Abstract
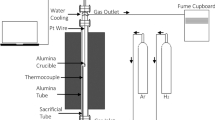
Metal production is facing new challenges due to climate change and declining primary ore quality; thus, new technologies with sustainable processing routes are needed. One way to address this is to recover metals from secondary resources such as end-of-life batteries using a cleaner reducing agent hydrogen that would reduce direct carbon dioxide emissions during pyrometallurgical processing. Recycling of Li-ion battery cathode materials using hydrogen would result in a more benign metal production process and would simultaneously provide a sustainable solution to treat wastes. This study investigated the kinetics of H2 reduction of LiCoO2 at 600–1000 °C up to 180 min reaction time using an isothermal mass change analysis supported with detailed microstructure evolution observations. The overall reduction mechanism appeared to follow the shrinking-core model with a different rate-limiting step depending on the reduction temperature. The activation energy of reduction at 600–800 °C was calculated to be 37.4 kJ/mol and was controlled by the nucleation step. The information and data obtained are useful when comparing different recycling methods and optimizing the reduction parameters of spent Li-ion battery process.




















Similar content being viewed by others

References
G. Gaustad, E. Williams, and A. Leader: Resources Conserv. Recycl., 2021, vol. 167, p. 105213.
A. Tuncuk, V. Stazi, A. Akcil, E.Y. Yazici, and H. Deveci: Miner. Eng., 2012, vol. 25, pp. 28–37.
C. Erüst, A. Akcil, C.S. Gahan, A. Tuncuk, and H. Deveci: J. Chem. Technol. Biotechnol., 2013, vol. 88, pp. 2115–32.
A. Vidyadhar: A Review of Technology of Metal Recovery from Electronic Waste, Rijeka, IntechOpen, 2016, pp. 121–58.
R. Widmer, H. Oswald-Krapf, D. Sinha-Khetriwal, M. Schnellmann, and H. Böni: Environ. Impact Assess. Rev., 2005, vol. 25, pp. 436–58.
B.C.P. Forti, R. Kuehr, G. Bel, The Global E-waste Monitor 2020: Quantities, Flows and the Circular Economy Potential (Bonn/Geneva/Rotterdam, 2020)
IEA, Global EV Outlook 2022 (IEA, Paris, 2022)
IEA, Net Zero by 2050 (IEA, Paris, 2021)
M. Chen, X. Ma, B. Chen, R. Arsenault, P. Karlson, N. Simon, and Y. Wang: Joule, 2019, vol. 3, pp. 2622–46.
Capgemini, Second Life Batteries: A Sustainable Business Opportunity, Not a Conundrum (Capgemini Group, 2019). https://www.capgemini.com/2019/04/second-life-batteries-a-sustainable-business-opportunity-not-a-conundrum/. Accessed 2 Sept 2022
S. Mridha: Metallic Materials, Elsevier, 2016, pp. 1–7.
B. Balasubramaniam, N. Singh, S. Verma, and R.K. Gupta: Recycling of Lithium From Li-ion Batteries, Elsevier, Oxford, 2020, pp. 546–54.
E. Hsu, K. Barmak, A.C. West, and A.H.A. Park: Green Chem., 2019, vol. 21, pp. 919–36.
D.W. Blowes, C.J. Ptacek, J.L. Jambor, C.G. Weisener, D. Paktunc, W.D. Gould, and D.B. Johnson: 115—The Geochemistry of Acid Mine Drainage, Elsevier, Oxford, 2014, pp. 131–90.
M. Aarabi-Karasgani, F. Rashchi, N. Mostoufi, and E. Vahidi: Hydrometallurgy, 2010, vol. 102, pp. 14–21.
T. Havlik, D. Orac, M. Petranikova, A. Miskufova, F. Kukurugya, and Z. Takacova: J. Hazard. Mater., 2010, vol. 183, pp. 866–73.
Y. Huo, Z. Chang, W. Li, S. Liu, and B. Dong: Waste and Biomass Valorization, 2015, vol. 6, pp. 159–65.
W. Gu, J. Bai, B. Dong, X. Zhuang, J. Zhao, C. Zhang, J. Wang, and K. Shih: Chem. Eng. J., 2017, vol. 324, pp. 122–29.
M. Wędrychowicz, A. Piotrowicz, T. Skrzekut, P. Noga, and A. Bydalek: Materials (Basel), 2022, vol. 15, p. 2089.
D. Spreitzer and J. Schenk: Steel Res. Int., 2019, vol. 90, p. 1900108.
S. Luidold and H. Antrekowitsch: JOM, 2007, vol. 59, pp. 58–62.
Z. Huang, F. Liu, B. Makuza, D. Yu, X. Guo, and Q. Tian: ACS Sustain. Chem. Eng., 2022, vol. 10, pp. 756–65.
Z. Huang, D. Yu, B. Makuza, Q. Tian, X. Guo, and K. Zhang: Front. Chem., 2022, vol. 10, p. 1019493.
H. Pinegar, R. Marthi, P. Yang, and Y.R. Smith: ACS Sustain. Chem. Eng., 2021, vol. 9, pp. 7447–53.
B.A. Nuraeni, K. Avarmaa, L.H. Prentice, W.J. Rankin, and M.A. Rhamdhani: SM&T, 2022, vol. 34, p. e00526.
B.A. Nuraeni, K. Avarmaa, L.H. Prentice, W.J. Rankin, and M.A. Rhamdhani: Metall. Mater. Trans. B, 2023, vol. 54(2), pp. 602–20.
B.A. Nuraeni, K.L. Avarmaa, L.H. Prentice, W.J. Rankin, M.I. Pownceby, and M.A. Rhamdhani: Metall. Mater. Trans. B, 2023, vol. 54(4), pp. 2011–36.
ILO and WHO, Lithium Hydroxide Monohydrate (ILO, 2021). https://www.ilo.org/dyn/icsc/showcard.display?p_version=2&p_card_id=0914. Accessed 1 Sept 2022
O. Levenspiel: Chemical Reaction Engineering, 3rd ed. Wiley, New York, 1999, p. 566.
A. Khawam and D.R. Flanagan: J. Phys. Chem. B, 2006, vol. 110, pp. 17315–28.
L. Mooij and B. Dam: Phys. Chem. Chem. Phys., 2013, vol. 15(27), pp. 11501–10.
F. Pyrczak: Making Sense of Statistics: A Conceptual Overview, 5th ed. Pyczak Pub, Glendale, 2010, pp. 141–45.
B. Tomić-Tucaković, D. Majstorović, D. Jelić, and S. Mentus: Thermochim. Acta. Acta, 2012, vol. 541, pp. 15–24.
N.G. Gallegos and J.M.P. Lopez: Mater. Chem. Phys., 1988, vol. 19, pp. 431–46.
H.Y. Lin and Y.W. Chen: Mater. Chem. Phys., 2004, vol. 85, pp. 171–75.
L.E. Revell and B.E. Williamson: J. Chem. Educ., 2013, vol. 90, pp. 1024–27.
Acknowledgments
The authors thank and acknowledge the financial support from Swinburne University’s Automotive Engineering Graduate Program (AEGP) by the Australian Government through the Department of Industry, Science, and Resources, and the Commonwealth Scientific and Industrial Research Organisation (CSIRO).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nuraeni, B.A., Avarmaa, K.L., Prentice, L.H. et al. Hydrogen Reduction of LiCoO2 Cathode Material: A Kinetic Study. Metall Mater Trans B 55, 319–336 (2024). https://doi.org/10.1007/s11663-023-02960-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-023-02960-9