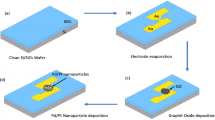
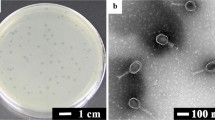
Abstract
Vaccinia is caused by the vaccinia virus and affects thousands of people, particularly in underprivileged communities, in over 100 countries worldwide. In Brazil, more than 10,000 cases are registered between January and June 2023, with some resulting in fatalities. This study investigates the use of pencil graphite electrodes (PGEs) modified with polymeric films from 2-hydroxybenzamide (2HB) and 4-hydroxybenzamide (4HB) to develop a biosensor to diagnose vaccinia virus (VACV) infection. Polymeric films were produced by electropolymerization of the respective monomers using cyclic voltammetry. The modified electrodes showed a reduced response to the potassium ferro/ferricyanide solution and increased charge transfer resistance (Rct). Two recognition strategies have been proposed: immobilizing VACV for detecting anti-VACV antibodies and immobilizing anti-VACV for virus detection. In the detection of anti-VACV, Rct increased by approximately 162% for PGE/poly(2HB) and 55% for PGE/poly(4HB), with no significant changes observed for non-specific antibodies. For VACV detection, the Rct response increased by 30 and 190% in the presence of the viral particles for PGE/poly(2HB) and PGE/poly(4HB), respectively. The biosensors displayed a significant increase in Rct response in the presence of viral particles, demonstrating their potential for detecting viral antigens or antibodies. PGE/poly(2HB) and PGE/poly(4HB) were found to be suitable for detecting anti-VACV and the vaccinia virus itself, respectively, using the applied method. These findings highlight the versatility of the biosensor for a wide range of applications, including diagnostic tests and vaccine quality control, and emphasize its potential for VACV diagnosis. A biosensor that detects both vaccinia antigens and viruses can aid in monitoring immune responses, diagnosing infections, ensuring vaccine safety, and tracking outbreaks.
Graphical abstract








Similar content being viewed by others
References
Abrahão JS, Guedes MIM, Trindade GS et al (2009) One more piece in the VACV ecological puzzle: could peridomestic rodents be the link between wildlife and bovine vaccinia outbreaks in Brazil? PLoS ONE 4:e7428. https://doi.org/10.1371/JOURNAL.PONE.0007428
Ahmadi M, Ahour F (2020) An electrochemical biosensor based on a graphene oxide modified pencil graphite electrode for direct detection and discrimination of double-stranded DNA sequences. Anal Methods 12:4541–4550. https://doi.org/10.1039/D0AY01128B
Alcamí A (2023) Pathogenesis of the circulating mpox virus and its adaptation to humans. Proc Natl Acad Sci USA 120:e2301662120. https://doi.org/10.1073/PNAS.2301662120/ASSET/861AC145-DD1C-4ADC-97D2-347F7D4929B6/ASSETS/IMAGES/LARGE/PNAS.2301662120FIG01.JPG
Barouki R, Kogevinas M, Audouze K et al (2021) The COVID-19 pandemic and global environmental change: emerging research needs. Environ Int 146:106272. https://doi.org/10.1016/J.ENVINT.2020.106272
Blum LJ, Coulet PR (1991) Biosensor principles and applications, 1a. M. Dekker
Borremans B, Faust C, Manlove KR et al (2019) Cross-species pathogen spillover across ecosystem boundaries: mechanisms and theory. Philos Trans R Soc B. https://doi.org/10.1098/RSTB.2018.0344
Buleandra M, Rabinca AA, Badea IA et al (2017) (2017) Voltammetric determination of dihydroxybenzene isomers using a disposable pencil graphite electrode modified with cobalt-phthalocyanine. Microchim Acta 1845(184):1481–1488. https://doi.org/10.1007/S00604-017-2153-Z
Bunge EM, Hoet B, Chen L et al (2022) The changing epidemiology of human monkeypox—a potential threat? A systematic review. PLoS Negl Trop Dis 16:e0010141. https://doi.org/10.1371/JOURNAL.PNTD.0010141
Burek DG (1993) Biosensors: theory and applications. Florida, Boca Raton
Instituto Butantan (2020) Como surgiu o novo coronavírus? Conheça as teorias mais aceitas sobre sua origem
