang, J. Hu, H. Chang, B. Xu, Y. Li, S. Xu, Maternal urinary bisphenol A levels and infant low birth weight: a nested case-control study of the Health Baby Cohort in China. Environ. Int. 85, 96–103 (2015). https://doi.org/10.1016/j.envint.2015.09.005
Y.H. Chiu, L. Mínguez-Alarcón, J.B. Ford, M. Keller, E.W. Seely, C. Messerlian, J. Petrozza, P.L. Williams, X. Ye, A.M. Calafat, R. Hauser, T. James-Todd, Trimester-specific urinary bisphenol A concentrations and blood glucose levels among pregnant women from a fertility clinic. J. Clin. Endocrinol. Metab. 102(4), 1350–1357 (2017). https://doi.org/10.1210/jc.2017-00022
V. Pergialiotis, P. Kotrogianni, E. Christopoulos-Timogiannakis, D. Koutaki, G. Daskalakis, N. Papantoniou, Bisphenol A and adverse pregnancy outcomes: a systematic review of the literature. J. Matern. Fetal Neonatal Med. 31(24), 3320–3327 (2018). https://doi.org/10.1080/14767058.2017.1368076
Z. Yu, Y. Han, R. Shen, K. Huang, Y.Y. Xu, Q.N. Wang, S.S. Zhou, D.X. Xu, F.B. Tao, Gestational di-(2-ethylhexyl) phthalate exposure causes fetal intrauterine growth restriction through disturbing placental thyroid hormone receptor signaling. Toxicol. Lett. 294, 1–10 (2018). https://doi.org/10.1016/j.toxlet.2018.05.013
S. Kim, Y.H. Cho, I. Lee, W. Kim, S. Won, J.L. Ku, H.B. Moon, J. Park, S. Kim, G. Choi, K. Choi, Prenatal exposure to persistent organic pollutants and methylation of LINE-1 and imprinted genes in placenta: a CHECK cohort study. Environ. Int. 119, 398–406 (2018). https://doi.org/10.1016/j.envint.2018.06.039
A. Derfoul, F.J. Lin, E.M. Awumey, T. Kolodzeski, D.J. Hall, R.S. Tuan, Estrogenic endocrine disruptive components interfere with calcium handling and differentiation of human trophoblast cells. J. Cell Biochem. 89(4), 755–770 (2003). https://doi.org/10.1002/jcb.10558
V.L. Bosquiazzo, J. Varayoud, M. Muñoz-de-Toro, E.H. Luque, J.G. Ramos, Effects of neonatal exposure to bisphenol A on steroid regulation of vascular endothelial growth factor expression and endothelial cell proliferation in the adult rat uterus. Biol. Reprod. 82(1), 86–95 (2010). https://doi.org/10.1095/biolreprod.109.078543
P.W. Chu, Z.J. Yang, H.H. Huang, A.A. Chang, Y.C. Cheng, G.J. Wu, H.C. Lan, Low-dose bisphenol A activates the ERK signaling pathway and attenuates steroidogenic gene expression in human placental cells. Biol. Reprod. 98(2), 250–258 (2018). https://doi.org/10.1093/biolre/iox162
R. Alarcón, O.E. Rivera, P.I. Ingaramo, M.V. Tschopp, G.H. Dioguardi, M.M. Milesi, M. Muñoz-de-Toro, E.H. Luque, Neonatal exposure to a glyphosate-based herbicide alters the uterine differentiation of prepubertal ewe lambs. Environ. Pollut. 265, 114874 (2020). https://doi.org/10.1016/j.envpol.2020.114874
V. Lorenz, G. Pacini, E.H. Luque, J. Varayoud, M.M. Milesi, Perinatal exposure to glyphosate or a glyphosate-based formulation disrupts hormonal and uterine milieu during the receptive state in rats. Food Chem. Toxicol. 143, 111560 (2020). https://doi.org/10.1016/j.fct.2020.111560
R.L. Ruhlen, K.L. Howdeshell, J. Mao, J.A. Taylor, F.H. Bronson, R.R. Newbold, W.V. Welshons, F.S. vom Saal, Low phytoestrogen levels in feed increase fetal serum estradiol resulting in the “fetal estrogenization syndrome” and obesity in CD-1 mice. Environ. Health Perspect. 116, 322–328 (2008). https://doi.org/10.1289/ehp.10448
