Abstract
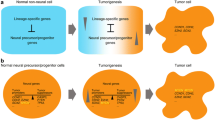
Tumorigenic cells are similar to neural stem cells or embryonic neural cells in regulatory networks, tumorigenicity and pluripotent differentiation potential. By integrating the evidence from developmental biology, tumor biology and evolution, I will make a detailed discussion on the observations and propose that neural stemness underlies two coupled cell properties, tumorigenicity and pluripotent differentiation potential. Neural stemness property of tumorigenic cells can hopefully integrate different observations/concepts underlying tumorigenesis. Neural stem cells and tumorigenic cells share regulatory networks; both exhibit neural stemness, tumorigenicity and pluripotent differentiation potential; both depend on expression or activation of ancestral genes; both rely primarily on aerobic glycolytic metabolism; both can differentiate into various cells/tissues that are derived from three germ layers, leading to tumor formation resembling severely disorganized or more degenerated process of embryonic tissue differentiation; both are enriched in long genes with more splice variants that provide more plastic scaffolds for cell differentiation, etc. Neural regulatory networks, which include higher levels of basic machineries of cell physiological functions and developmental programs, work concertedly to define a basic state with fast cell cycle and proliferation. This is predestined by the evolutionary advantage of neural state, the ground or initial state for multicellularity with adaptation to an ancient environment. Tumorigenesis might represent a process of restoration of neural ground state, thereby restoring a state with fast proliferation and pluripotent differentiation potential in somatic cells. Tumorigenesis and pluripotent differentiation potential might be better understood from understanding neural stemness, and cancer therapy should benefit more from targeting neural stemness.
Graphical Abstract




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Hajdu, S. I. (2011). A note from history: landmarks in history of cancer, part 1. Cancer, 117, 1097–1102
Burrell, R. A., McGranahan, N., Bartek, J., & Swanton, C. (2013). The causes and consequences of genetic heterogeneity in cancer evolution. Nature, 501, 338–345
Meacham, C. E., & Morrison, S. J. (2013). Tumour heterogeneity and cancer cell plasticity. Nature, 501, 328–337
Friedmann-Morvinski, D., & Verma, I. M. (2014). Dedifferentiation and reprogramming: origins of cancer stem cells. EMBO Reports, 15, 244–253
Prasetyanti, P. R., & Medema, J. P. (2017). Intra-tumor heterogeneity from a cancer stem cell perspective. Molecular Cancer, 16, 41
Dagogo-Jack, I., & Shaw, A. T. (2018). Tumour heterogeneity and resistance to cancer therapies. Nature Reviews. Clinical Oncology, 15, 81–94
Paduch, R. (2015). Theories of cancer origin. European Journal of Cancer Prevention, 24, 57–67
Hanselmann, R. G., & Welter, C. (2016). Origin of cancer: an information, energy, and matter disease. Frontiers in Cell and Developmental Biology, 4, 121
Gilbert, S. F., & Barresi, M. J. (2016). Early amphibian development. In Developmental Biology (11th Ed., pp. 333-364). Sinauer Associates, Inc
Grunz, H., & Tacke, L. (1989). Neural differentiation of Xenopus laevis ectoderm takes place after disaggregation and delayed reaggregation without inducer. Cell Differentiation and Development, 28, 211–217
Godsave, S. F., & Slack, J. M. (1989). Clonal analysis of mesoderm induction in Xenopus laevis. Developmental Biology, 134, 486–490
Sato, S. M., & Sargent, T. D. (1989). Development of neural inducing capacity in dissociated Xenopus embryos. Developmental Biology, 134, 263–266
Harland, R. (2000). Neural induction. Current Opinion in Genetics & Development, 10, 357–362
De Robertis, E. M., & Kuroda, H. (2004). Dorsal-ventral patterning and neural induction in Xenopus embryos. Annual Review of Cell and Developmental Biology, 20, 285–308
De Robertis, E. M. (2006). Spemann’s organizer and self-regulation in amphibian embryos. Nature Reviews. Molecular Cell Biology, 7, 296–302
Muñoz-Sanjuán, I., & Brivanlou, A. H. (2002). Neural induction, the default model and embryonic stem cells. Nature Reviews. Neuroscience, 3, 271–280
Tropepe, V., Hitoshi, S., Sirard, C., Mak, T. W., Rossant, J., & van der Kooy, D. (2001). Direct neural fate specification from embryonic stem cells: a primitive mammalian neural stem cell stage acquired through a default mechanism. Neuron, 30, 65–78
Ying, Q. L., Stavridis, M., Griffiths, D., Li, M., & Smith, A. (2003). Conversion of embryonic stem cells into neuroectodermal precursors in adherent monoculture. Nature Biotechnology, 21, 183–186
Smukler, S. R., Runciman, S. B., Xu, S., & van der Kooy, D. (2006). Embryonic stem cells assume a primitive neural stem cell fate in the absence of extrinsic influences. The Journal of Cell Biology, 172, 79–90
Ying, Q. L., Nichols, J., Chambers, I., & Smith, A. (2003). BMP induction of Id proteins suppresses differentiation and sustains embryonic stem cell self-renewal in collaboration with STAT3. Cell, 115, 281–292
Malaguti, M., Nistor, P. A., Blin, G., Pegg, A., Zhou, X., & Lowell, S. (2013). Bone morphogenic protein signalling suppresses differentiation of pluripotent cells by maintaining expression of E-Cadherin. eLife, 2, e01197
Ben-David, U., & Benvenisty, N. (2011). The tumorigenicity of human embryonic and induced pluripotent stem cells. Nature Reviews. Cancer, 11, 268–277
Zhang, Z., Lei, A., Xu, L., Chen, L., Chen, Y., Zhang, X., & Cao, Y. (2017). Similarity in gene-regulatory networks suggests that cancer cells share characteristics of embryonic neural cells. The Journal of Biological Chemistry, 292, 12842–12859
