Abstract
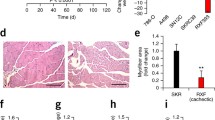
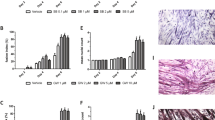
Conjugated linoleic acid (CLA) is thought to have anti-proliferative and anti-inflammatory properties, but its effect on cancer cachexia is unknown. Two effects were here investigated: that of CLA on inflammatory mediator production in human lung cancer cells, and that of reduced mediators on the myogenic differentiation of murine muscle C2C12 cells. The latter cells were grown in medium conditioned by human lung cancer A427 cells, with or without CLA, to mimic only the effect of molecules released from the tumor “in vivo”, excluding the effect of host-produced cachectic factors. The results obtained show that CLA was found to reduce the production of tumor necrosis factor-α, interleukin (IL)-1β and prostaglandin E2 (PGE2), but had no effect on IL-6 production. The mechanisms underlying the effect of CLA on cytokine or PGE2 release in A427 cells are probably mediated by activation of peroxisome proliferator-activated receptor (PPAR)α, which increased at 24 h CLA treatment. In turn, the reduced content of inflammatory mediators in medium conditioned by A427 cells, in the presence of CLA, allowed muscle cells to proliferate, again by inducing PPAR. The involvement of PPARα was demonstrated by treatment with the antagonist MK-886. The findings demonstrate the anti-inflammatory and myogenic action of CLA and point to its possible application as a novel dietary supplement and therapeutic agent in inflammatory disease states, such as cachexia.







Similar content being viewed by others
Abbreviations
- CLA:
-
Conjugated linoleic acid
- COX:
-
Cyclooxygenase
- IL:
-
Interleukin
- LDH:
-
Lactate dehydrogenase
- PGE2:
-
Prostaglandin E2
- PPAR:
-
Peroxisome proliferator-activated receptor
- TNF:
-
Tumour necrosis factor
References
Costelli P, Baccino FM (2000) Cancer cachexia: from experimental models to patient management. Curr Opin Clin Nutr Metab Care 3:177–181
Fearon K, Moses A (2002) Cancer cachexia. Int J Cardiol 85:73–81
Argiles JM, Busquets S, López-Soriano FJ (2005) The pivotal role of cytokines in muscle wasting during cancer. Int J Biochem Cell Biol 37:2036–2046
Tisdale MJ (1997) Cancer cachexia: metabolic alterations and clinical manifestations. Nutrition 13:1–7
Hawke TJ, Garry DJ (2001) Myogenic satellite cells: physiology and molecular biology. J Appl Physiol 91:534–551
Argiles JM, Busquets S, Toledo M, López-Soriano FJ (2009) The role of cytokines in cancer cachexia. Curr Opin Support Palliat Care 3:263–268
Kemik O, Sumer A, Kemik AS, Itik V, Dulger AC, Purisa S, Tuzun S (2010) The relationship among acute-phase response proteins, cytokines and hormones in cachectic patients with colon cancer. World J Surg Oncol 28:8–85
Strassmann G, Fong M, Freter CE, Windsor S, D’Alessandro F, Nordan RP (1993) Suramin interfers with interleukin-6 receptor binding in vitro and inhibits colon-26-mediated experimental cancer cachexia in vivo. J Clin Invest 92:2152–2159
Enomoto A, Rho MC, Fukami A, Hiraku O, Komiyama K, Hayashi M (2004) Suppression of cancer cachexia by 20S, 21-epoxy-resibufogenin-3-acetate-a novel nonpeptide IL-6 receptor antagonist. Biochem Biophys Res Commun 323:1096–1102
Garcıa-Martınez C, Lopez-Soriano FJ, Argiles JM (1994) Interleukin-6 does not activate protein breakdown in rat skeletal muscle. Cancer Lett 76:1–4
Zhang D, Zheng H, Zhou Y, Tang X, Yu B, Li J (2007) Association of IL-1beta gene polymorphism with cachexia from locally advanced gastric cancer. BMC Cancer 14:7–45
Tisdale MJ (2001) Cancer anorexia and cachexia. Nutrition 17:438–442
Bhattacharya A, Banu J, Rahman M, Causey J, Fernandes G (2006) Biological effects of conjugated linoleic acids in health and disease. J Nutr Biochem 17:789–810
