Abstract
Purpose of Review
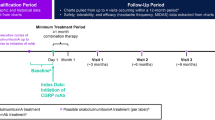
Monoclonal antibodies against calcitonin gene-related peptide (CGRP) or its receptor have become part of the standard treatment for migraine in clinical practice. The current review focuses on the clinical evidence of CGRP monoclonal antibodies in patients with chronic migraine (CM), including more challenging cases.
Recent Findings
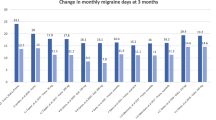
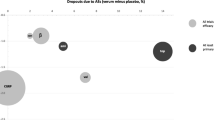
CGRP monoclonal antibodies were more effective than placebo in reducing the number of monthly migraine days (MMDs), and the change relative to placebo in the treatment group was between − 1.2 and − 2.7 days at 3 months. CGRP monoclonal antibodies resulted in ≥ 50% response in 27.5 to 61.4% of patients, and doubled the odds for having ≥ 50% response. The findings were generally consistent in patients with coexisting medication overuse or with treatment failures to multiple preventive medications, including onabotulinumtoxinA. The results from real-world studies (RWS) were similar to those seen in clinical trials, and the changes from baseline in the number of MMDs and the response rates largely fell within the ranges of those reported in the treatment group in pivotal trials. The therapeutic effects typically started within a few days, and remained steady after regular treatment for up to 1 year. These agents were generally well tolerated, and the discontinuation rates due to adverse events in clinical trials and in many RWS were < 4.5%.
Summary
CGRP monoclonal antibodies are effective and safe in the treatment of patients with CM, including clinical challenging cases. However, the role of CGRP monoclonal antibodies in a number of conditions, such as cardiovascular or cerebrovascular diseases, pregnancy, and overuse of opioids or barbiturates, needs to be further clarified.

Similar content being viewed by others
References
May A, Schulte LH. Chronic migraine: risk factors, mechanisms and treatment. Nat Rev Neurol. 2016;12(8):455–64.
Stark RJ, Ravishankar K, Siow HC, Lee KS, Pepperle R, Wang SJ. Chronic migraine and chronic daily headache in the Asia-Pacific region: a systematic review. Cephalalgia. 2013;33(4):266–83.
Lu SR, Fuh JL, Chen WT, Juang KD, Wang SJ. Chronic daily headache in Taipei, Taiwan: prevalence, follow-up and outcome predictors. Cephalalgia. 2001;21(10):980–6.
Wang SJ, Fuh JL, Lu SR, Liu CY, Hsu LC, Wang PN, et al. Chronic daily headache in Chinese elderly: prevalence, risk factors, and biannual follow-up. Neurology. 2000;54(2):314–9.
Buse DC, Manack AN, Fanning KM, Serrano D, Reed ML, Turkel CC, et al. Chronic migraine prevalence, disability, and sociodemographic factors: results from the American Migraine Prevalence and Prevention Study. Headache. 2012;52(10):1456–70.
Buse D, Manack A, Serrano D, Reed M, Varon S, Turkel C, et al. Headache impact of chronic and episodic migraine: results from the American Migraine Prevalence and Prevention study. Headache. 2012;52(1):3–17.
Juang KD, Wang SJ, Fuh JL, Lu SR, Su TP. Comorbidity of depressive and anxiety disorders in chronic daily headache and its subtypes. Headache. 2000;40(10):818–23.
Bigal ME, Serrano D, Buse D, Scher A, Stewart WF, Lipton RB. Acute migraine medications and evolution from episodic to chronic migraine: a longitudinal population-based study. Headache. 2008;48(8):1157–68.
Scher AI, Stewart WF, Ricci JA, Lipton RB. Factors associated with the onset and remission of chronic daily headache in a population-based study. Pain. 2003;106(1–2):81–9.