Cao, Y. (2017). Tumorigenesis as a process of gradual loss of original cell identity and gain of properties of neural precursor/progenitor cells. Cell & Bioscience, 7, 61
Lei, A., Chen, L., Zhang, M., Yang, X., Xu, L., Cao, N., & Cao, Y. (2019). EZH2 regulates protein stability via recruiting USP7 to mediate neuronal gene expression in cancer cells. Frontiers in Genetics, 10, 422
Neman, J., Termini, J., Wilczynski, S., Vaidehi, N., Choy, C., Kowolik, C. M., & Jandial, R. (2014). Human breast cancer metastases to the brain display GABAergic properties in the neural niche. Proceedings of the National Academy of Sciences of the United States of America, 111, 984–989
Boire, A., Brastianos, P. K., Garzia, L., & Valiente, M. (2020). Brain metastasis. Nature Reviews Cancer, 20, 4–11
Venkataramani, V., Tanev, D. I., Strahle, C., Studier-Fischer, A., Fankhauser, L., Kessler, T., & Kuner, T. (2019). Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature, 573(7775), 532–538
Venkatesh, H. S., Morishita, W., Geraghty, A. C., Silverbush, D., Gillespie, S. M., Arzt, M. … Monje, M. (2019). Electrical and synaptic integration of glioma into neural circuits. Nature, 573, 539–545
Zeng, Q., Michael, I. P., Zhang, P., Saghafinia, S., Knott, G., Jiao, W. … Hanahan, D. (2019). Synaptic proximity enables NMDAR signalling to promote brain metastasis. Nature, 573, 526–531
Reavis, H. D., Chen, H. I., & Drapkin, R. (2020). Tumor innervation: cancer has some nerve. Trends in Cancer, 6, 1059–1067
Mauffrey, P., Tchitchek, N., Barroca, V., Bemelmans, A. P., Firlej, V., Allory, Y. … Magnon, C. (2019). Progenitors from the central nervous system drive neurogenesis in cancer. Nature, 569, 672–678
Li, Z., Guo, X., Huang, H., Wang, C., Yang, F., Zhang, Y. … Xi, R. (2020). A switch in tissue stem cell identity causes neuroendocrine tumors in drosophila gut. Cell Reports, 30, 1724–17344
Müller, F. J., Goldmann, J., Löser, P., & Loring, J. F. (2010). A call to standardize teratoma assays used to define human pluripotent cell lines. Cell Stem Cell, 6, 412–414
Yasuda, S., & Sato, Y. (2015). Tumorigenicity assessment of human cell-processed therapeutic products. Biologicals, 43, 416–421
Germain, N. D., Hartman, N. W., Cai, C., Becker, S., Naegele, J. R., & Grabel, L. B. (2012). Teratocarcinoma formation in embryonic stem cell-derived neural progenitor hippocampal transplants. Cell Transplantation, 21, 1603–1611
Deng, J., Zhang, Y., Xie, Y., Zhang, L., & Tang, P. (2018). Cell transplantation for spinal cord injury: tumorigenicity of induced pluripotent stem cell-derived neural stem/progenitor cells. Stem Cells International, 2018, 5653787
Amariglio, N., Hirshberg, A., Scheithauer, B. W., Cohen, Y., Loewenthal, R., Trakhtenbrot, L. … Rechavi, G. (2009). Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient. PLoS Medicine, 6, e1000029
Xu, L., Zhang, M., Shi, L., Yang, X., Chen, L., Cao, N. … Cao, Y. (2021). Neural stemness contributes to cell tumorigenicity. Cell & Bioscience, 11, 21
Mullen, A. C., & Wrana, J. L. (2017). TGF-β family signaling in embryonic and somatic stem-cell renewal and differentiation. Cold Spring Harbor Perspectives in Biology, 9, a022186
Massagué, J. (2008). TGFbeta in cancer. Cell, 134, 215–230
Seoane, J., & Gomis, R. R. (2017). TGF-β family signaling in tumor suppression and cancer progression. Cold Spring Harbor Perspectives in Biology, 9, a022277
Clarke, D. L., Johansson, C. B., Wilbertz, J., Veress, B., Nilsson, E., Karlström, H. … Frisén, J. (2000). Generalized potential of adult neural stem cells. Science, 288, 1660–1663
Serakinci, N., Tulay, P., & Kalkan, R. (2018). Role of mesenchymal stem cells in cancer development and their use in cancer therapy. Advances in Experimental Medicine and Biology, 1083, 45–62
Ra, J. C., Shin, I. S., Kim, S. H., Kang, S. K., Kang, B. C., Lee, H. Y. … Kwon, E. (2011). Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells and Development, 20, 1297–1308
Sykova, E., & Forostyak, S. (2013). Stem cells in regenerative medicine. Laser Therapy, 22, 87–92
Yong, K. W., Choi, J. R., Dolbashid, A. S., & Safwani, W. (2018). Biosafety and bioefficacy assessment of human mesenchymal stem cells: what do we know so far? Regenerative Medicine, 13, 219–232
Lauvrak, S. U., Munthe, E., Kresse, S. H., Stratford, E. W., Namløs, H. M., Meza-Zepeda, L. A., & Myklebost, O. (2013). Functional characterisation of osteosarcoma cell lines and identification of mRNAs and miRNAs associated with aggressive cancer phenotypes. British Journal of Cancer, 109, 2228–2236
Méndez-Ferrer, S., Michurina, T. V., Ferraro, F., Mazloom, A. R., Macarthur, B. D., Lira, S. A. … Frenette, P. S. (2010). Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature, 466, 829–834
Ricci-Vitiani, L., Lombardi, D. G., Pilozzi, E., Biffoni, M., Todaro, M., Peschle, C., & De Maria, R. (2007). Identification and expansion of human colon-cancer-initiating cells. Nature, 445, 111–115
Fox, R. G., Lytle, N. K., Jaquish, D. V., Park, F. D., Ito, T., Bajaj, J. … Reya, T. (2016). Image-based detection and targeting of therapy resistance in pancreatic adenocarcinoma. Nature, 534, 407–411
Boumahdi, S., Driessens, G., Lapouge, G., Rorive, S., Nassar, D., Le Mercier, M. … Blanpain, C. (2014). SOX2 controls tumour initiation and cancer stem-cell functions in squamous-cell carcinoma. Nature, 511, 246–250
Nakanishi, Y., Seno, H., Fukuoka, A., Ueo, T., Yamaga, Y., Maruno, T. … Chiba, T. (2013). Dclk1 distinguishes between tumor and normal stem cells in the intestine. Nature Genetics, 45, 98–103
McGrail, M., Batz, L., Noack, K., Pandey, S., Huang, Y., Gu, X., & Essner, J. J. (2010). Expression of the zebrafish CD133/prominin1 genes in cellular proliferation zones in the embryonic central nervous system and sensory organs. Developmental Dynamics, 239, 1849–1857
