Abstract
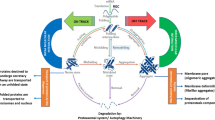
Proteins that misfold into hyper-stable/degradation-resistant species during aging may accumulate and disrupt protein homeostasis (i.e., proteostasis), thereby posing a survival risk to any organism. Using the method diagonal two-dimensional (D2D) SDS-PAGE, which separates hyper-stable SDS-resistant proteins at a proteomics level, we analyzed the plasma of healthy young (<30 years) and older (60–80 years) adults. We discovered the presence of soluble SDS-resistant protein aggregates in the plasma of older adults, but found significantly lower levels in the plasma of young adults. We identified the inflammation-related chaperone protein haptoglobin as the main component of the hyper-stable aggregates. This observation is consistent with the growing link between accumulations of protein aggregates and aging across many organisms. It is plausible higher amounts of SDS-resistant protein aggregates in the plasma of older adults may reflect a compromise in proteostasis that may potentially indicate cellular aging and/or disease risk. The results of this study have implications for further understanding the link between aging and the accumulation of protein aggregates, as well as potential for the development of aging-related biomarkers. More broadly, this novel application of D2D SDS-PAGE may be used to identify, quantify, and characterize the degradation-resistant protein aggregates in human plasma or any biological system.



Similar content being viewed by others
References
Baig UI, Bhadbhade BJ, Watve MG (2014) Evolution of aging and death: what insights bacteria can provide. Q Rev Biol 89:209–223
Bailey RA (2008) Design of comparative experiments. Cambridge University Press, New York
Baker GT, Sprott RL (1988) Biomarkers of aging. Exp Gerontol 23:223–239
Balch WE, Morimoto RI, Dillin A, Kelly JW (2008) Adapting proteostasis for disease intervention. Science 319:916–919
Byerley LO, Leamy L, Tam SW, Chou CW, Ravussin E, Study LHA (2010) Development of a serum profile for healthy aging. Age 32:497–507
Casella G (2008) Statistical design. Springer-Verlag, New York
David DC (2012) Aging and the aggregating proteome. Front Genet 3:247
Ding J, Kopchick JJ (2011) Plasma biomarkers of mouse aging. Age 33:291–307
Emanuele E (2014) Can trehalose prevent neurodegeneration? Insights from experimental studies. Curr Drug Targets 15:551–557
Eng JK, McCormack AL, Yates JR (1994) An approach to correlate tandem mass spectral data of peptides with amino acid sequences in a protein database. J Am Soc Mass Spectrom 5:976–989
Ferrucci L et al. (2006) Relationship of plasma polyunsaturated fatty acids to circulating inflammatory markers. J Clin Endocrinol Metab 91:439–446
Jones OR et al. (2014) Diversity of ageing across the tree of life. Nature 505:169–173
Kang R et al. (2008) Neural palmitoyl-proteomics reveals dynamic synaptic palmitoylation. Nature 456:904–909
Katz PR, Karuza J, Gutman SI, Bartholomew W, Richman G (1990) A comparison between erythrocyte sedimentation rate (ESR) and selected acute-phase proteins in the elderly. Am J Clin Pathol 94:637–640
Knoefler D, Thamsen M, Koniczek M, Niemuth NJ, Diederich AK, Jakob U (2012) Quantitative in vivo redox sensors uncover oxidative stress as an early event in life. Mol Cell 47:767–776
Koga H, Kaushik S, Cuervo AM (2011) Protein homeostasis and aging: the importance of exquisite quality control. Ageing Res Rev 10:205–215
Labbadia J and Morimoto RI. (2014) Proteostasis and longevity: when does aging really begin? F1000Prime Rep 6: 7
Lepez-Otin C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153:1194–1217
Lindner AB, Madden R, Demarez A, Stewart EJ, Taddei F (2008) Asymmetric segregation of protein aggregates is associated with cellular aging and rejuvenation. Proc Natl Acad Sci U S A 105:3076–3081
