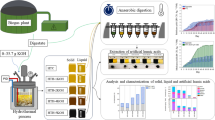
Abstract
In this present work, the transformation of the Moroccan phosphogypsum (PG) waste, considered a potential source of sulfate, into potassium sulfate compound could help reduce environmental impact and create a new value chain for the phosphate industry. Generally, solid–liquid equilibria are frequently applied in chemical industries. They are a valuable aid in visualizing the precipitation, separation, and purification of a solid phase and the pathways by which crystallization can occur. This process aims to produce potassium sulfate (K2SO4), a high-value fertilizer, from sulfate solutions obtained after dissolving PG in a NaOH medium. The quaternary phase diagram Na+, K+//Cl−, SO42−-H2O at 25 °C was especially used to determine the operating conditions and the design of a crystallization process during the PG conversion into K2SO4. The Jänecke representation of this system enables the determination of the optimal trajectory in the phase diagram for the double decomposition reaction. X-ray fluorescent (XRF) and X-ray diffraction (XRD) techniques were conducted to identify the crystalline phases formed during our process. In summary, the results of this study could contribute to the development of a sustainable valorization PG. Furthermore, K2SO4 represents a good alternative to potassium chloride for chloride-sensitive crops.
Graphical Abstract









Similar content being viewed by others
Data availability
All data derived during the experiments are given in the paper and referenced.
References
Aagli A, Tamer N, Atbir A et al (2005) Conversion of phosphogypsum to potassium sulfate: part I. The effect of temperature on the solubility of calcium sulfate in concentrated aqueous chloride solutions. J Therm Anal Calorim 82:395–399. https://doi.org/10.1007/s10973-005-0908-y
Abe M, Tanaka S, Noguchi M, Yamasaki A (2021) Investigation of mineral carbonation with direct bubbling into concrete sludge. ACS Omega 6:15564–15571. https://doi.org/10.1021/acsomega.0c04758
Abu-Eishah SI, Bani-Kananeh AA, Allawzi MA (2000) K2SO4 production via the double decomposition reaction of KCl and phosphogypsum. Chem Eng J 76:197–207. https://doi.org/10.1016/S1385-8947(99)00158-8
Agayr K, Chanouri H, Achiou B et al (2022) Study on the kinetics of the conversion of Moroccan phosphogypsum into X2SO4 (X = Na, NH4). J Mater Cycles Waste Manag 24:2015–2029
Avşar C, Tümük D, Yüzbaşıoğlu AE, Gezerman AO (2022) Focusing on the Merseburg process: benefits on industrial decarbonization and waste minimization. Environ Technol Rev 11:148–155
Belahbib L, Arhouni FE, Boukhair A et al (2021) Impact of phosphate industry on natural radioactivity in sediment, seawater, and coastal marine fauna of El Jadida Province, Morocco. J Hazardous, Toxic, Radioact Waste 25:04020064. https://doi.org/10.1061/(asce)hz.2153-5515.0000563
Berland J-M, Juery C (2002) Les procédés membranaires pour le traitement de l’eau. Doc Tech FNDAE 4:1–71
Biyoune MG, Bouargane B, Bari H et al (2020) Water quality depends on remineralization’s method in the desalination plant. Mediterr J Chem 10:162–170. https://doi.org/10.13171/mjc10202002141228mgb
Biyoune MG, Bouargane B, Idboufrade A et al (2021) New procedure for water-salinity reduction using phosphogypsum waste and carbon dioxide resulting in useful compounds formation. Nanotechnol Environ Eng 6:1–18. https://doi.org/10.1007/s41204-021-00125-0
Bouargane B, Marrouche A, El IS et al (2019) Recovery of Ca(OH)2, CaCO3, and Na2SO4 from Moroccan phosphogypsum waste. J Mater Cycles Waste Manag. https://doi.org/10.1007/s10163-019-00910-9
Bouargane B, Biyoune MG, Mabrouk A et al (2020) Experimental investigation of the effects of synthesis parameters on the precipitation of calcium carbonate and portlandite from Moroccan phosphogypsum and pure gypsum using carbonation route. Waste and Biomass Valorization. https://doi.org/10.1007/s12649-019-00923-3