Mizuguchi, M., Qin, J., Yamada, M., Ikeda, K., & Takashima, S. (1999). High expression of doublecortin and KIAA0369 protein in fetal brain suggests their specific role in neuronal migration. The American Journal of Pathology, 155, 1713–1721
Matsumoto, N., Pilz, D. T., & Ledbetter, D. H. (1999). Genomic structure, chromosomal mapping, and expression pattern of human DCAMKL1 (KIAA0369), a homologue of DCX (XLIS). Genomics, 56, 179–183
Thisse, B., Pflumio, S., Fürthauer, M., Loppin, B., Heyer, V., Degrave, A. … Thisse, C. (2001). Expression of the zebrafish genome during embryogenesis (NIH R01 RR15402). ZFIN Direct Data Submission (http://zfin.org)
Han, R., Wang, R., Zhao, Q., Han, Y., Zong, S., Miao, S. … Wang, L. (2016). Trim69 regulates zebrafish brain development by ap-1 pathway. Scientific Reports, 6, 24034
Chen, J., Niu, N., Zhang, J., Qi, L., Shen, W., Donkena, K. V. … Liu, J. (2019). Polyploid Giant Cancer Cells (PGCCs): the evil roots of cancer. Current Cancer Drug Targets, 19, 360–367
Moein, S., Adibi, R., da Silva Meirelles, L., Nardi, N. B., & Gheisari, Y. (2020). Cancer regeneration: Polyploid cells are the key drivers of tumor progression. Biochimica et Biophysica Acta. Reviews on Cancer, 1874, 188408
Was, H., Borkowska, A., Olszewska, A., Klemba, A., Marciniak, M., Synowiec, A., & Kieda, C. (2021). Polyploidy formation in cancer cells: How a Trojan horse is born. Seminars in Cancer Biology, S1044-579X(21), 00053–00055 Advance online publication
Saleh, T., Carpenter, V. J., Bloukh, S., & Gewirtz, D. A. (2020). Targeting tumor cell senescence and polyploidy as potential therapeutic strategies. Seminars in Cancer Biology, S1044-579X(20), 30270–30274 Advance online publication
Saini, G., Joshi, S., Garlapati, C., Li, H., Kong, J., Krishnamurthy, J. … Aneja, R. (2021). Polyploid giant cancer cell characterization: New frontiers in predicting response to chemotherapy in breast cancer. Seminars in Cancer Biology, S1044-579 × (21)00067-5
Niu, N., Mercado-Uribe, I., & Liu, J. (2017). Dedifferentiation into blastomere-like cancer stem cells via formation of polyploid giant cancer cells. Oncogene, 36, 4887–4900
Pruszak, J., Sonntag, K. C., Aung, M. H., Sanchez-Pernaute, R., & Isacson, O. (2007). Markers and methods for cell sorting of human embryonic stem cell-derived neural cell populations. Stem Cells, 25, 2257–2268
Chen, K. A., Laywell, E. D., Marshall, G., Walton, N., Zheng, T., & Steindler, D. A. (2006). Fusion of neural stem cells in culture. Experimental Neurology, 198, 129–135
Marusyk, A., Almendro, V., & Polyak, K. (2012). Intra-tumour heterogeneity: a looking glass for cancer? Nature Reviews. Cancer, 12, 323–334
McGranahan, N., & Swanton, C. (2015). Biological and therapeutic impact of intratumor heterogeneity in cancer evolution. Cancer Cell, 27, 15–26
Quintanal-Villalonga, Ã., Chan, J. M., Yu, H. A., Pe’er, D., Sawyers, C. L., Sen, T., & Rudin, C. M. (2020). Lineage plasticity in cancer: a shared pathway of therapeutic resistance. Nature Reviews. Clinical Oncology, 17, 360–371
Clevers, H. (2011). The cancer stem cell: premises, promises and challenges. Nature Medicine, 17, 313–319
Beck, B., & Blanpain, C. (2013). Unravelling cancer stem cell potential. Nature Reviews. Cancer, 13, 727–738
Shmelkov, S. V., Butler, J. M., Hooper, A. T., Hormigo, A., Kushner, J., Milde, T., … Rafii, S. (2008). CD133 expression is not restricted to stem cells, and both CD133+ and CD133- metastatic colon cancer cells initiate tumors. The Journal of Clinical Investigation, 118, 2111–2120
Beier, D., Hau, P., Proescholdt, M., Lohmeier, A., Wischhusen, J., Oefner, P. J. … Beier, C. P. (2007). CD133(+) and CD133(-) glioblastoma-derived cancer stem cells show differential growth characteristics and molecular profiles. Cancer Research, 67, 4010–4015
Mintz, B., & Illmensee, K. (1975). Normal genetically mosaic mice produced from malignant teratocarcinoma cells. Proceedings of the National Academy of Sciences of the United States of America, 72, 3585–3589
Papaioannou, V. E., McBurney, M. W., Gardner, R. L., & Evans, M. J. (1975). Fate of teratocarcinoma cells injected into early mouse embryos. Nature, 258, 70–73
Scaffidi, P., & Misteli, T. (2011). In vitro generation of human cells with cancer stem cell properties. Nature Cell Biology, 13, 1051–1061
Ricci-Vitiani, L., Pallini, R., Biffoni, M., Todaro, M., Invernici, G., Cenci, T. … De Maria, R. (2010). Tumour vascularization via endothelial differentiation of glioblastoma stem-like cells. Nature, 468, 824–828
Wang, R., Chadalavada, K., Wilshire, J., Kowalik, U., Hovinga, K. E., Geber, A. … Tabar, V. (2010). Glioblastoma stem-like cells give rise to tumour endothelium. Nature, 468, 829–833
Cheng, L., Huang, Z., Zhou, W., Wu, Q., Donnola, S., Liu, J. K. … Bao, S. (2013). Glioblastoma stem cells generate vascular pericytes to support vessel function and tumor growth. Cell, 153, 139–152
Vermeulen, L., Todaro, M., de Sousa Mello, F., Sprick, M. R., Kemper, K., Perez Alea, M. … Medema, J. P. (2008). Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proceedings of the National Academy of Sciences of the United States of America, 105, 13427–13432
Kim, K., Higashi, M., Fumino, S., & Tajiri, T. (2019). Derivation of neural stem cells from human teratomas. Stem Cell Research, 41, 101633
Yachida, S., Fukushima, N., Nakanishi, Y., Akasu, T., Kitamura, H., Sakamoto, M., & Shimoda, T. (2003). Alpha-fetoprotein-producing carcinoma of the colon: report of a case and review of the literature. Diseases of the Colon and Rectum, 46, 826–831
Anzai, H., Kazama, S., Kiyomatsu, T., Nishikawa, T., Tanaka, T., Tanaka, J. … Watanabe, T. (2015). Alpha-fetoprotein-producing early rectal carcinoma: a rare case report and review. World Journal of Surgical Oncology, 13, 180