N. Chernis, P. Masschelin, A.R. Cox, S.M. Hartig, Bisphenol AF promotes inflammation in human white adipocytes. Am. J. Physiol. Cell Physiol. 318, 63–72 (2020). https://doi.org/10.1152/ajpcell.00175.2019
H.X. Wang, Y. Zhou, C.X. Tang, J.G. Wu, Y. Chen, Q.W. Jiang, Association between bisphenol A exposure and body mass index in Chinese school children: a cross-sectional study. Environ. Health 11, 79 (2012). https://doi.org/10.1186/1476-069X-11-79
S. Kirchner, T. Kieu, C. Chow, S. Casey, B. Blumberg, Prenatal exposure to the environmental obesogen tributyltin predisposes multipotent stem cells to become adipocytes. Mol. Endocrinol. 24(3), 526–539 (2010). https://doi.org/10.1210/me.2009-0261
F. Grün, H. Watanabe, Z. Zamanian, L. Maeda, K. Arima, R. Cubacha, D.M. Gardiner, J. Kanno, T. Iguchi, B. Blumberg, Endocrine-disrupting organotin compounds are potent inducers of adipogenesis in vertebrates. Mol. Endocrinol. 20(9), 2141–2155 (2006). https://doi.org/10.1210/me.2005-0367
B. Leppert, S. Strunz, B. Seiwert, L. Schlittenbauer, R. Schlichting, C. Pfeiffer, S. Röder, M. Bauer, M. Borte, G.I. Stangl, T. Schöneberg, A. Schulz, I. Karkossa, U.E. Rolle-Kampczyk, L. Thürmann, M. von Bergen, B.I. Escher, K.M. Junge, T. Reemtsma, I. Lehmann, T. Polte, Maternal paraben exposure triggers childhood overweight development. Nat. Commun. 11, 561 (2020). https://doi.org/10.1038/s41467-019-14202-1
M. La Merrill, E. Karey, E. Moshier, C. Lindtner, M.R. La Frano, J.W. Newman, C. Buettner, Perinatal exposure of mice to the pesticide DDT impairs energy expenditure and metabolism in adult female offspring. PLoS One 9(7), 103337 (2014). https://doi.org/10.1371/journal.pone.0103337
M. Warner, A. Wesselink, K.G. Harley, A. Bradman, K. Kogut, B. Eskenazi, Prenatal exposure to dichlorodiphenyltrichloroethane and obesity at 9 years of age in the CHAMACOS study cohort. Am. J. Epidemiol. 179(11), 1312–1322 (2014). https://doi.org/10.1093/aje/kwu046
P. Gigante, M. Berni, S. Bussolati, F. Grasselli, S. Grolli, R. Ramoni, G. Basini, Glyphosate affects swine ovarian and adipose stromal cell functions. Anim. Reprod. Sci. 195, 185–196 (2018). https://doi.org/10.1016/j.anireprosci.2018.05.023
B.C. Gladen, N.B. Ragan, W.J. Rogan, Pubertal growth and development and prenatal and lactational exposure to polychlorinated biphenyls and dichlorodiphenyl dichloroethene. J. Pediatr. 136(4), 490–496 (2000). https://doi.org/10.1016/s0022-3476(00)90012-x
Y. Sakamoto, A. Naka, N. Ohara, K. Kondo, K. Iida, Daidzein regulates proinflammatory adipokines thereby improving obesity-related inflammation through PPARγ. Mol. Nutr. Food Res. 58, 718–726 (2014). https://doi.org/10.1002/mnfr.201300482
R.R. Newbold, Lessons learned from perinatal exposure to diethylstilbestrol. Toxicol. Appl Pharm. 199(2), 142–150 (2004). https://doi.org/10.1016/j.taap.2003.11.033
H. MacKay, Z.R. Patterson, A. Abizaid, Perinatal exposure to low-dose bisphenol-a disrupts the structural and functional development of the hypothalamic feeding circuitry. Endocrinology 158(4), 768–777 (2017). https://doi.org/10.1210/en.2016-1718
F. Ariemma, V. D’Esposito, D. Liguoro, F. Oriente, S. Cabaro, A. Liotti, I. Cimmino, M. Longo, F. Beguinot, P. Formisano, R. Valentino, Low-dose bisphenol-A impairs adipogenesis and generates dysfunctional 3T3-L1 adipocytes. PLoS One 11(3), 0150762 (2016). https://doi.org/10.1371/journal.pone.0150762