Goadsby PJ, Edvinsson L, Ekman R. Vasoactive peptide release in the extracerebral circulation of humans during migraine headache. Ann Neurol. 1990;28(2):183–7.
Goadsby PJ, Edvinsson L. The trigeminovascular system and migraine: studies characterizing cerebrovascular and neuropeptide changes seen in humans and cats. Ann Neurol. 1993;33(1):48–56.
Edvinsson L, Haanes KA, Warfvinge K, Krause DN. CGRP as the target of new migraine therapies - successful translation from bench to clinic. Nat Rev Neurol. 2018;14(6):338–50.
Olesen J, Diener HC, Husstedt IW, Goadsby PJ, Hall D, Meier U, et al. Calcitonin gene-related peptide receptor antagonist BIBN 4096 BS for the acute treatment of migraine. N Engl J Med. 2004;350(11):1104–10.
Ho TW, Mannix LK, Fan X, Assaid C, Furtek C, Jones CJ, et al. Randomized controlled trial of an oral CGRP receptor antagonist, MK-0974, in acute treatment of migraine. Neurology. 2008;70(16):1304–12.
Ho TW, Ferrari MD, Dodick DW, Galet V, Kost J, Fan X, et al. Efficacy and tolerability of MK-0974 (telcagepant), a new oral antagonist of calcitonin gene-related peptide receptor, compared with zolmitriptan for acute migraine: a randomised, placebo-controlled, parallel-treatment trial. Lancet. 2008;372(9656):2115–23.
Ho TW, Connor KM, Zhang Y, Pearlman E, Koppenhaver J, Fan X, et al. Randomized controlled trial of the CGRP receptor antagonist telcagepant for migraine prevention. Neurology. 2014;83(11):958–66.
Ho TW, Ho AP, Ge YJ, Assaid C, Gottwald R, MacGregor EA, et al. Randomized controlled trial of the CGRP receptor antagonist telcagepant for prevention of headache in women with perimenstrual migraine. Cephalalgia. 2016;36(2):148–61.
Agostoni EC, Barbanti P, Calabresi P, Colombo B, Cortelli P, Frediani F, et al. Current and emerging evidence-based treatment options in chronic migraine: a narrative review. J Headache Pain. 2019;20(1):92.
Yang CP, Zeng BY, Chang CM, Shih PH, Yang CC, Tseng PT, et al. Comparative effectiveness and tolerability of the pharmacology of monoclonal antibodies targeting the calcitonin gene-related peptide and its receptor for the prevention of chronic migraine: a network meta-analysis of randomized controlled trials. Neurotherapeutics. 2021;18:2639–50.
Bigal ME, Edvinsson L, Rapoport AM, Lipton RB, Spierings EL, Diener HC, et al. Safety, tolerability, and efficacy of TEV-48125 for preventive treatment of chronic migraine: a multicentre, randomised, double-blind, placebo-controlled, phase 2b study. Lancet Neurol. 2015;14(11):1091–100.
Dodick DW, Lipton RB, Silberstein S, Goadsby PJ, Biondi D, Hirman J, et al. Eptinezumab for prevention of chronic migraine: a randomized phase 2b clinical trial. Cephalalgia. 2019;39(9):1075–85.
Detke HC, Goadsby PJ, Wang S, Friedman DI, Selzler KJ, Aurora SK. Galcanezumab in chronic migraine: the randomized, double-blind, placebo-controlled REGAIN study. Neurology. 2018;91(24):e2211–21.
Tepper S, Ashina M, Reuter U, Brandes JL, Dolezil D, Silberstein S, et al. Safety and efficacy of erenumab for preventive treatment of chronic migraine: a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Neurol. 2017;16(6):425–34.
Silberstein SD, Dodick DW, Bigal ME, Yeung PP, Goadsby PJ, Blankenbiller T, et al. Fremanezumab for the preventive treatment of chronic migraine. N Engl J Med. 2017;377(22):2113–22.