Gong, W., Su, Y., Liu, A., Liu, J., Sun, D., Jiang, T., … Sun, P. (2018). Clinical characteristics and treatments of patients with alpha-fetoprotein producing gastric carcinoma. Neoplasma, 65, 326–330
Abid, H., & Gnanajothy, R. (2019). Osteoclast giant cell tumor of pancreas: a case report and literature review. Cureus, 11, e4710
Njoumi, N., Elalami, F. H., Attolou, G., Saoud, O., Elabsi, M., Echarrab, M. … Chkoff, M. R. (2014). Undifferentiated pancreatic carcinoma with osteoclast-like giant cells: a case report. Journal of Gastrointestinal Cancer, 45(Suppl 1), 96–98
Sah, S. K., Li, Y., & Li, Y. (2015). Undifferentiated carcinoma of the pancreas with osteoclast-like giant cells: a rare case report and review of the literature. International Journal of Clinical and Experimental Pathology, 8, 11785–11791
Togawa, Y., Tonouchi, A., Chiku, T., Sano, W., Doki, T., Yano, K. … Toyoda, A. (2010). A case report of undifferentiated carcinoma with osteoclast-like giant cells of the pancreas and literature review. Clinical Journal of Gastroenterology, 3, 195–203
Bauditz, J., Rudolph, B., & Wermke, W. (2006). Osteoclast-like giant cell tumors of the pancreas and liver. World Journal of Gastroenterology, 12, 7878–7883
Dioscoridi, L., Bisogni, D., & Freschi, G. (2015). Hepatocellular carcinoma with osteoclast-like giant cells: report of the seventh case in the literature. Case Reports in Surgery, 2015, 836105
Ikeda, T., Seki, S., Maki, M., Noguchi, N., Kawamura, T., Arii, S. … Hirokawa, K. (2003). Hepatocellular carcinoma with osteoclast-like giant cells: possibility of osteoclastogenesis by hepatocyte-derived cells. Pathology International, 53, 450–456
Kuwano, H., Sonoda, T., Hashimoto, H., & Enjoji, M. (1984). Hepatocellular carcinoma with osteoclast-like giant cells. Cancer, 54, 837–842
Rosai, J. (1990). Liver cell carcinoma with osteoclast-like giant cells: nonepitheliogenic giant cells in diverse malignancies. Hepatology, 12, 782–783
Goel, G., Rao, S., & Khurana, N. (2011). Malignant melanoma with osteoclast-like giant cells: A report of two cases. Journal of Cancer Research and Therapeutics, 7, 336–338
Al-Brahim, N., & Salama, S. (2005). Malignant melanoma with osteoclast-like giant cells: an unusual host response: immunohistochemical and ultrastructural study of three cases and literature review. The American Journal of Dermatopathology, 27, 126–129
Jiménez-Heffernan, J. A., Adrados, M., Muñoz-Hernández, P., Fernández-Rico, P., Ballesteros-García, A. I., & Fraga, J. (2018). Cytologic Features of Malignant Melanoma with Osteoclast-Like Giant Cells. Acta Cytologica, 62, 151–154
Houang, M., Castillo, C., La Marca, S., Combemale, P., Wang, Q., Paindavoine, S. … de la Fouchardiere A. (2015). An unusual case of desmoplastic melanoma containing an osteoclast-like giant cell-rich nodule. The American Journal of Dermatopathology, 37, 299–304
Nakahashi, H., Tsuneyoshi, M., Ishida, T., Minagawa, S., Owaki, Y., Momii, S., & Eimoto, T. (1987). Undifferentiated carcinoma of the lung with osteoclast-like giant cells. The Japanese Journal of Surgery, 17, 199–203
Matsukuma, S., Takeo, H., Kato, K., & Sato, K. (2014). Numerous osteoclast-like giant cells in metastases from lung adenocarcinoma, but absent from primary tumor. Thoracic Cancer, 5, 354–357
Lindholm, K. E., Kalhor, N., & Moran, C. A. (2019). Osteoclast-like giant cell-rich carcinomas of the lung: a clinicopathological, immunohistochemical, and molecular study of 3 cases. Human Pathology, 85, 168–173
Kong, L., Peng, W., Liu, J., Wang, W., Gong, P., Yu, G., & Li, J. (2015). Squamous cell carcinoma of lung associated with osteoclast-like giant cells: report of a case. International Journal of Clinical and Experimental Pathology, 8, 11823–11825
Dahm, H. H. (2017). Non-small cell carcinoma of the lung with osteoclast-like giant cells. International Journal of Surgical Pathology, 25, 258–261
Stewart, C. J., & Mutch, A. F. (1991). Breast carcinoma with osteoclast-like giant cells. Cytopathology, 2, 215–219
Agnantis, N. T., & Rosen, P. P. (1979). Mammary carcinoma with osteoclast-like giant cells. A study of eight cases with follow-up data. American Journal of Clinical Pathology, 72, 383–389
Fadare, O., & Gill, S. A. (2009). Solid neuroendocrine carcinoma of the breast with osteoclast-like giant cells. The Breast Journal, 15, 205–206
Ginter, P. S., Petrova, K., & Hoda, S. A. (2015). The grossly “rusty” tumor of breast: invasive ductal carcinoma with osteoclast-like giant cells. International Journal of Surgical Pathology, 23, 32–33
Ohashi, R., Hayama, A., Matsubara, M., Watarai, Y., Sakatani, T., Naito, Z., & Shimizu, A. (2018). Breast carcinoma with osteoclast-like giant cells: A cytological-pathological correlation with a literature review. Annals of Diagnostic Pathology, 33, 1–5
Hoorweg, J. J., Loftus, B. M., & Hilgers, F. J. (1997). Osteoid and bone formation in a nasal mucosal melanoma and its metastasis. Histopathology, 31, 465–468
Dekkers, I. A., Cleven, A., Lamb, H. J., & Kroon, H. M. (2019). Primary Osteosarcoma of the Breast. Radiographics, 39, 626–629
Drummond, C. J., Hanna, J. A., Garcia, M. R., Devine, D. J., Heyrana, A. J., Finkelstein, D. … Hatley, M. E. (2018). Hedgehog Pathway Drives Fusion-Negative Rhabdomyosarcoma Initiated From Non-myogenic Endothelial Progenitors. Cancer Cell, 33, 108–1245
Rubin, H. (1985). Cancer as a dynamic developmental disorder. Cancer Research, 45, 2935–2942
Solter, D. (2006). From teratocarcinomas to embryonic stem cells and beyond: a history of embryonic stem cell research. Nature Reviews. Genetics, 7, 319–327
Knoepfler, P. S. (2009). Deconstructing stem cell tumorigenicity: a roadmap to safe regenerative medicine. Stem Cells, 27, 1050–1056
