Abstract
Background
Little is known about the risk factors and frequency of metronidazole-associated neurological adverse events.
Objective
To investigate the risk factors and frequency of metronidazole-associated neurological adverse events.
Design
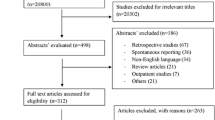
This retrospective study contained two parts. First, we investigated metronidazole treatment-associated neurologic adverse events by performing a population-based cohort study using the Korea Adverse Event Reporting System (KAERS) database from January 2011 to December 2020. Second, we conducted a matched case-control study based on a retrospective cohort of patients treated with metronidazole between January 2006 and July 2021 at a tertiary hospital in South Korea. The data analysis was performed from August 2021 to April 2022.
Participants
In the case-control study, case patients were defined as those diagnosed with metronidazole-associated encephalopathy or peripheral neuropathy during the study period with causal assessment based on the clinical diagnoses and findings from associated tests. In a ratio of 1:3, case patients were compared to a control group of patients prescribed metronidazole without neurologic adverse events matched for age and cumulative dose of metronidazole.
Main Measures
Frequency and risk factors for metronidazole-associated neurological adverse events.
Key Results
Overall, 2,309 cases of neurologic adverse events were reported to the KAERS from 2011 to 2020, and the number of reported neurological adverse events showed an increasing trend. Further, 92,838 patients were prescribed metronidazole during the study period at the Severance Hospital; 54 patients were diagnosed with metronidazole-associated encephalopathy or peripheral neuropathy, 40 with central and 28 with peripheral nervous system adverse events. Liver cirrhosis, chronic kidney disease, intravenous administration, and lower body weight were identified as risk factors for these adverse events.
Conclusions
The number of reported metronidazole-associated neurological adverse events are increasing. Prolonged metronidazole treatment in patients with the aforementioned factors requires careful examination for neurological adverse events.


Similar content being viewed by others
Data Availability:
The datasets during and/or analysed during the current study available from the corresponding author on reasonable request.
References
Hernández Ceruelos A, Romero-Quezada LC, Ruvalcaba Ledezma JC, López Contreras L. Therapeutic uses of metronidazole and its side effects: an update. Eur Rev Med Pharmacol Sci. 2019;23(1):397-401. https://doi.org/10.26355/eurrev_201901_16788.
Tamma PD, Avdic E, Li DX, Dzintars K, Cosgrove SE. Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med. 2017;177(9):1308-15. https://doi.org/10.1001/jamainternmed.2017.1938.
Löfmark S, Edlund C, Nord CE. Metronidazole is still the drug of choice for treatment of anaerobic infections. Clin Infect Dis. 2010;50 Suppl 1:S16-23. https://doi.org/10.1086/647939.
Carroll MW, Jeon D, Mountz JM, et al. Efficacy and safety of metronidazole for pulmonary multidrug-resistant tuberculosis. Antimicrob Agents Chemother. 2013;57(8):3903-9. https://doi.org/10.1128/aac.00753-13.
Brandt LJ, Bernstein LH, Boley SJ, Frank MS. Metronidazole therapy for perineal Crohn's disease: a follow-up study. Gastroenterology. 1982;83(2):383-7.
Lee SU, Jung IE, Kim HJ, Kim JS. Metronidazole-induced combined peripheral and central vestibulopathy. J Neurol Sci. 2016;365:31-3. https://doi.org/10.1016/j.jns.2016.03.043.
Kim J, Chun J, Park JY, et al. Metronidazole-induced encephalopathy in a patient with Crohn's disease. Intest Res. 2017;15(1):124-9. https://doi.org/10.5217/ir.2017.15.1.124.
Goolsby TA, Jakeman B, Gaynes RP. Clinical relevance of metronidazole and peripheral neuropathy: a systematic review of the literature. Int J Antimicrob Agents. 2018;51(3):319-25. https://doi.org/10.1016/j.ijantimicag.2017.08.033.
Alston TA, Abeles RH. Enzymatic conversion of the antibiotic metronidazole to an analog of thiamine. Arch Biochem Biophys. 1987;257(2):357-62. https://doi.org/10.1016/0003-9861(87)90577-7.
Bradley WG, Karlsson IJ, Rassol CG. Metronidazole neuropathy. Br Med J. 1977;2(6087):610-1. https://doi.org/10.1136/bmj.2.6087.610.
Rao DN, Mason RP. Generation of nitro radical anions of some 5-nitrofurans, 2- and 5-nitroimidazoles by norepinephrine, dopamine, and serotonin. A possible mechanism for neurotoxicity caused by nitroheterocyclic drugs. J Biol Chem. 1987;262(24):11731–6.
Sørensen CG, Karlsson WK, Amin FM, Lindelof M. Metronidazole-induced encephalopathy: a systematic review. J Neurol. 2020;267(1):1-13. https://doi.org/10.1007/s00415-018-9147-6.
Hobbs K, Stern-Nezer S, Buckwalter MS, Fischbein N, Finley Caulfield A. Metronidazole-induced encephalopathy: not always a reversible situation. Neurocrit Care. 2015;22(3):429-36. https://doi.org/10.1007/s12028-014-0102-9.
Daneman N, Cheng Y, Gomes T, et al. Metronidazole-associated Neurologic Events: A Nested Case-control Study. Clin Infect Dis. 2021;72(12):2095-100. https://doi.org/10.1093/cid/ciaa395.