Sakai F, Suzuki N, Kim BK, Igarashi H, Hirata K, Takeshima T, et al. Efficacy and safety of fremanezumab for chronic migraine prevention: multicenter, randomized, double-blind, placebo-controlled, parallel-group trial in Japanese and Korean patients. Headache. 2021;61(7):1092–101.
Lipton RB, Goadsby PJ, Smith J, Schaeffler BA, Biondi DM, Hirman J, et al. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology. 2020;94(13):e1365–77.
Jhingran P, Osterhaus JT, Miller DW, Lee JT, Kirchdoerfer L. Development and validation of the Migraine-Specific Quality of Life Questionnaire. Headache. 1998;38(4):295–302.
Yang M, Rendas-Baum R, Varon SF, Kosinski M. Validation of the Headache Impact Test (HIT-6) across episodic and chronic migraine. Cephalalgia. 2011;31(3):357–67.
Stewart WF, Lipton RB, Kolodner K, Liberman J, Sawyer J. Reliability of the migraine disability assessment score in a population-based sample of headache sufferers. Cephalalgia. 1999;19(2):107–14; discussion 74.
Lipton RB, Tepper SJ, Reuter U, Silberstein S, Stewart WF, Nilsen J, et al. Erenumab in chronic migraine: patient-reported outcomes in a randomized double-blind study. Neurology. 2019;92(19):e2250–60.
Chiang CC, Schwedt TJ, Wang SJ, Dodick DW. Treatment of medication-overuse headache: a systematic review. Cephalalgia. 2016;36(4):371–86.
Silberstein SD, Lipton RB, Dodick DW, Freitag FG, Ramadan N, Mathew N, et al. Efficacy and safety of topiramate for the treatment of chronic migraine: a randomized, double-blind, placebo-controlled trial. Headache. 2007;47(2):170–80.
Diener HC, Bussone G, Van Oene JC, Lahaye M, Schwalen S, Goadsby PJ, et al. Topiramate reduces headache days in chronic migraine: a randomized, double-blind, placebo-controlled study. Cephalalgia. 2007;27(7):814–23.
Aurora SK, Dodick DW, Turkel CC, DeGryse RE, Silberstein SD, Lipton RB, et al. OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 1 trial. Cephalalgia. 2010;30(7):793–803.
Diener HC, Dodick DW, Aurora SK, Turkel CC, DeGryse RE, Lipton RB, et al. OnabotulinumtoxinA for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia. 2010;30(7):804–14.
Silberstein SD, Blumenfeld AM, Cady RK, Turner IM, Lipton RB, Diener HC, et al. OnabotulinumtoxinA for treatment of chronic migraine: PREEMPT 24-week pooled subgroup analysis of patients who had acute headache medication overuse at baseline. J Neurol Sci. 2013;331(1–2):48–56.
Wang YF. OnabotulinumtoxinA injection in the treatment of chronic migraine. Prog Brain Res. 2020;255:171–206.
Diener HC, Dodick DW, Goadsby PJ, Bigal ME, Bussone G, Silberstein SD, et al. Utility of topiramate for the treatment of patients with chronic migraine in the presence or absence of acute medication overuse. Cephalalgia. 2009;29(10):1021–7.
Dodick DW, Doty EG, Aurora SK, Ruff DD, Stauffer VL, Jedynak J, et al. Medication overuse in a subgroup analysis of phase 3 placebo-controlled studies of galcanezumab in the prevention of episodic and chronic migraine. Cephalalgia. 2021;41(3):340–52.
Tepper SJ, Diener HC, Ashina M, Brandes JL, Friedman DI, Reuter U, et al. Erenumab in chronic migraine with medication overuse: Subgroup analysis of a randomized trial. Neurology. 2019;92(20):e2309–20.
Silberstein SD, Cohen JM, Seminerio MJ, Yang R, Ashina S, Katsarava Z. The impact of fremanezumab on medication overuse in patients with chronic migraine: subgroup analysis of the HALO CM study. J Headache Pain. 2020;21(1):114.