Riggs, J. W., Barrilleaux, B. L., Varlakhanova, N., Bush, K. M., Chan, V., & Knoepfler, P. S. (2013). Induced pluripotency and oncogenic transformation are related processes. Stem Cells and Development, 22, 37–50
Cheng, L., Hu, W., Qiu, B., Zhao, J., Yu, Y., Guan, W. … Pei, G. (2014). Generation of neural progenitor cells by chemical cocktails and hypoxia. Cell Research, 24, 665–679
Domazet-Loso, T., Brajković, J., & Tautz, D. (2007). A phylostratigraphy approach to uncover the genomic history of major adaptations in metazoan lineages. Trends in Genetics, 23, 533–539
Sebé-Pedrós, A., Degnan, B. M., & Ruiz-Trillo, I. (2017). The origin of Metazoa: a unicellular perspective. Nature Reviews. Genetics, 18, 498–512
Clarke, L., & van der Kooy, D. (2009). Low oxygen enhances primitive and definitive neural stem cell colony formation by inhibiting distinct cell death pathways. Stem Cells, 27, 1879–1886
Vernon, A. E., & Philpott, A. (2003). The developmental expression of cell cycle regulators in Xenopus laevis. Gene Expression Patterns, 3, 179–192
Hatch, V. L., Marin-Barba, M., Moxon, S., Ford, C. T., Ward, N. J., Tomlinson, M. L. … Wheeler, G. N. (2016). The positive transcriptional elongation factor (P-TEFb) is required for neural crest specification. Developmental Biology, 416, 361–372
Baserga, R. (2007). Is cell size important? Cell Cycle, 6, 814–816
Turi, Z., Lacey, M., Mistrik, M., & Moudry, P. (2019). Impaired ribosome biogenesis: mechanisms and relevance to cancer and aging. Aging, 11, 2512–2540
Robson, A., Owens, N. D., Baserga, S. J., Khokha, M. K., & Griffin, J. N. (2016). Expression of ribosomopathy genes during Xenopus tropicalis embryogenesis. BMC Developmental Biology, 16, 38
Altmann, C. R., Bell, E., Sczyrba, A., Pun, J., Bekiranov, S., Gaasterland, T., & Brivanlou, A. H. (2001). Microarray-based analysis of early development in Xenopus laevis. Developmental Biology, 236, 64–75
Weinstein, D. C., Honoré, E., & Hemmati-Brivanlou, A. (1997). Epidermal induction and inhibition of neural fate by translation initiation factor 4AIII. Development, 124, 4235–4242
Yan, B., Neilson, K. M., & Moody, S. A. (2010). Microarray identification of novel downstream targets of FoxD4L1/D5, a critical component of the neural ectodermal transcriptional network. Developmental Dynamics, 239, 3467–3480
Chau, K. F., Shannon, M. L., Fame, R. M., Fonseca, E., Mullan, H., Johnson, M. B. … Lehtinen, M. K. (2018). Downregulation of ribosome biogenesis during early forebrain development. eLife, 7, e36998
Hasegawa, K., Shiraishi, T., & Kinoshita, T. (1999). Xoom: a novel oocyte membrane protein maternally expressed and involved in the gastrulation movement of Xenopus embryos. The International Journal of Developmental Biology, 43, 479–485
Parain, K., Mazurier, N., Bronchain, O., Borday, C., Cabochette, P., Chesneau, A. … Perron, M. (2012). A large scale screen for neural stem cell markers in Xenopus retina. Developmental Neurobiology, 72, 491–506
Hu, R., Huang, W., Liu, J., Jin, M., Wu, Y., Li, J. … Cao, Y. (2019). Mutagenesis of putative ciliary genes with the CRISPR/Cas9 system in zebrafish identifies genes required for retinal development. FASEB Journal, 33, 5248–5256
Bush, S. J., Chen, L., Tovar-Corona, J. M., & Urrutia, A. O. (2017). Alternative splicing and the evolution of phenotypic novelty. Philosophical transactions of the Royal Society of London. Series B, Biological sciences, 372, 20150474
Baralle, F. E., & Giudice, J. (2017). Alternative splicing as a regulator of development and tissue identity. Nature Reviews. Molecular Cell Biology, 18, 437–451
Dichmann, D. S., Fletcher, R. B., & Harland, R. M. (2008). Expression cloning in Xenopus identifies RNA-binding proteins as regulators of embryogenesis and Rbmx as necessary for neural and muscle development. Developmental Dynamics, 237, 1755–1766
Voigt, J., Chen, J. A., Gilchrist, M., Amaya, E., & Papalopulu, N. (2005). Expression cloning screening of a unique and full-length set of cDNA clones is an efficient method for identifying genes involved in Xenopus neurogenesis. Mechanisms of Development, 122, 289–306
Liu, K. J., & Harland, R. M. (2005). Inhibition of neurogenesis by SRp38, a neuroD-regulated RNA-binding protein. Development, 132, 1511–1523
Li, C., Hu, R., Hou, N., Wang, Y., Wang, Z., Yang, T. … Li, T. (2018). Alteration of the Retinoid Acid-CBP Signaling Pathway in Neural Crest Induction Contributes to Enteric Nervous System Disorder. Frontiers in Pediatrics, 6, 382
Schwenty-Lara, J., Nürnberger, A., & Borchers, A. (2019). Loss of function of Kmt2d, a gene mutated in Kabuki syndrome, affects heart development in Xenopus laevis. Developmental Dynamics, 248, 465–476
Nicetto, D., Hahn, M., Jung, J., Schneider, T. D., Straub, T., David, R. … Rupp, R. A. (2013). Suv4-20 h histone methyltransferases promote neuroectodermal differentiation by silencing the pluripotency-associated Oct-25 gene. PLoS Genetics, 9, e1003188
Saritas-Yildirim, B., Childers, C. P., Elsik, C. G., & Silva, E. M. (2015). Identification of REST targets in the Xenopus tropicalis genome. BMC Genomics, 16, 380
Kawaguchi, A., Ochi, H., Sudou, N., & Ogino, H. (2012). Comparative expression analysis of the H3K27 demethylases, JMJD3 and UTX, with the H3K27 methylase, EZH2, in Xenopus. The International Journal of Developmental Biology, 56, 295–300
Wang, C. D., Guo, X. F., Wong, T., Wang, H., Qi, X. F., Cai, D. Q. … Zhao, H. (2019). Developmental expression of three prmt genes in Xenopus. Zoological Research, 40, 102–107
Zhang, X., Gao, Y., Lu, L., Zhang, Z., Gan, S., Xu, L. … Cao, Y. (2015). JmjC domain-containing protein 6 (Jmjd6) derepresses the transcriptional repressor transcription factor 7-like 1 (Tcf7l1) and is required for body axis patterning during Xenopus embryogenesis. The Journal of Biological Chemistry, 290, 20273–20283