MacDonald HB (2000) Conjugated linoleic acid and disease prevention: a review of current knowledge. J Am Coll Nutr 19:111–118
Belury M (2002) Dietary conjugated linoleic acid in health: physiological effects and mechanisms of action. Annu Rev Nutr 22:505–531
Pariza MW, Park Y, Cook ME (2001) The biologically active isomers of conjugated linoleic acid. Prog Lipid Res 40:283–298
De la Torre A, Debiton E, Durand D, Chardigny JM, Berdeaux O, Loreau O, Barthomeuf C, Bauchart D, Gruffat D (2005) Conjugated linoleic acid isomers and their conjugated derivatives inhibit growth of human cancer cell lines. Anticancer Res 25:3943–3949
Pariza MW, Park Y, Cook ME (1999) Conjugated linoleic acid and the control of cancer and obesity. Toxicol Sci 52:107–110
Pariza MW, Park Y, Cook ME (2000) Mechanisms of action of conjugated linoleic acid: evidence and speculation. Proc Soc Exp Biol Med 223:8–13
Kelley NS, Hubbard NE, Erickson KL (2007) Conjugated linoleic acid isomers and cancer. J Nutr 137:2599–2607
Maggiora M, Bologna M, Cerù MP, Possati L, Angelucci A, Cimini A, Miglietta A, Bozzo F, Margiotta C, Muzio G, Canuto RA (2004) An overview of the effect of linoleic and conjugated-linoleic acids on the growth of several human tumor cell lines. Int J Cancer 112:909–919
Zulet MA, Marti A, Parra MD, Martínez JA (2005) Inflammation and conjugated linoleic acid: mechanisms of action and implications for human health. J Physiol Biochem 61:483–494
Gonçalves DC, Lira FS, Carnevali LC Jr, Rosa JC, Pimentel GD, Seelaender M (2010) Conjugated linoleic acid: good or bad nutrient. Diabetol Metab Syndr 30:2–62
Tian M, Kliewer KL, Asp ML, Stout MB, Belury MA (2011) c9t11-Conjugated linoleic acid-rich oil fails to attenuate wasting in colon-26 tumor-induced late-stage cancer cachexia in male CD2F1 mice. Mol Nutr Food Res 55:268–277
Graves E, Hitt A, Pariza MW, Cook ME, McCarthy DO (2005) Conjugated linoleic acid preserves gastrocnemius muscle mass in mice bearing the colon-26 adenocarcinoma. Res Nurs Health 28:48–55
McCarthy DO, Graves E (2006) Conjugated linoleic acid preserves muscle mass in mice bearing the Lewis lung carcinoma, but not the B16 melanoma. Res Nurs Health 29:98–104
Larsen AE, Crowe TC (2009) Effects of conjugated linoleic acid on myogenic and inflammatory responses in a human primary muscle and tumor coculture model. Nutr Cancer 61:687–695
Martinasso G, Maggiora M, Trombetta A, Canuto RA, Muzio G (2006) Effects of di(2-ethylhexyl) phthalate, a widely used peroxisome proliferator and plasticizer, on cell growth in the human keratinocyte cell line NCTC 2544. J Toxicol Environ Health A 69:353–365
Li G, Barnes D, Butz D, Bjorling D, Cook ME (2005) 10t,12c-conjugated linoleic acid inhibits lipopolysaccharide-induced cyclooxygenase expression in vitro and in vivo. J Lipid Res 46:2134–2142
Lee JH, Tachibana H, Morinaga Y, Fujimura Y, Yamada K (2009) Modulation of proliferation and differentiation of C2C12 skeletal muscle cells by fatty acids. Life Sci 84:415–420
Tisdale MJ (2009) Mechanisms of cancer cachexia. Physiol Rev 89:381–410
Alvarez B, Quinn LS, Busquets S, López-Soriano FJ, Argilés JM (2002) TNF-alpha modulates cytokine and cytokine receptors in C2C12 myotubes. Cancer Lett 175:181–185
Bishop-Bailey D, Bystrom J (2009) Emerging roles of peroxisome proliferator-activated receptor-beta/delta in inflammation. Pharmacol Ther 124:141–150
Cuzzocrea S, Pisano B, Dugo L, Ianaro A, Maffia P, Patel NS, Di Paola R, Ialenti A, Genovese T, Chatterjee PK, Di Rosa M, Caputi AP, Thiemermann C (2004) Rosiglitazone, a ligand of the peroxisome proliferator-activated receptor-γ, reduces acute inflammation. Eur J Pharmacol 483:79–93