Diener HC, Marmura MJ, Tepper SJ, Cowan R, Starling AJ, Diamond ML, et al. Efficacy, tolerability, and safety of eptinezumab in patients with a dual diagnosis of chronic migraine and medication-overuse headache: subgroup analysis of PROMISE-2. Headache. 2021;61(1):125–36.
Headache Classification Committee of the International Headache Society. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33(9):629–808.
Ferrari MD, Diener HC, Ning X, Galic M, Cohen JM, Yang R, et al. Fremanezumab versus placebo for migraine prevention in patients with documented failure to up to four migraine preventive medication classes (FOCUS): a randomised, double-blind, placebo-controlled, phase 3b trial. Lancet. 2019;394(10203):1030–40.
Mulleners WM, Kim BK, Lainez MJA, Lanteri-Minet M, Pozo-Rosich P, Wang S, et al. Safety and efficacy of galcanezumab in patients for whom previous migraine preventive medication from two to four categories had failed (CONQUER): a multicentre, randomised, double-blind, placebo-controlled, phase 3b trial. Lancet Neurol. 2020;19(10):814–25.
Lambru G, Hill B, Murphy M, Tylova I, Andreou AP. A prospective real-world analysis of erenumab in refractory chronic migraine. J Headache Pain. 2020;21(1):61.
Scheffler A, Messel O, Wurthmann S, Nsaka M, Kleinschnitz C, Glas M, et al. Erenumab in highly therapy-refractory migraine patients: First German real-world evidence. J Headache Pain. 2020;21(1):84.
Torres-Ferrus M, Gallardo VJ, Alpuente A, Caronna E, Gine-Cipres E, Pozo-Rosich P. The impact of anti-CGRP monoclonal antibodies in resistant migraine patients: a real-world evidence observational study. J Neurol. 2021;268(10):3789–98.
Bottiroli S, De Icco R, Vaghi G, Pazzi S, Guaschino E, Allena M, et al. Psychological predictors of negative treatment outcome with erenumab in chronic migraine: data from an open label long-term prospective study. J Headache Pain. 2021;22(1):114.
de Vries LS, Verhagen IE, van den Hoek TC, MaassenVanDenBrink A, Terwindt GM. Treatment with the monoclonal calcitonin gene-related peptide receptor antibody erenumab: a real-life study. Eur J Neurol. 2021;28(12):4194–203.
Pensato U, Favoni V, Pascazio A, Benini M, Asioli GM, Merli E, et al. Erenumab efficacy in highly resistant chronic migraine: a real-life study. Neurol Sci. 2020;41(Suppl 2):457–9.
Alex A, Vaughn C, Rayhill M. Safety and tolerability of 3 CGRP monoclonal antibodies in practice: a retrospective cohort study. Headache. 2020;60(10):2454–62.
Caronna E, Gallardo VJ, Alpuente A, Torres-Ferrus M, Pozo-Rosich P. Anti-CGRP monoclonal antibodies in chronic migraine with medication overuse: real-life effectiveness and predictors of response at 6 months. J Headache Pain. 2021;22(1):120.
Vernieri F, Altamura C, Brunelli N, Costa CM, Aurilia C, Egeo G, et al. Galcanezumab for the prevention of high frequency episodic and chronic migraine in real life in Italy: a multicenter prospective cohort study (the GARLIT study). J Headache Pain. 2021;22(1):35.
Ailani J, Pearlman E, Zhang Q, Nagy AJ, Schuh K, Aurora SK. Positive response to galcanezumab following treatment failure to onabotulinumtoxinA in patients with migraine: post hoc analyses of three randomized double-blind studies. Eur J Neurol. 2020;27(3):542–9.