Maisonneuve E, Ezraty B, Dukan S (2008) Protein aggregates: an aging factor involved in cell death. J Bacteriol 190:6070–6075
Manning M, Colon W (2004) Structural basis of protein kinetic stability: resistance to sodium dodecyl sulfate suggests a central role for rigidity and a bias toward beta-sheet structure. Biochemistry 43:11248–11254
Miura Y et al. (2011) Proteomic analysis of plasma proteins in Japanese semisuper centenarians. Exp Gerontol 46:81–85
Morimoto RI (2008) Proteotoxic stress and inducible chaperone networks in neurodegenerative disease and aging. Genes Dev 22:1427–1438
Napolioni V, Gianni P, Carpi FM, Concetti F, Lucarini N (2011) Haptoglobin (HP) polymorphisms and human longevity: a cross-sectional association study in a Central Italy population. Clin Chim Acta 412:574–577
Pareja-Galeano H, Alis R, Sanchis-Gomar F, Lucia A, Emanuele E (2015) Vitamin D, precocious acute myocardial infarction, and exceptional longevity. Int J Cardiol 199:405–406
Rana A, Rera M, Walker DW (2013) Parkin overexpression during aging reduces proteotoxicity, alters mitochondrial dynamics, and extends lifespan. Proc Natl Acad Sci U S A 110:8638–8643
Rousseau F, Serrano L, Schymkowitz JW (2006) How evolutionary pressure against protein aggregation shaped chaperone specificity. J Mol Biol 355:1037–1047
Rowe JW, Kahn RL (1987) Human aging—usual and successful. Science 237:143–149
Shamsi KS, Pierce A, Ashton AS, Halade DG, Richardson A, Espinoza SE (2012) Proteomic screening of glycoproteins in human plasma for frailty biomarkers. J Gerontol A Biol Sci Med Sci 67:853–864
Song IU, Kim YD, Chung SW, Cho HJ (2015) Association between serum haptoglobin and the pathogenesis of Alzheimer’s disease. Intern Med 54:453–457
Spagnuolo MS et al. (2014a) Haptoglobin interacts with apolipoprotein E and beta-amyloid and influences their crosstalk. ACS Chem Neurosci 5:837–847
Spagnuolo MS et al. (2014b) Haptoglobin increases with age in rat hippocampus and modulates Apolipoprotein E mediated cholesterol trafficking in neuroblastoma cell lines. Front Cell Neurosci 8:1–13
Tartaglia GG, Pellarin R, Cavalli A, Caflisch A (2005) Organism complexity anti-correlates with proteomic beta-aggregation propensity. Protein Sci 14:2735–2740
Teunissen CE et al. (2011) Consensus guidelines for CSF and blood biobanking for CNS biomarker studies. Mult Scler Int 2011:246412
Theilgaard-Monch K et al. (2006) Haptoglobin is synthesized during granulocyte differentiation, stored in specific granules, and released by neutrophils in response to activation. Blood 108:353–361
Xia K, Manning M, Hesham H, Lin Q, Bystroff C, Colon W (2007) Identifying the subproteome of kinetically stable proteins via diagonal 2D SDS/PAGE. Proc Natl Acad Sci U S A 104:17329–17334
Xia K, Zhang S, Bathrick B, Liu S, Garcia Y, Colon W (2012) Quantifying the kinetic stability of hyperstable proteins via time-dependent SDS trapping. Biochemistry 51:100–107
Yerbury JJ, Rybchyn MS, Easterbrook-Smith SB, Henriques C, Wilson MR (2005) The acute phase protein haptoglobin is a mammalian extracellular chaperone with an action similar to clusterin. Biochemistry 44:10914–10925
Acknowledgments
This work was supported in part by a grant (#1158375) from the National Science Foundation to W. Colón.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All participants were recruited with informed consent and approval by the Institutional Review Board.
Electronic supplementary material
About this article
Cite this article
Xia, K., Trasatti, H., Wymer, J.P. et al. Increased levels of hyper-stable protein aggregates in plasma of older adults. AGE 38, 56 (2016). https://doi.org/10.1007/s11357-016-9919-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11357-016-9919-9