Chomel BB (2009) Zoonoses. Encycl Microbiol. https://doi.org/10.1016/B978-012373944-5.00213-3
Cohen JI, Hohman P, Preuss JC et al (2007) Detection of vaccinia virus DNA, but not infectious virus, in the blood of smallpox vaccine recipients. Vaccine 25:4571–4574. https://doi.org/10.1016/j.vaccine.2007.03.044
Cordeiro TAR, Martins HR, Franco DL et al (2020) Impedimetric immunosensor for rapid and simultaneous detection of chagas and visceral leishmaniasis for point of care diagnosis. Biosens Bioelectron 169:112573. https://doi.org/10.1016/j.bios.2020.112573
Cordeiro TAR, de Resende MAC, dos Moraes SC, S, et al (2021) Electrochemical biosensors for neglected tropical diseases: a review. Talanta 234:122617. https://doi.org/10.1016/j.talanta.2021.122617
da Cruz Santos C, Pimenta TC, Thomasini RL, Verly RM, Franco DL, Ferreira LF (2019) Electropolymerization of phenol and aniline derivatives: synthesis, characterization and application as electrochemical transducers. J Electroanal Chem 846:113163. https://doi.org/10.1016/j.jelechem.2019.05.045
da Cruz Santos C, Santos PCM, Rocha KLS, Thomasini RL, de Oliveira DB, Franco DL, Ferreira LF (2020) A new tool for dengue virus diagnosis: optimization and detection of anti-NS1 antibodies in serum samples by impedimetric transducers. Microchem J 154:104544. https://doi.org/10.1016/j.microc.2019.104544
Davison AJ, Moss B (1989) Structure of vaccinia virus early promoters. J Mol Biol 210:749–769. https://doi.org/10.1016/0022-2836(89)90107-1
de Fátima Alves M, de Souza Corrêa RAM, da Cruz FS, Franco DL, Ferreira LF (2018) Electrochemical enzymatic fenitrothion sensor based on a tyrosinase/poly (2-hydroxybenzamide)-modified graphite electrode. Analytical Biochem 553:15–23. https://doi.org/10.1016/j.ab.2018.05.014
Dedavid BA, Gomes CI, Machado G (2007) Microscopia eletrônica de varredura microscopia eletrônica de varredura aplicações e preparação de amostras aplicações e preparação de amostras. Pontifícia Univ Católica do Rio Gd do Sul
Donaldson KA, Kramer MF, Lim DV (2004) A rapid detection method for Vaccinia virus, the surrogate for smallpox virus. Biosens Bioelectron 20:322–327. https://doi.org/10.1016/j.bios.2004.01.029
Drumond BP, Leite JA, da Fonseca FG et al (2008) Brazilian Vaccinia virus strains are genetically divergent and differ from the Lister vaccine strain. Microbes Infect 10:185–197. https://doi.org/10.1016/J.MICINF.2007.11.005
Ebrahimiasl S, Fathi E, Ahmad M (2018) Electrochemical detection of insulin in blood serum using Ppy/GF nanocomposite modified pencil graphite electrode. Nanomed Res J 3(4):219–228
Ensafi AA, Kazemnadi N, Amini M, Rezaei B (2015) Impedimetric DNA-biosensor for the study of dopamine induces DNA damage and investigation of inhibitory and repair effects of some antioxidants. Bioelectrochemistry 104:71–78. https://doi.org/10.1016/j.bioelechem.2015.03.008
Erdem A, Karadeniz H, Caliskan A (2011) Dendrimer modified graphite sensors for detection of anticancer drug Daunorubicin by voltammetry and electrochemical impedance spectroscopy. Analyst 136:1041–1045. https://doi.org/10.1039/C0AN00357C
Evtugyn G (2014) Biosensors: essentials, 1a. Springer, Heidelberg New York, Kazan, Rússia
Fiocruz FOC (2010) Pesquisadores explicam a atual importância do vírus vaccinia
Fundação Nacional de Saúde F (2002) Guia de Vigilância Epidemiológica, 1st edn. Brasília
Goebel SJ, Johnson GP, Perkus ME et al (1990) The complete DNA sequence of vaccinia virus. Virology 179:247–266. https://doi.org/10.1016/0042-6822(90)90294-2
Gong ZQ, Sujari ANA, Ab Ghani S (2012) Electrochemical fabrication, characterization and application of carboxylic multi-walled carbon nanotube modified composite pencil graphite electrodes. Electrochim Acta 65:257–265. https://doi.org/10.1016/J.ELECTACTA.2012.01.057
Goud KY, Reddy KK, Khorshed A et al (2021) Electrochemical diagnostics of infectious viral diseases: trends and challenges. Biosens Bioelectron 180:113112. https://doi.org/10.1016/j.bios.2021.113112