Xu, Y., Zhang, M., Li, W., Zhu, X., Bao, X., Qin, B. … Esteban, M. A. (2016). Transcriptional control of somatic cell reprogramming. Trends in Cell Biology, 26, 272–288
Buitrago-Delgado, E., Nordin, K., Rao, A., Geary, L., & LaBonne, C. (2015). NEURODEVELOPMENT. Shared regulatory programs suggest retention of blastula-stage potential in neural crest cells. Science, 348, 1332–1335
Bellmeyer, A., Krase, J., Lindgren, J., & LaBonne, C. (2003). The protooncogene c-myc is an essential regulator of neural crest formation in xenopus. Developmental Cell, 4, 827–839
Janesick, A., Abbey, R., Chung, C., Liu, S., Taketani, M., & Blumberg, B. (2013). ERF and ETV3L are retinoic acid-inducible repressors required for primary neurogenesis. Development, 140, 3095–3106
Thisse, B., & Thisse, C. (2004). Fast release clones: a high throughput expression analysis. ZFIN Direct Data Submission (http://zfin.org)
Light, W., Vernon, A. E., Lasorella, A., Iavarone, A., & LaBonne, C. (2005). Xenopus Id3 is required downstream of Myc for the formation of multipotent neural crest progenitor cells. Development, 132, 1831–1841
Neff, A. W., King, M. W., Harty, M. W., Nguyen, T., Calley, J., Smith, R. C., & Mescher, A. L. (2005). Expression of Xenopus XlSALL4 during limb development and regeneration. Developmental Dynamics, 233, 356–367
Reijnen, M. J., Hamer, K. M., den Blaauwen, J. L., Lambrechts, C., Schoneveld, I., van Driel, R., & Otte, A. P. (1995). Polycomb and bmi-1 homologs are expressed in overlapping patterns in Xenopus embryos and are able to interact with each other. Mechanisms of Development, 53, 35–46
Li, I. C., Chan, C. T., Lu, Y. F., Wu, Y. T., Chen, Y. C., Li, G. B. … Hwang, S. P. (2011). Zebrafish krüppel-like factor 4a represses intestinal cell proliferation and promotes differentiation of intestinal cell lineages. PloS One, 6, e20974
Bertrand, S., Thisse, B., Tavares, R., Sachs, L., Chaumot, A., Bardet, P. L. … Laudet, V. (2007). Unexpected novel relational links uncovered by extensive developmental profiling of nuclear receptor expression. PLoS Genetics, 3, e188
Seo, S., Richardson, G. A., & Kroll, K. L. (2005). The SWI/SNF chromatin remodeling protein Brg1 is required for vertebrate neurogenesis and mediates transactivation of Ngn and NeuroD. Development, 132, 105–115
Tessmar, K., Loosli, F., & Wittbrodt, J. (2002). A screen for co-factors of Six3. Mechanisms of Development, 117, 103–113
Iwano, H., Nakamura, M., & Tajima, S. (2004). Xenopus MBD3 plays a crucial role in an early stage of development. Developmental Biology, 268, 416–428
Aldiri, I., & Vetter, M. L. (2009). Characterization of the expression pattern of the PRC2 core subunit Suz12 during embryonic development of Xenopus laevis. Developmental Dynamics, 238, 3185–3192
Bibonne, A., Néant, I., Batut, J., Leclerc, C., Moreau, M., & Gilbert, T. (2013). Three calcium-sensitive genes, fus, brd3 and wdr5, are highly expressed in neural and renal territories during amphibian development. Biochimica et Biophysica Acta, 1833, 1665–1671
Sen, R., Pezoa, S. A., Carpio Shull, L., Hernandez-Lagunas, L., Niswander, L. A., & Artinger, K. B. (2018). Kat2a and Kat2b Acetyltransferase Activity Regulates Craniofacial Cartilage and Bone Differentiation in Zebrafish and Mice. Journal of Developmental Biology, 6, 27
Wang, Y., Shupenko, C. C., Melo, L. F., & Strauss, P. R. (2006). DNA repair protein involved in heart and blood development. Molecular and Cellular Biology, 26, 9083–9093
Pan, Y., Kelly, L. E., & El-Hodiri, H. M. (2018). Identification of retinal homeobox (rax) gene-dependent genes by a microarray approach: The DNA endoglycosylase neil3 is a major downstream component of the rax genetic pathway. Developmental Dynamics, 247, 1199–1210
Gu, A., Ji, G., Yan, L., & Zhou, Y. (2013). The 8-oxoguanine DNA glycosylase 1 (ogg1) decreases the vulnerability of the developing brain to DNA damage. DNA Repair, 12, 1094–1104
Bustelo, X. R., & Dosil, M. (2018). Ribosome biogenesis and cancer: basic and translational challenges. Current Opinion in Genetics & Development, 48, 22–29
Catez, F., Dalla Venezia, N., Marcel, V., Zorbas, C., Lafontaine, D., & Diaz, J. J. (2019). Ribosome biogenesis: An emerging druggable pathway for cancer therapeutics. Biochemical Pharmacology, 159, 74–81
Pelletier, J., Thomas, G., & Volarević, S. (2018). Ribosome biogenesis in cancer: new players and therapeutic avenues. Nature Reviews. Cancer, 18, 51–63
Bhat, M., Robichaud, N., Hulea, L., Sonenberg, N., Pelletier, J., & Topisirovic, I. (2015). Targeting the translation machinery in cancer. Nature Reviews. Drug Discovery, 14, 261–278
Chen, Y., Zhang, Y., & Guo, X. (2017). Proteasome dysregulation in human cancer: implications for clinical therapies. Cancer Metastasis Reviews, 36, 703–716
Soave, C. L., Guerin, T., Liu, J., & Dou, Q. P. (2017). Targeting the ubiquitin-proteasome system for cancer treatment: discovering novel inhibitors from nature and drug repurposing. Cancer Metastasis Reviews, 36, 717–736
Rousseau, A., & Bertolotti, A. (2018). Regulation of proteasome assembly and activity in health and disease. Nature Reviews. Molecular Cell Biology, 19, 697–712
Dvinge, H., Kim, E., Abdel-Wahab, O., & Bradley, R. K. (2016). RNA splicing factors as oncoproteins and tumour suppressors. Nature Reviews. Cancer, 16, 413–430
Wang, B. D., & Lee, N. H. (2018). Aberrant RNA splicing in cancer and drug resistance. Cancers, 10, 458
Bennett, R. L., & Licht, J. D. (2018). Targeting epigenetics in cancer. Annual Review of Pharmacology and Toxicology, 58, 187–207
Mohammad, H. P., Barbash, O., & Creasy, C. L. (2019). Targeting epigenetic modifications in cancer therapy: erasing the roadmap to cancer. Nature Medicine, 25, 403–418