R.J. Denver, R.M. Bonett, G.C. Boorse, Evolution of leptin structure and function. Neuroendocrinology 94(1), 21–38 (2011). https://doi.org/10.1159/000328435
A. Ewart-Toland, K. Mounzih, J. Qiu, F.F. Chehab, Effect of the genetic background on the reproduction of leptin-deficient obese mice. Endocrinology 140(2), 732–738 (1999). https://doi.org/10.1210/endo.140.2.6470
A. Pérez-Pérez, F. Sánchez-Jiménez, J. Maymó, J.L. Dueñas, C. Varone, V. Sánchez-Margalet, Role of leptin in female reproduction. Clin. Chem. Lab. Med. 53(1), 15–28 (2015). https://doi.org/10.1515/cclm-2014-0387
M. Zerani, C. Boiti, D. Zampini, G. Brecchia, C. Dall’Aglio, P. Ceccarelli, A. Gobbetti, Ob receptor in rabbit ovary and leptin in vitro regulation of corpora lutea. J. Endocrinol. 183(2), 279–288 (2004). https://doi.org/10.1677/joe.1.05507
M.P. Magariños, V. Sánchez-Margalet, M. Kotler, J.C. Calvo, C.L. Varone, Leptin promotes cell proliferation and survival of trophoblastic cells. Biol. Reprod. 76(2), 203–210 (2007). https://doi.org/10.1095/biolreprod.106.051391
Z.V. Wang, P.E. Scherer, Adiponectin, the past two decades. J. Mol. Cell Biol. 8(2), 93–100 (2016). https://doi.org/10.1093/jmcb/mjw011
H. Fang, R.L. Judd, Adiponectin regulation and function. Compr. Physiol. 8(3), 1031–1063 (2018). https://doi.org/10.1002/cphy.c170046
M. Lu, Q. Tang, J.M. Olefsky, P.L. Mellon, N.J.G. Webster, Adiponectin activates adenosine monophosphate-activated protein kinase and decreases luteinizing hormone secretion in LbetaT2 gonadotropes. Mol. Endocrinol. 22(3), 760–771 (2008). https://doi.org/10.1210/me.2007-0330
D. Lagaly, P. Aad, J. Grado-Ahuir, L. Hulsey, L. Spicer, Role of adiponectin in regulating ovarian theca and granulose cell function. Mol. Cell. Endocrinol. 284, 38–45 (2008). https://doi.org/10.1016/j.mce.2008.01.007
J. Li, X.J. Ma, X. Wu, S.J. Si, C. Li, P.K. Yang, G.X. Li, X.J. Liu, Y.D. Tian, X.T. Kang, Adiponectin modulates steroid hormone secretion, granulosa cell proliferation and apoptosis via binding its receptors during hens’ high laying period. Poult. Sci. 100(7), 101197 (2021). https://doi.org/10.1016/j.psj.2021.101197
E.A. Adu-Gyamfi, L.A. Fondjo, W.K.B.A. Owiredu, A. Czika, W. Nelson, J. Lamptey, Y.X. Wang, Y.B. Ding, The role of adiponectin in placentation and preeclampsia. Cell Biochem. Funct. 38(1), 106–117 (2020). https://doi.org/10.1002/cbf.3458
K. Bozaoglu, K. Bolton, J. McMillan, P. Zimmet, J. Jowett, G. Collier, K. Walder, D. Segal, Chemerin is a novel adipokine associated with obesity and metabolic syndrome. Endocrinology 148(10), 4687–4694 (2007). https://doi.org/10.1210/en.2007-0175
N. Smolinska, M. Kiezun, K. Dobrzyn, E. Rytelewska, K. Kisielewska, M. Gudelska, E. Zaobidna, K. Bogus-Nowakowska, J. Wyrebek, K. Bors, G. Kopij, B. Kaminska, T. Kaminski, Expression of chemerin and its receptors in the porcine hypothalamus and plasma chemerin levels during the oestrous cycle and early pregnancy. Int. J. Mol. Sci. 20(16), 3887 (2019). https://doi.org/10.3390/ijms20051128
M. Diot, M. Reverchon, C. Rame, P. Froment, J.P. Brillard, S. Briere, G. Leveque, D. Guillaume, J. Dupont, Expression of adiponectin, chemerin and visfatin in plasma and different tissues during a laying season in turkeys. Reprod. Biol. Endocrinol. 13, 81 (2015). https://doi.org/10.1186/s12958-015-0081-5