Management KIoDSR. Introduction of KAERS. https://www.drugsafe.or.kr/iwt/ds/en/report/WhatIsKAERS.do. Accessed 2023–07–20.
Shin YS, Lee YW, Choi YH, et al. Spontaneous reporting of adverse drug events by Korean regional pharmacovigilance centers. Pharmacoepidemiol Drug Saf. 2009;18(10):910-5. https://doi.org/10.1002/pds.1796.
Choi NK, Park BJ. [Adverse drug reaction surveillance system in Korea]. J Prev Med Public Health. 2007;40(4):278-84. https://doi.org/10.3961/jpmph.2007.40.4.278.
WHO-ART 092 and SOC Korean terminology. Korea Institute of Drug Safety & Risk Management. 2015. https://www.drugsafe.or.kr/iwt/ds/ko/bbs/EgovBbs.do?bbsId=BBSMSTR_000000000251&nttId=1593&pageIndex=3&searchCnd=0&searchWrd=. Accessed 2023–07–11.
Stevens PE, Levin A. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013;158(11):825-30. https://doi.org/10.7326/0003-4819-158-11-201306040-00007.
KASL clinical practice guidelines for liver cirrhosis: Varices, hepatic encephalopathy, and related complications. Clin Mol Hepatol. 2020;26(2):83-127. https://doi.org/10.3350/cmh.2019.0010n.
Singal AK, Bataller R, Ahn J, Kamath PS, Shah VH. ACG Clinical Guideline: Alcoholic Liver Disease. Am J Gastroenterol. 2018;113(2):175-94. https://doi.org/10.1038/ajg.2017.469.
ATC/DDD Index 2023. WHO Collaborating Centre for Drug Statistics Methodology https://www.whocc.no/atc_ddd_index/. Accessed 2023–07–11.
Thornhill MH, Dayer MJ, Durkin MJ, Lockhart PB, Baddour LM. Risk of adverse reactions to oral antibiotics prescribed by dentists. J Dent Res. 2019;98(10):1081-7. https://doi.org/10.1177/0022034519863645.
Ludwig E, Csiba A, Magyar T, Szöcs G, Graber H. Age-associated pharmacokinetic changes of metronidazole. Int J Clin Pharmacol Ther Toxicol. 1983;21(2):87-91.
Loft S, Sonne J, Døssing M, Andreasen PB. Metronidazole pharmacokinetics in patients with hepatic encephalopathy. Scand J Gastroenterol. 1987;22(1):117-23. https://doi.org/10.3109/00365528708991867.
Knorr JP, Javed I, Sahni N, Cankurtaran CZ, Ortiz JA. Metronidazole-induced encephalopathy in a patient with end-stage liver disease. Case Reports Hepatol. 2012;2012:209258. https://doi.org/10.1155/2012/209258.
Sarna JR, Furtado S, Brownell AK. Neurologic complications of metronidazole. Can J Neurol Sci. 2013;40(6):768-76. https://doi.org/10.1017/s0317167100015870.
Sonthalia N, Pawar SV, Mohite AR, et al. Metronidazole-Induced Encephalopathy in Alcoholic Liver Disease: A Diagnostic and Therapeutic Challenge. J Emerg Med. 2016;51(4):e79-e83. https://doi.org/10.1016/j.jemermed.2016.05.038.
Aloy B, Launay-Vacher V, Bleibtreu A, et al. Antibiotics and chronic kidney disease: Dose adjustment update for infectious disease clinical practice. Med Mal Infect. 2020;50(4):323-31. https://doi.org/10.1016/j.medmal.2019.06.010.
Houghton GW, Smith J, Thorne PS, Templeton R. The pharmacokinetics of oral and intravenous metronidazole in man. J Antimicrob Chemother. 1979;5(5):621-3. https://doi.org/10.1093/jac/5.5.621.
Polasek TM, Lin FP, Miners JO, Doogue MP. Perpetrators of pharmacokinetic drug-drug interactions arising from altered cytochrome P450 activity: a criteria-based assessment. Br J Clin Pharmacol. 2011;71(5):727-36. https://doi.org/10.1111/j.1365-2125.2011.03903.x.
Zemanová N, Lněničková K, Vavrečková M, et al. Gut microbiome affects the metabolism of metronidazole in mice through regulation of hepatic cytochromes P450 expression. PLoS ONE. 2021;16(11):e0259643. https://doi.org/10.1371/journal.pone.0259643.
Makunts T, Abagyan R. How can proton pump inhibitors damage central and peripheral nervous systems? Neural Regen Res. 2020;15(11):2041-2. https://doi.org/10.4103/1673-5374.282252.
Chey WD, Leontiadis GI, Howden CW, Moss SF. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am J Gastroenterol. 2017;112(2):212-39. https://doi.org/10.1038/ajg.2016.563.
Sedgwick P. Bias in observational study designs: case-control studies. Bmj. 2015;350:h560. https://doi.org/10.1136/bmj.h560.
Acknowledgements
Contributors: We would like to thank Editage (www.editage.co.kr) for English language editing.
Funding
None.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest:
None reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Prior presentations None.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, S.J., Kim, J., Lee, K.H. et al. Frequency and Risk Factor Analysis for Metronidazole-Associated Neurologic Adverse Events. J GEN INTERN MED 39, 912–920 (2024). https://doi.org/10.1007/s11606-023-08566-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11606-023-08566-w