Gubser C, Hué S, Kellam P, Smith GL (2004) Poxvirus genomes: a phylogenetic analysis. J Gen Virol 85:105–117. https://doi.org/10.1099/VIR.0.19565-0
Güner A, Çevik E, Şenel M, Alpsoy L (2017) An electrochemical immunosensor for sensitive detection of Escherichia coli O157:H7 by using chitosan, MWCNT, polypyrrole with gold nanoparticles hybrid sensing platform. Food Chem 229:358–365. https://doi.org/10.1016/J.FOODCHEM.2017.02.083
Harrison SC, Alberts B, Ehrenfeld E et al (2004) Discovery of antivirals against smallpox. Proc Natl Acad Sci U S A 101:11178–11192. https://doi.org/10.1073/PNAS.0403600101
Jones KE, Patel NG, Levy MA et al (2008) (2008) Global trends in emerging infectious diseases. Nat 4517181(451):990–993. https://doi.org/10.1038/nature06536
Karimi-Maleh H, Tahernejad-Javazmi F, Atar N et al (2015) A novel DNA biosensor based on a pencil graphite electrode modified with polypyrrole/functionalized multiwalled carbon nanotubes for determination of 6-mercaptopurine anticancer drug. Ind Eng Chem Res 54:3634–3639. https://doi.org/10.1021/IE504438Z
Labib M, Zamay AS, Muharemagic D et al (2012) Electrochemical sensing of aptamer-facilitated virus immunoshielding. Anal Chem 84:1677–1686. https://doi.org/10.1021/ac202978r
Lasia A (2011) Electrochemical impedance spectroscopy EIS, and corrosion, 1a. Springer, New York, Sherbrooke, Québec
Leik NKO, Ahmedy F, Mac GR, Baharuddin DMP (2021) Covid-19 vaccine and its consequences in pregnancy: brief review. Ann Med Surg 72:103103. https://doi.org/10.1016/J.AMSU.2021.103103
Lu T, Wu Z, Jiang S et al (2022) The current emergence of monkeypox: the recurrence of another smallpox? Biosaf Heal 4:369–375. https://doi.org/10.1016/j.bsheal.2022.09.004
Luques MN, Oliveira RL, Hir S et al (2023) Co-circulation of vaccinia and monkeypox viruses in rural areas of Brazil: importance of differential molecular diagnosis. Travel Med Infect Dis 53:102578. https://doi.org/10.1016/j.tmaid.2023.102578
Magee WC, Shahhosseini S, Lin YCJ et al (2009) Production and characterization of antibodies against vaccinia virus DNA polymerase. J Virol Methods 161:44–51. https://doi.org/10.1016/J.JVIROMET.2009.05.012
Martin C da S (2013) Estudo e caracterização da eletropolimerização de moléculas nanoestruturadas a Base de Schiff. Universidade Estadual Paulista “Júlio de Mesquita Filho”
Matho MH, Schlossman A, Meng X et al (2015) Structural and functional characterization of anti-A33 antibodies reveal a potent cross-species orthopoxviruses neutralizer. PLOS Pathog 11:e1005148. https://doi.org/10.1371/JOURNAL.PPAT.1005148
Matho MH, Schlossman A, Gilchuk IM et al (2018) Structure-function characterization of three human antibodies targeting the vaccinia virus adhesion molecule D8. J Biol Chem 293:390–401. https://doi.org/10.1074/JBC.M117.814541
Ministério da Saúde (2005) Guia de Vigilância Epidemiológica, 6th edn. MINISTÉRIO DA SAÚDE, Brasília - DF
Morens DM, Fauci AS (2020) Emerging pandemic diseases: how we got to COVID-19. Cell 182:1077–1092. https://doi.org/10.1016/J.CELL.2020.08.021
Moussatche N, Condit RC (2015) Fine structure of the vaccinia virion determined by controlled degradation and immunolocalization. Virology 475:204–218. https://doi.org/10.1016/J.VIROL.2014.11.020
Oliveira N, Souza E, Ferreira D et al (2015) A sensitive and selective label-free electrochemical DNA biosensor for the detection of specific dengue virus serotype 3 sequences. Sensors 15:15562–15577. https://doi.org/10.3390/s150715562
Oliveira JSD, Figueiredo PDO, Costa GB, Assis FLD, Drumond BP, Da Fonseca FG, Trindade GDS (2017) Vaccinia virus natural infections in brazil: the good, the bad, and the ugly. Viruses 9(11):340. https://doi.org/10.3390/V9110340
Ozcan HM, Aydin UD (2021) A simple immunosensor for thyroid stimulating hormone. Artif Cells Nanomed Biotechnol 49:61–70. https://doi.org/10.1080/21691401.2020.1867153