M. Reverchon, M. Cornuau, C. Rame, F. Guerif, D. Royere, J. Dupont, Chemerin inhibits IGF-1-induced progesterone and estradiol secretion in human granulosa cells. Hum. Reprod. 27(6), 1790–1800 (2012). https://doi.org/10.1093/humrep/des089
M. Reverchon, M.J. Bertoldo, C. Rame, P. Froment, J. Dupont, CHEMERIN (RARRES2) decreases in vitrogranulosa cell steroidogenesis and blocks oocyte meiotic progression in bovine species. Biol. Reprod. 90(5), 102 (2014). https://doi.org/10.1095/biolreprod.113.117044
K. Tatemoto, M. Hosoya, Y. Habata, R. Fujii, T. Kakegawa, M.X. Zou, Y. Kawamata, S. Fukusumi, S. Hinuma, C. Kitada, T. Kurokawa, H. Onda, M. Fujino, Isolation and characterization of a novel endogenous peptide ligand for the human APJ receptor. Biochem. Biophys. Res. Commun. 251, 471–476 (1998). https://doi.org/10.1006/bbrc.1998.9489
A.M. O’Carroll, S.J. Lolait, L.E. Harris, G.R. Pope, The apelin receptor APJ: journey from an orphan to a multifaceted regulator of homeostasis. J. Endocrinol. 219(1), 13–35 (2013). https://doi.org/10.1530/JOE-13-0227
A. Rak, E. Drwal, C. Rame, K. Knapczyk-Stwora, M. Slomczynska, J. Dupont, E.L. Gregoraszczuk, Expression of apelin and apelin receptor (APJ) in porcine ovarian follicles and in vitro effect of apelin on steroidogenesis and proliferation through APJ activation and different signaling pathways. Theriogenology 96, 126–135 (2017). https://doi.org/10.1016/j.theriogenology.2017.04.014
M. Różycka, P. Kurowska, M. Grzesiak, M. Kotula-Balak, W. Tworzydło, C. Rame, E. Gregoraszczuk, J. Dupont, A. Rak, Apelin and apelin receptor at different stages of corpus luteum development and effect of apelin on progesterone secretion and 3β-hydroxysteroid dehydrogenase (3β-HSD) in pigs. Anim. Reprod. Sci. 192, 251–260 (2018). https://doi.org/10.1016/j.anireprosci.2018.03.021
J. Roche, C. Rame, M. Reverchon, N. Mellouk, A. Rak, P. Froment, J. Dupont, Apelin (APLN) regulates progesterone secretion and oocyte maturation in bovine ovarian cells. Reproduction 153(5), 589–603 (2017). https://doi.org/10.1530/REP-16-0677
T. Shimizu, N. Kosaka, C. Murayama, M. Tetsuka, A. Miyamoto, Apelin and APJ receptor expression in granulosa and theca cells during different stages of follicular development in the bovine ovary: Involvement of apoptosis and hormonal regulation. Anim. Reprod. Sci. 116, 28–37 (2009). https://doi.org/10.1016/j.anireprosci.2009.01.009
K. Tatemoto, K. Takayama, M.X. Zou, I. Kumaki, W. Zhang, K. Kumano, M. Fujimiya, The novel peptide apelin lowers blood pressure via a nitric oxide-dependent mechanism. Regul. Pep. 99, 87–92 (2001). https://doi.org/10.1016/s0167-0115(01)00236-1
M. Dawid, E. Mlyczynska, P. Kurowska, M. Sierpowski, A. Rak, Apelin decreased placental hormone secretion by human trophoblast BeWo cells via apelin receptor, protein kinase A and extracellular signal-regulated kinases 1/2 activation. J. Physiol. Pharmacol. 70(6), 895–907 (2019). https://doi.org/10.26402/jpp.2019.6.08
E. Mlyczyńska, M. Myszka, P. Kurowska, M. Dawid, T. Milewicz, M. Bałajewicz-Nowak, P. Kowalczyk, A. Rak, Anti-apoptotic effect of apelin in human placenta: studies on BeWo cells and villous explants from third-trimester human pregnancy. Int. J. Mol. Sci. 22(5), 2760 (2021). https://doi.org/10.3390/ijms22052760