Brown, J. S., O’Carrigan, B., Jackson, S. P., & Yap, T. A. (2017). Targeting DNA repair in cancer: beyond PARP inhibitors. Cancer Discovery, 7, 20–37
Klinakis, A., Karagiannis, D., & Rampias, T. (2020). Targeting DNA repair in cancer: current state and novel approaches. Cellular and Molecular Life Sciences, 77, 677–703
Gabel, H. W., Kinde, B., Stroud, H., Gilbert, C. S., Harmin, D. A., Kastan, N. R. … Greenberg, M. E. (2015). Disruption of DNA-methylation-dependent long gene repression in Rett syndrome. Nature, 522, 89–93
Zylka, M. J., Simon, J. M., & Philpot, B. D. (2015). Gene length matters in neurons. Neuron, 86, 353–355
Domazet-Loso, T., & Tautz, D. (2010). Phylostratigraphic tracking of cancer genes suggests a link to the emergence of multicellularity in metazoa. BMC Biology, 8, 66
Bussey, K. J., Cisneros, L. H., Lineweaver, C. H., & Davies, P. (2017). Ancestral gene regulatory networks drive cancer. Proceedings of the National Academy of Sciences of the United States of America, 114, 6160–6162
Trigos, A. S., Pearson, R. B., Papenfuss, A. T., & Goode, D. L. (2017). Altered interactions between unicellular and multicellular genes drive hallmarks of transformation in a diverse range of solid tumors. Proceedings of the National Academy of Sciences of the United States of America, 114, 6406–6411
Trigos, A. S., Pearson, R. B., Papenfuss, A. T., & Goode, D. L. (2018). How the evolution of multicellularity set the stage for cancer. British Journal of Cancer, 118, 145–152
Anatskaya, O. V., Vinogradov, A. E., Vainshelbaum, N. M., Giuliani, A., & Erenpreisa, J. (2020). Phylostratic Shift of Whole-Genome Duplications in Normal Mammalian Tissues towards Unicellularity Is Driven by Developmental Bivalent Genes and Reveals a Link to Cancer. International Journal of Molecular Sciences, 21, 8759
Alfarouk, K. O., Shayoub, M. E., Muddathir, A. K., Elhassan, G. O., & Bashir, A. H. (2011). Evolution of Tumor Metabolism might Reflect Carcinogenesis as a Reverse Evolution process (Dismantling of Multicellularity). Cancers, 3, 3002–3017
Chen, H., Lin, F., Xing, K., & He, X. (2015). The reverse evolution from multicellularity to unicellularity during carcinogenesis. Nature Communications, 6, 6367
Kim, D. Y., Rhee, I., & Paik, J. (2014). Metabolic circuits in neural stem cells. Cellular and Molecular Life Sciences, 71, 4221–4241
Zheng, X., Boyer, L., Jin, M., Mertens, J., Kim, Y., Ma, L. … Hunter, T. (2016). Metabolic reprogramming during neuronal differentiation from aerobic glycolysis to neuronal oxidative phosphorylation. eLife, 5, e13374
Intlekofer, A. M., & Finley, L. (2019). Metabolic signatures of cancer cells and stem cells. Nature Metabolism, 1, 177–188
Nishimura, K., Fukuda, A., & Hisatake, K. (2019). Mechanisms of the Metabolic Shift during Somatic Cell Reprogramming. International Journal of Molecular Sciences, 20, 2254
Rabelo, I., Arnaud-Sampaio, V. F., Adinolfi, E., Ulrich, H., & Lameu, C. (2021). Cancer metabostemness and metabolic reprogramming via P2 × 7 receptor. Cells, 10, 1782
Ribeiro, D. E., Glaser, T., Oliveira-Giacomelli, Ã., & Ulrich, H. (2019). Purinergic receptors in neurogenic processes. Brain Research Bulletin, 151, 3–11
Oliveira, Ã., Illes, P., & Ulrich, H. (2016). Purinergic receptors in embryonic and adult neurogenesis. Neuropharmacology, 104, 272–281
Yona, A. H., Manor, Y. S., Herbst, R. H., Romano, G. H., Mitchell, A., Kupiec, M. … Dahan, O. (2012). Chromosomal duplication is a transient evolutionary solution to stress. Proceedings of the National Academy of Sciences of the United States of America, 109, 21010–21015
Cisneros, L., Bussey, K. J., Orr, A. J., Miočević, M., Lineweaver, C. H., & Davies, P. (2017). Ancient genes establish stress-induced mutation as a hallmark of cancer. PloS One, 12, e0176258
Varela, C., Denis, J. A., Polentes, J., Feyeux, M., Aubert, S., Champon, B. … Lefort, N. (2012). Recurrent genomic instability of chromosome 1q in neural derivatives of human embryonic stem cells. The Journal of Clinical Investigation, 122, 569–574
Peterson, S. E., & Loring, J. F. (2014). Genomic instability in pluripotent stem cells: implications for clinical applications. The Journal of Biological Chemistry, 289, 4578–4584
Sahakyan, A. B., & Balasubramanian, S. (2016). Long genes and genes with multiple splice variants are enriched in pathways linked to cancer and other multigenic diseases. BMC Genomics, 17, 225
Sogabe, S., Hatleberg, W. L., Kocot, K. M., Say, T. E., Stoupin, D., Roper, K. E. … Degnan, B. M. (2019). Pluripotency and the origin of animal multicellularity. Nature, 570, 519–522
Cao, Y., Siegel, D., & Knöchel, W. (2006). Xenopus POU factors of subclass V inhibit activin/nodal signaling during gastrulation. Mechanisms of Development, 123, 614–625
Snir, M., Ofir, R., Elias, S., & Frank, D. (2006). Xenopus laevis POU91 protein, an Oct3/4 homologue, regulates competence transitions from mesoderm to neural cell fates. The EMBO Journal, 25, 3664–3674
Lujan, E., & Wernig, M. (2012). The many roads to Rome: induction of neural precursor cells from fibroblasts. Current Opinion in Genetics & Development, 22, 517–522
Shyamala, K., Yanduri, S., Girish, H. C., & Murgod, S. (2015). Neural crest: The fourth germ layer. Journal of Oral and Maxillofacial Pathology: JOMFP, 19, 221–229
Ozair, M. Z., Kintner, C., & Brivanlou, A. H. (2013). Neural induction and early patterning in vertebrates. Wiley Interdisciplinary Reviews. Developmental Biology, 2, 479–498
Itoh, F., Watabe, T., & Miyazono, K. (2014). Roles of TGF-β family signals in the fate determination of pluripotent stem cells. Seminars in Cell & Developmental Biology, 32, 98–106
Meyers, E. A., & Kessler, J. A. (2017). TGF-β family signaling in neural and neuronal differentiation, development, and function. Cold Spring Harbor Perspectives in Biology, 9, a022244