Prasad BB, Madhuri R, Tiwari MP, Sharma PS (2010) Imprinting molecular recognition sites on multiwalled carbon nanotubes surface for electrochemical detection of insulin in real samples. Electrochim Acta 55:9146–9156. https://doi.org/10.1016/j.electacta.2010.09.008
Qasim Almajidi Y, Algahtani SM, Sajjad Alsawad O et al (2023) Recent applications of microfluidic immunosensors. Microchem J 190:108733. https://doi.org/10.1016/J.MICROC.2023.108733
Rahman MT, Sobur MA, Islam MS et al (2020) Zoonotic diseases: etiology, impact, and control. Microorganisms 8:1–34. https://doi.org/10.3390/MICROORGANISMS8091405
Rai M, Reshetilov A, Plekhanova Y, Ingle AP (2021) Macro, micro, and nano-biosensors : potential applications and possible limitations, 1a. Springer
Reghunath R, Devi K, Singh KK (2021) Recent advances in graphene based electrochemical glucose sensor. Nano-Struct Nano-Objects 26:100750. https://doi.org/10.1016/j.nanoso.2021.100750
Rehfeld IS, Guedes MIMC, Fraiha ALS et al (2015) Vaccinia virus transmission through experimentally contaminated milk using a murine model. PLoS ONE 10:e0127350. https://doi.org/10.1371/JOURNAL.PONE.0127350
Santos PCM, Lima TM, Soares PI et al (2022) Electropolymerization in multilayers of aromatic monomers over graphite electrodes for the development of a biosensor for Chagas disease. Mater Chem Phys 288:126364. https://doi.org/10.1016/j.matchemphys.2022.126364
Sharma A, Majdinasab M, Khan R et al (2021) Nanomaterials in fluorescence-based biosensors: defining key roles. Nano-Struct Nano-Objects 27:100774. https://doi.org/10.1016/j.nanoso.2021.100774
Souto DEP, Volpe J, de Gonçalves C, C et al (2019) A brief review on the strategy of developing SPR-based biosensors for application to the diagnosis of neglected tropical diseases. Talanta 205:120122. https://doi.org/10.1016/J.TALANTA.2019.120122
Souza MJ (2011) One Health: zoonoses in the exotic animal practice. Vet Clin North Am Exot Anim Pract 14:421–426. https://doi.org/10.1016/J.CVEX.2011.05.007
Trindade GS, Emerson GL, Carroll DS et al (2007) Brazilian vaccinia viruses and their origins. Emerg Infect Dis 13:965. https://doi.org/10.3201/EID1307.061404
Trindade GDS, Emerson GL, Sammons S, Frace M, Govil D, Fernandes Mota BE, Damon IK (2016) Serro 2 virus highlights the fundamental genomic and biological features of a natural vaccinia virus infecting humans. Viruses 8(12):328. https://doi.org/10.3390/V8120328
Wang H, Dong P, Di D et al (2013) Interdigitated microelectrodes biosensor with nanodot arrays for thyroid-stimulating hormone detection. Micro Nano Lett 8:11–14. https://doi.org/10.1049/mnl.2012.0776
Xiang Y, Lane RK (2021) Vaccinia Virus (Poxviridae). Encycl Virol 854–859. https://doi.org/10.1016/B978-0-12-814515-9.00047-3
Zeng YK, Zhou XL, An L et al (2016) A high-performance flow-field structured iron-chromium redox flow battery. J Power Sour 324:738–744. https://doi.org/10.1016/j.jpowsour.2016.05.138
Acknowledgements
The authors gratefully acknowledge financial support from Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasil (CAPES), Finance Code 001.
Author information
Authors and Affiliations
Contributions
RMC performed conceptualization, methodology, investigation, writing—original draft. KLS analyzed resources, methodology, investigation, writing—original draft. AOA and PIS conducted investigation. DBO provided resources, conceptualization, writing—review, review & editing. ACP and DLF drafted writing—review & editing. LFF approved conceptualization, supervision, project administration, funding acquisition, writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Coelho, R.M., de Oliveira Almeida, A., Soares, P.I. et al. A low-cost electrochemical biosensor for vaccinia virus using pencil graphite electrodes modified with poly(hydroxybenzamide). Chem. Pap. 77, 7563–7575 (2023). https://doi.org/10.1007/s11696-023-03073-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-023-03073-y