M.S. Jamaluddin, S.M. Weakley, Q. Yao, C. Chen, Resistin: functional roles and therapeutic considerations for cardiovascular disease. Br. J. Pharm. 165(3), 622–632 (2012). https://doi.org/10.1111/j.1476-5381.2011.01369.x
D. Tripathi, S. Kant, S. Pandey, N.Z. Ehtesham, Resistin in metabolism, inflammation, and disease. FEBS J. 287(15), 3141–3149 (2020). https://doi.org/10.1111/febs.15322
A. Rak-Mardyła, M. Duda, E.Ł. Gregoraszczuk, A role for resistin in the ovary during the estrous cycle. Horm. Metab. Res. 46(7), 493–498 (2014). https://doi.org/10.1055/s-0034-1370909
A. Rak-Mardyła, M. Durak, E.L. Gregoraszczuk, Effects of resistin on porcine ovarian follicle steroidogenesis in prepubertal animals: an in vitro study. Reprod. Biol. Endocrinol. 11, 45 (2013). https://doi.org/10.1186/1477-7827-11-45
V. Maillard, P. Froment, C. Rame, S. Uzbekova, S. Elis, J. Dupont, Expression and effect of resistin on bovine and rat granulosa cell steroidogenesis and proliferation. Reproduction 141(4), 467–479 (2011). https://doi.org/10.1530/REP-10-0419
A. Rak, E. Drwal, A. Wróbel, E.Ł. Gregoraszczuk, Resistin is a survival factor for porcine ovarian follicular cells. Reproduction 150(4), 343–355 (2015). https://doi.org/10.1530/REP-15-0255
P. Kurowska, M. Sroka, M. Dawid, E. Mlyczyńska, N. Respekta, M. Jurek, D. Klimczyk, M. Grzesiak, J. Dupont, A. Rak, Expression and role of resistin on steroid secretion in the porcine corpus luteum. Reproduction 162(4), 237–248 (2021). https://doi.org/10.1530/REP-21-0236
R. Tassinari, L. Narciso, S. Tait, L. Busani, A. Martinelli, A. Di Virgilio, F. Carli, A. Deodati, C. La Rocca, F. Maranghi, Juvenile toxicity rodent model to study toxicological effects of bisphenol A (BPA) at dose levels derived from italian children biomonitoring study. Toxicol. Sci. 173, 387–401 (2020). https://doi.org/10.1093/toxsci/kfz226
B.M. Angle, R.P. Do, D. Ponzi, R.W. Stahlhut, B.E. Drury, S.C. Nagel, W.V. Welshons, C.L. Besch-Williford, P. Palanza, S. Parmigiani, F.S. vom Saal, J.A. Taylor, Metabolic disruption in male mice due to fetal exposure to low but not high doses of bisphenol A (BPA): evidence for effects on body weight, food intake, adipocytes, leptin, adiponectin, insulin and glucose regulation. Reprod. Toxicol. 42, 256–268 (2013). https://doi.org/10.1016/j.reprotox.2013.07.017
T. Kidani, S. Kamei, J. Miyawaki, J. Aizawa, K. Sakayama, H. Masuno, Bisphenol A downregulates Akt signaling and inhibits adiponectin production and secretion in 3T3-L1 adipocytes. J. Atheroscler. Thromb. 17, 834–43 (2010). https://doi.org/10.5551/jat.4051
C. Menale, A. Grandone, C. Nicolucci, G. Cirillo, S. Crispi, A. Di Sessa, P. Marzuillo, S. Rossi, D.G. Mita, L. Perrone, N. Diano, E. Miraglia Del Giudice, Bisphenol A is associated with insulin resistance and modulates adiponectin and resistin gene expression in obese children. Pediatr. Obes. 12, 380–387 (2017). https://doi.org/10.1111/ijpo.12154
D.J. Watkins, K.E. Peterson, K.K. Ferguson, A. Mercado-García, M. Tamayo Ortiz, A. Cantoral, J.D. Meeker, M.M. Téllez-Rojo, Relating phthalate and BPA exposure to metabolism in peripubescence: the role of exposure timing, sex, and puberty. J. Clin. Endocrinol. Metab. 101, 79–88 (2016). https://doi.org/10.1210/jc.2015-2706