Alpuente A, Gallardo VJ, Caronna E, Torres-Ferrus M, Pozo-Rosich P. Partial and nonresponders to onabotulinumtoxinA can benefit from anti-CGRP monoclonal antibodies preventive treatment: a real-world evidence study. Eur J Neurol. 2021;28(7):2378–82.
Talbot J, Stuckey R, Crawford L, Weatherby S, Mullin S. Improvements in pain, medication use and quality of life in onabotulinumtoxinA-resistant chronic migraine patients following erenumab treatment - real world outcomes. J Headache Pain. 2021;22(1):5.
Pellesi L, Do TP, Ashina H, Ashina M, Burstein R. Dual therapy with Anti-CGRP monoclonal antibodies and botulinum toxin for migraine prevention: is there a rationale? Headache. 2020;60(6):1056–65.
Armanious M, Khalil N, Lu Y, Jimenez-Sanders R. Erenumab and onabotulinumtoxinA combination therapy for the prevention of intractable chronic migraine without aura: a retrospective analysis. J Pain Palliat Care Pharmacother. 2021;35(1):1–6.
Blumenfeld AM, Frishberg BM, Schim JD, Iannone A, Schneider G, Yedigarova L, et al. Real-world evidence for control of chronic migraine patients receiving CGRP monoclonal antibody therapy added to onabotulinumtoxinA: a retrospective chart review. Pain Ther. 2021;10(2):809–26.
Winner PK, Spierings ELH, Yeung PP, Aycardi E, Blankenbiller T, Grozinski-Wolff M, et al. Early onset of efficacy with fremanezumab for the preventive treatment of chronic migraine. Headache. 2019;59(10):1743–52.
Schwedt T, Reuter U, Tepper S, Ashina M, Kudrow D, Broessner G, et al. Early onset of efficacy with erenumab in patients with episodic and chronic migraine. J Headache Pain. 2018;19(1):92.
Tepper SJ, Ashina M, Reuter U, Brandes JL, Dolezil D, Silberstein SD, et al. Long-term safety and efficacy of erenumab in patients with chronic migraine: results from a 52-week, open-label extension study. Cephalalgia. 2020;40(6):543–53.
Detke H, Pozo-Rosisch P, Reuter U, Dolezil D, Li LQ, Wang S, Aurora SK. One-year treatment with galcanezumab in patients with chronic migraine: results from the open-label phase of the REGAIN Study (P2.10–010). Neurology. 2019;92:2.10–010.
Goadsby PJ, Silberstein SD, Yeung PP, Cohen JM, Ning X, Yang R, et al. Long-term safety, tolerability, and efficacy of fremanezumab in migraine: a randomized study. Neurology. 2020;95(18):e2487–99.
Blumenfeld AM, Stevanovic DM, Ortega M, Cohen JM, Seminerio MJ, Yang R, et al. No “wearing-off effect” seen in quarterly or monthly dosing of fremanezumab: subanalysis of a randomized long-term study. Headache. 2020;60(10):2431–43.
Sacco S, Bendtsen L, Ashina M, Reuter U, Terwindt G, Mitsikostas DD, et al. European headache federation guideline on the use of monoclonal antibodies acting on the calcitonin gene related peptide or its receptor for migraine prevention. J Headache Pain. 2019;20(1):6.
Raffaelli B, Mussetto V, Israel H, Neeb L, Reuter U. Erenumab and galcanezumab in chronic migraine prevention: effects after treatment termination. J Headache Pain. 2019;20(1):66.
Gantenbein AR, Agosti R, Gobbi C, Flugel D, Schankin CJ, Viceic D, et al. Impact on monthly migraine days of discontinuing anti-CGRP antibodies after one year of treatment - a real-life cohort study. Cephalalgia. 2021;41(11–12):1181–6.
De Matteis E, Affaitati G, Frattale I, Caponnetto V, Pistoia F, Giamberardino MA, et al. Early outcomes of migraine after erenumab discontinuation: data from a real-life setting. Neurol Sci. 2021;42(8):3297–303.