Sancho, M., & Rodríguez, T. A. (2013). Ready, set, differentiate!. eLife, 2, e01839
Gomes Fernandes, M., Dries, R., Roost, M. S., Semrau, S., de Melo Bernardo, A., Davis, R. P. … de Lopes, S., A., &, S. M. (2016). BMP-SMAD Signaling Regulates Lineage Priming, but Is Dispensable for Self-Renewal in Mouse Embryonic Stem Cells. Stem Cell Reports, 6, 85–94
Nichols, S. A., Dirks, W., Pearse, J. S., & King, N. (2006). Early evolution of animal cell signaling and adhesion genes. Proceedings of the National Academy of Sciences of the United States of America, 103, 12451–12456
King, N., Westbrook, M. J., Young, S. L., Kuo, A., Abedin, M., Chapman, J. … Rokhsar, D. (2008). The genome of the choanoflagellate Monosiga brevicollis and the origin of metazoans. Nature, 451(7180), 783–788
Srivastava, M., Simakov, O., Chapman, J., Fahey, B., Gauthier, M. E., Mitros, T. … Rokhsar, D. S. (2010). The Amphimedon queenslandica genome and the evolution of animal complexity. Nature, 466(7307), 720–726
Adamska, M., Degnan, S. M., Green, K. M., Adamski, M., Craigie, A., Larroux, C., & Degnan, B. M. (2007). Wnt and TGF-beta expression in the sponge Amphimedon queenslandica and the origin of metazoan embryonic patterning. PloS One, 2, e1031
Barker, N. (2014). Adult intestinal stem cells: critical drivers of epithelial homeostasis and regeneration. Nature Reviews. Molecular Cell Biology, 15, 19–33
Li, L., Mignone, J., Yang, M., Matic, M., Penman, S., Enikolopov, G., & Hoffman, R. M. (2003). Nestin expression in hair follicle sheath progenitor cells. Proceedings of the National Academy of Sciences of the United States of America, 100, 9958–9961
Jiang, M. H., Cai, B., Tuo, Y., Wang, J., Zang, Z. J., Tu, X. … Xiang, A. P. (2014). Characterization of Nestin-positive stem Leydig cells as a potential source for the treatment of testicular Leydig cell dysfunction. Cell Research, 24, 1466–1485
Mokrý, J., Cízková, D., Filip, S., Ehrmann, J., Osterreicher, J., Kolár, Z., & English, D. (2004). Nestin expression by newly formed human blood vessels. Stem Cells and Development, 13, 658–664
Hagey, D. W., Klum, S., Kurtsdotter, I., Zaouter, C., Topcic, D., Andersson, O. … Muhr, J. (2018). SOX2 regulates common and specific stem cell features in the CNS and endoderm derived organs. PLoS Genetics, 14, e1007224
Casanova, J. (2012). Stemness as a cell default state. EMBO Reports, 13, 396–397
Pesaresi, M., Sebastian-Perez, R., & Cosma, M. P. (2019). Dedifferentiation, transdifferentiation and cell fusion: in vivo reprogramming strategies for regenerative medicine. The FEBS Journal, 286, 1074–1093
Kienle, G., & Kiene, H. (2012). From Reductionism to Holism: Systems-oriented Approaches in Cancer Research. Global Advances in Health and Medicine, 1, 68–77
Jopling, C., Boue, S., & Izpisua Belmonte, J. C. (2011). Dedifferentiation, transdifferentiation and reprogramming: three routes to regeneration. Nature Reviews. Molecular Cell Biology, 12, 79–89
Jumabay, M., & Boström, K. I. (2015). Dedifferentiated fat cells: A cell source for regenerative medicine. World Journal of Stem Cells, 7, 1202–1214
Lim, B., Lin, Y., & Navin, N. (2020). Advancing cancer research and medicine with single-cell genomics. Cancer Cell, 37, 456–470
Weinberg, R. A. (2014). Coming full circle-from endless complexity to simplicity and back again. Cell, 157, 267–271
Holohan, C., Van Schaeybroeck, S., Longley, D. B., & Johnston, P. G. (2013). Cancer drug resistance: an evolving paradigm. Nature Reviews. Cancer, 13, 714–726
Ramos, P., & Bentires-Alj, M. (2015). Mechanism-based cancer therapy: resistance to therapy, therapy for resistance. Oncogene, 34, 3617–3626
Lytle, N. K., Barber, A. G., & Reya, T. (2018). Stem cell fate in cancer growth, progression and therapy resistance. Nature Reviews. Cancer, 18, 669–680
Sharma, P., Hu-Lieskovan, S., Wargo, J. A., & Ribas, A. (2017). Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell, 168, 707–723
Syn, N. L., Teng, M., Mok, T., & Soo, R. A. (2017). De-novo and acquired resistance to immune checkpoint targeting. The Lancet Oncology, 18, e731–e741
de Miguel, M., & Calvo, E. (2020). Clinical challenges of immune checkpoint inhibitors. Cancer Cell, 38, 326–333
Frelaut, M., Le Tourneau, C., & Borcoman, E. (2019). Hyperprogression under Immunotherapy. International Journal of Molecular Sciences, 20, 2674
Adashek, J. J., Subbiah, I. M., Matos, I., Garralda, E., Menta, A. K., Ganeshan, D. M., & Subbiah, V. (2020). Hyperprogression and immunotherapy: fact, fiction, or alternative fact? Trends in Cancer, 6, 181–191
Krah, N. M., Narayanan, S. M., Yugawa, D. E., Straley, J. A., Wright, C., MacDonald, R. J., & Murtaugh, L. C. (2019). Prevention and reversion of pancreatic tumorigenesis through a differentiation-based mechanism. Developmental Cell, 50, 744–7544
Ishay-Ronen, D., Diepenbruck, M., Kalathur, R., Sugiyama, N., Tiede, S., Ivanek, R., ... Christofori, G. (2019). Gain fat-lose metastasis: converting invasive breast cancer cells into adipocytes inhibits cancer metastasis. Cancer Cell, 35, 17–326
Funding
This work was supported by Shenzhen Science and Technology Program (Grant No.: JCYJ20210324120205015), and National Science Foundation of China (Grant No.: 31671499).
Author information
Authors and Affiliations
Contributions
Ying Cao conceived and wrote the review.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing Interests
The author declares no potential competing interests.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, Y. Neural is Fundamental: Neural Stemness as the Ground State of Cell Tumorigenicity and Differentiation Potential. Stem Cell Rev and Rep 18, 37–55 (2022). https://doi.org/10.1007/s12015-021-10275-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-021-10275-y