J. Ashley-Martin, L. Dodds, T.E. Arbuckle, A.S. Ettinger, G.D. Shapiro, M. Fisher, A.S. Morisset, S. Taback, M.F. Bouchard, P. Monnier, R. Dallaire, W.D. Fraser, A birth cohort study to investigate the association between prenatal phthalate and bisphenol A exposures and fetal markers of metabolic dysfunction. Environ. Health 13, 84 (2014). https://doi.org/10.1186/1476-069X-13-84
M. Rönn, L. Lind, J. Örberg, J. Kullberg, S. Söderberg, A. Larsson, L. Johansson, H. Ahlström, P.M. Lind, Bisphenol A is related to circulating levels of adiponectin, leptin and ghrelin, but not to fat mass or fat distribution in humans. Chemosphere 112, 42–48 (2014). https://doi.org/10.1016/j.chemosphere.2014.03.042
L. Ceotto Freitas-Lima, E. Merlo, M. Campos Zicker, J.M. Navia-Pelaez, M. de Oliveira, L. Dos Santos Aggum Capettini, C.R. Nogueira, A. Versiani Matos Ferreira, S.H. Sousa Santos, J. Bernardes Graceli, Tributyltin impacts in metabolic syndrome development through disruption of angiotensin II receptor signaling pathways in white adipose tissue from adult female rats. Toxicol. Lett. 299, 21–31 (2018). https://doi.org/10.1016/j.toxlet.2018.08.018
C. Hao, X. Cheng, J. Guo, H. Xia, X. Ma, Perinatal exposure to diethyl-hexyl-phthalate induces obesity in mice. Front. Biosci. (Elite Ed.) 5, 725–733 (2013). https://doi.org/10.2741/e653
L. Zhou, H. Chen, Q. Xu, X. Han, Y. Zhao, X. Song, T. Zhao, L. Ye, The effect of di-2-ethylhexyl phthalate on inflammation and lipid metabolic disorder in rats. Ecotoxicol. Environ. Saf. 170, 391–398 (2019). https://doi.org/10.1016/j.ecoenv.2018.12.009
A. Rak, K. Zajda, E.Ł. Gregoraszczuk, Endocrine disrupting compounds modulates adiponectin secretion, expression of its receptors and action on steroidogenesis in ovarian follicle. Reprod. Toxicol. 69, 204–211 (2017). https://doi.org/10.1016/j.reprotox.2017.03.004
R.G. Ahmed, Early weaning PCB 95 exposure alters the neonatal endocrine system: thyroid adipokine dysfunction. J. Endocrinol. 219, 205–215 (2013). https://doi.org/10.1530/JOE-13-0302
J.E. Lim, S.H. Jee, Association between serum levels of adiponectin and polychlorinated biphenyls in Korean men and women. Endocrine 48, 211–7 (2015). https://doi.org/10.1007/s12020-014-0231-0
G. Howell III, L. Mangum, Exposure to bioaccumulative organochlorine compounds alters adipogenesis, fatty acid uptake, and adipokine production in NIH3T3-L1 cells. Toxicol. Vitr. 25(1), 394–402 (2011). https://doi.org/10.1016/j.tiv.2010.10.015
M.J. Lee, H. Lin, C.W. Liu, M.H. Wu, W.J. Liao, H.H. Chang, H.C. Ku, Y.S. Chien, W.H. Ding, Y.H. Kao, Octylphenol stimulates resistin gene expression in 3T3-L1 adipocytes via the estrogen receptor and extracellular signal-regulated kinase pathways. Am. J. Physiol. Cell Physiol. 294(6), 1542–1551 (2008). https://doi.org/10.1152/ajpcell.00403.2007
M. Hoffmann, A. Rak, A. Ptak, Bisphenol A and its derivatives decrease expression of chemerin, which reverses its stimulatory action in ovarian cancer cells. Toxicol. Lett. 291, 61–69 (2018). https://doi.org/10.1016/j.toxlet.2018.04.004
M. Hoffmann, E. Fiedor, A. Ptak, Bisphenol A and its derivatives tetrabromobisphenol A and tetrachlorobisphenol A induce apelin expression and secretion in ovarian cancer cells through a peroxisome proliferator-activated receptor gamma-dependent mechanism. Toxicol. Lett. 269, 15–22 (2017). https://doi.org/10.1016/j.toxlet.2017.01.006