Raffaelli B, Terhart M, Overeem LH, Mecklenburg J, Neeb L, Steinicke M, et al. Migraine evolution after the cessation of CGRP(-receptor) antibody prophylaxis: a prospective, longitudinal cohort study. Cephalalgia. 2021:3331024211046617.
Vandervorst F, Van Deun L, Van Dycke A, Paemeleire K, Reuter U, Schoenen J, et al. CGRP monoclonal antibodies in migraine: an efficacy and tolerability comparison with standard prophylactic drugs. J Headache Pain. 2021;22(1):128.
Raffaelli B, Kalantzis R, Mecklenburg J, Overeem LH, Neeb L, Gendolla A, et al. Erenumab in chronic migraine patients who previously failed five first-line oral prophylactics and onabotulinumtoxinA: a dual-center retrospective observational study. Front Neurol. 2020;11:417.
Ashina M, Kudrow D, Reuter U, Dolezil D, Silberstein S, Tepper SJ, et al. Long-term tolerability and nonvascular safety of erenumab, a novel calcitonin gene-related peptide receptor antagonist for prevention of migraine: a pooled analysis of four placebo-controlled trials with long-term extensions. Cephalalgia. 2019;39(14):1798–808.
Saely S, Croteau D, Jawidzik L, Brinker A, Kortepeter C. Hypertension: a new safety risk for patients treated with erenumab. Headache. 2021;61(1):202–8.
Dodick DW, Tepper SJ, Ailani J, Pannacciulli N, Navetta MS, Loop B, et al. Risk of hypertension in erenumab-treated patients with migraine: analyses of clinical trial and postmarketing data. Headache. 2021;61(9):1411–20.
Yang CP, Lee CF, Dell’Agnello G, Hundemer HP, Lipsius S, Wang SJ. Safety and efficacy of galcanezumab in Taiwanese patients: a post-hoc analysis of phase 3 studies in episodic and chronic migraine. Curr Med Res Opin. 2020;36(10):1653–66.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
This review was sponsored in part by Taiwan Ministry of Science and Technology [MOST 109–2314-B-075 -054 and MOST 110–2314-B-075 -041 -MY3 (to YFW), and MOST 104–2314-B-010–015-MY2, MOST106-2321-B-010–009, MOST 107–2321-B-010–001, MOST 108–2321-B-010–014 -MY2, 108–2321-B-010 001, MOST 108–2314-B-010–023-MY3, and MOST-110–2321-B-010–005 (to SJW)]; and Taipei Veterans General Hospital [VGH 108-C-092, VGH 109-C-096, and VGH 110-C-111 (to YFW)]; this work was also supported by the Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
YFW has received honoraria as a speaker from Taiwan branches of Allergan/AbbVie, Eli Lilly, Novartis, Pfizer, Sanofi, UCB, and Viatris, and Orient EuroPharma. He has received research grants from the Taiwan Ministry of Science and Technology, and Taipei Veterans General Hospital. SJW has served on the advisory boards of Daiichi-Sankyo, Eli Lilly and Novartis; has received honoraria as a moderator from Allergan/AbbVie, Pfizer, Eli Lilly, Biogen and Eisai and has been the PI in trials sponsored by Eli Lilly, Novartis, and Allergan/AbbVie. He has received research grants from the Taiwan Minister of Technology and Science (MOST), Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan, Taipei Veterans General Hospital, Taiwan Headache Society and Taiwan branches of Eli Lilly, Novartis, and Pfizer.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Chronic Daily Headache
Rights and permissions
About this article
Cite this article
Wang, YF., Wang, SJ. CGRP Targeting Therapy for Chronic Migraine—Evidence from Clinical Trials and Real-world Studies. Curr Pain Headache Rep 26, 543–554 (2022). https://doi.org/10.1007/s11916-022-01056-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11916-022-01056-4