Chen HH, Chen TW, Lin H (2009) Prostacyclin-induced peroxisome proliferator-activated receptor-alpha translocation attenuates NF-kappaB and TNF-alpha activation after renal ischemia-reperfusion injury. Am J Physiol Renal Physiol 297:1109–1118
Liang CJ, Tseng CP, Yang CM, Ma YH (2011) 20-Hydroxyeicosatetraenoic acid inhibits ATP-induced COX-2 expression via peroxisome proliferator activator receptor-α in vascular smooth muscle cells. Br J Pharmacol 163:815–825
Ramanan S, Kooshki M, Zhao W, Hsu FC, Robbins ME (2008) PPARalpha ligands inhibit radiation-induced microglial inflammatory responses by negatively regulating NF-kappaB and AP-1 pathways. Free Radic Biol Med 45:1695–1704
Delerive P, De Bosscher K, Besnard S, Vanden Berghe W, Peters JM, Gonzalez FJ, Fruchart JC, Tedgui A, Haegeman G, Staels B (1999) Peroxisome proliferator-activated receptor alpha negatively regulates the vascular inflammatory gene response by negative cross-talk with transcription factors NF-kappaB and AP-1. J Biol Chem 274:32048–32054
Jana M, Jana A, Liu X, Ghosh S, Pahan K (2007) Involvement of phosphatidylinositol 3-kinase-mediated up-regulation of I kappa B alpha in anti-inflammatory effect of gemfibrozil in microglia. J Immunol 179:4142–4152
Delerive P, Gervois P, Fruchart JC, Staels B (2000) Induction of IkappaBalpha expression as a mechanism contributing to the anti-inflammatory activities of peroxisome proliferator-activated receptor-alpha activators. J Biol Chem 275:36703–36707
Wysong A, Couch M, Shadfar S, Li L, Rodriguez JE, Asher S, Yin X, Gore M, Baldwin A, Patterson C, Willis MS (2011) NF-κB inhibition protects against tumor-induced cardiac atrophy in vivo. Am J Pathol 178:1059–1068
Zambon A, Gervois P, Pauletto P, Fruchart JC, Staels B (2006) Modulation of hepatic inflammatory risk markers of cardiovascular diseases by PPAR-alpha activators: clinical and experimental evidence. Arterioscler Thromb Vasc Biol 26:977–986
Giordano C, Rousseau AS, Wagner N, Gaudel C, Murdaca J, Jehl-Piétri C, Sibille B, Grimaldi PA, Lopez P (2009) Peroxisome proliferator-activated receptor beta activation promotes myonuclear accretion in skeletal muscle of adult and aged mice. Pflugers Arch 458:901–913
Allen DL, Monke SR, Talmadge RJ, Roy RR, Edgerton VR (1995) Plasticity of myonuclear number in hypertrophied and atrophied mammalian skeletal muscle fibers. J Appl Physiol 78:1969–1976
Roy RR, Monke SR, Allen DL, Edgerton VR (1999) Modulation of myonuclear number in functionally overloaded and exercised rat plantaris fibers. J Appl Physiol 87:634–642
Li P, Akimoto T, Zhang M, Williams RS, Yan Z (2006) Resident stem cells are not required for exercise-induced fiber-type switching and angiogenesis but are necessary for activity-dependent muscle growth. Am J Physiol Cell Physiol 290:1461–1468
Acknowledgments
This research was supported by grants from Compagnia di San Paolo, Italy, from Regione Piemonte, Italy and from the University of Turin, Italy. We thank Frances Cooper for revising the English and Prof. Sebastiano Colombatto for technical support.
Conflict of interest
All authors have read and agreed to the editorial policies, and declare that there are no financial or other conflicts of interest that might be construed to influence the results or interpretation of their manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Oraldi, M., Maggiora, M., Paiuzzi, E. et al. CLA Reduces Inflammatory Mediators from A427 Human Lung Cancer Cells and A427 Conditioned Medium Promotes Differentiation of C2C12 Murine Muscle Cells. Lipids 48, 29–38 (2013). https://doi.org/10.1007/s11745-012-3734-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-012-3734-6