W.C. Chou, J.L. Chen, C.F. Lin, Y.C. Chen, F.C. Shih, C.Y. Chuang, Biomonitoring of bisphenol A concentrations in maternal and umbilical cord blood in regard to birth outcomes and adipokine expression: a birth cohort study in Taiwan. Environ. Health 10, 94 (2011). https://doi.org/10.1186/1476-069X-10-94
G.C. Sena, L.C. Freitas-Lima, E. Merlo, P.L. Podratz, J.F. de Araújo, P.A. Brandão, M.T. Carneiro, M.C. Zicker, A.V. Ferreira, C.M. Takiya, C.M. de Lemos Barbosa, M.M. Morales, A.P. Santos-Silva, L. Miranda-Alves, I.V. Silva, J.B. Graceli, Environmental obesogen tributyltin chloride leads to abnormal hypothalamic-pituitary-gonadal axis function by disruption in kisspeptin/leptin signaling in female rats. Toxicol. Appl Pharm. 319, 22–38 (2017). https://doi.org/10.1016/j.taap.2017.01.021
A.V. Sirotkin, R. Alexa, A. Kádasi, A. Štochmaľová, M. Morovič, J. Laurinčik, A.H. Harrath, R. Grossmann, The isoflavone daidzein directly affects porcine ovarian cell functions and modifies the effect of follicle-stimulating hormone. J. Anim. Physiol. Anim. Nutr. (Berl.). 101, 127–135 (2017). https://doi.org/10.1111/jpn.12520
D. Franssen, Y.S. Ioannou, A. Alvarez-real, A. Gerard, J.K. Mueller, S. Heger, J.P. Bourguignon, A.S. Parent, Pubertal timing after neonatal diethylstilbestrol exposure in female rats: neuroendocrine vs peripheral effects and additive role of prenatal food restriction. Reprod. Toxicol. 44, 63–72 (2014). https://doi.org/10.1016/j.reprotox.2013.10.006
J. Boberg, S. Metzdorff, R. Wortziger, M. Axelstad, L. Brokken, A.M. Vinggaard, M. Dalgaard, C. Nellemann, Impact of diisobutyl phthalate and other PPAR agonists on steroidogenesis and plasma insulin and leptin levels in fetal rats. Toxicology 250, 75–81 (2008). https://doi.org/10.1016/j.tox.2008.05.020
B. Wahlang, J. Jin, J.E. Hardesty, K.Z. Head, H. Shi, K.C. Falkner, R.A. Prough, C.M. Klinge, M.C. Cave, Identifying sex differences arising from polychlorinated biphenyl exposures in toxicant-associated liver disease. Food Chem. Toxicol. 129, 64–76 (2019). https://doi.org/10.1016/j.fct.2019.04.007
S. Otokozawa, R. Tanaka, H. Akasaka, E. Ito, S. Asakura, H. Ohnishi, S. Saito, T. Miura, T. Saito, M. Mori, Associations of serum isoflavone, adiponectin and insulin levels with risk for epithelial ovarian cancer: results of a case-control study. Asian Pac. J. Cancer Prev. 16, 4987–4991 (2015). https://doi.org/10.7314/apjcp.2015.16.12.4987
Funding
Support was received from Région Centre Val de Loire (HAPOFERTI project number 32000858). P.K. obtained funding as part of the PhD scholarship ETIUDA programme: 2020/36/T/NZ9/00264.
Author information
Authors and Affiliations
Contributions
Conceptualisation: J.D., A.R.; Literature Search, Writing—Original Draft: P.K., E.M., M.D., N.R., K.P., L.S., J.D., A.R.; Writing—Review & Editing: P.K., J.D., A.R.; Supervision: A.R. All authors read and accepted final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kurowska, P., Mlyczyńska, E., Dawid, M. et al. Endocrine disruptor chemicals, adipokines and reproductive functions. Endocrine 78, 205–218 (2022). https://doi.org/10.1007/s12020-022-03061-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03061-4