Burnett WC, Schultz MK, Hull CD (1996) Radionuclide flow during the conversion of phosphogypsum to ammonium sulfate. J Environ Radioact 32:33–51. https://doi.org/10.1016/0265-931X(95)00078-O
Cai Q, Jiang J, Ma B et al (2021) Efficient removal of phosphate impurities in waste phosphogypsum for the production of cement. Sci Total Environ 780:146600. https://doi.org/10.1016/j.scitotenv.2021.146600
Cánovas CR, Macías F, Pérez-lópez R et al (2017) Valorization of wastes from the fertilizer industry: current status and future trends. J Clean Prod. https://doi.org/10.1016/j.jclepro.2017.10.293
Cao Y, Cui Y, Yu X et al (2021) Bibliometric analysis of phosphogypsum research from 1990 to 2020 based on literatures and patents. Environ Sci Pollut Res 28:66845–66857. https://doi.org/10.1007/s11356-021-15237-y
Cárdenas-Escudero C, Morales-Flórez V, Pérez-López R et al (2011) Procedure to use phosphogypsum industrial waste for mineral CO2 sequestration. J Hazard Mater 196:431–435. https://doi.org/10.1016/j.jhazmat.2011.09.039
Chaalal O, Madhuranthakam CMR, Moussa B, Hossain MM (2020) Sustainable approach for recovery of sulfur from phophogypsum. ACS Omega 5:8151–8157. https://doi.org/10.1021/acsomega.0c00420
Chauffour JP (2018) Morocco 2040: Emerging by investing in intangible capital. Directions in development—countries and regions. World Bank, Washington, DC. https://openknowledge.worldbank.org/handle/10986/28442
Cheng H, Wu L, Cao L et al (2019) Phase diagram of AlCl3-FeCl3-H2O(-HCl) salt water system at 298.15 K and its application in the crystallization of AlCl3·6H2O. J Chem Eng Data 64:5089–5094. https://doi.org/10.1021/acs.jced.9b00238
Chernysh Y, Yakhnenko O, Chubur V, Roubík H (2021) Phosphogypsum recycling: a review of environmental issues, current trends, and prospects. Appl Sci 11:1–22. https://doi.org/10.3390/app11041575
Churka Blum S, Caires EF, Alleoni LRF (2013) Lime and phosphogypsum application and sulfate retention in subtropical soils under no-till system. J Soil Sci Plant Nutr 13:279–300. https://doi.org/10.4067/S0718-95162013005000024
Contreras M, Pérez-López R, Gázquez MJ et al (2014) Fractionation and fluxes of metals and radionuclides during the recycling process of phosphogypsum wastes applied to mineral CO2 sequestration. Waste Manag 45:412–419. https://doi.org/10.1016/j.wasman.2015.06.046
Dávila JLW (2009) Production of ionic liquids through metathesis and melt crystallization. Thesis, University of Erlangen-Nuremberg. https://opus4.kobv.de/opus4-fau/files/831/JorgeLuisWongDavilaDissertation.pdf
Degirmenci N (2008) The using of waste phosphogypsum and natural gypsum in adobe stabilization. Constr Build Mater 22:1220–1224. https://doi.org/10.1016/j.conbuildmat.2007.01.027
El Zrelli R, Rabaoui L, Daghbouj N et al (2018) Characterization of phosphate rock and phosphogypsum from Gabes phosphate fertilizer factories (SE Tunisia): high mining potential and implications for environmental protection. Environ Sci Pollut Res 25:14690–14702. https://doi.org/10.1007/s11356-018-1648-4
Ennaciri Y, Mouahid FE, Bendriss A, Bettach M (2013) Conversion of phosphogypsum to potassium sulfate and calcium carbonate in aqueous solution. MATEC Web Conf 5:04006
Ezzahra F, Mohammed A, Ahmed H et al (2022) Better filterability and reduced radioactivity of phosphogypsum during phosphoric acid production in Morocco using a fly ash waste and pure silica additive. J Radioanal Nucl Chem 331:1609–1617. https://doi.org/10.1007/s10967-022-08235-y
Fezei R, Hammi H, Adel M (2008) Study of the sylvite transformation into arcanite at 25 ° C. Woeld J Agric Sci (3)390–397. https://www.idosi.org/wjas/wjas4(3)/17.pdf
Gadikota G (2021) Carbon mineralization pathways for carbon capture, storage and utilization. Commun Chem 4:1–5. https://doi.org/10.1038/s42004-021-00461-x
Gaudry A, Zeroual S, Gaie-Levrel F et al (2007) Heavy metals pollution of the atlantic marine environment by the Moroccan phosphate industry, as observed through their bioaccumulation in Ulva lactuca. Water Air Soil Pollut 178:267–285. https://doi.org/10.1007/s11270-006-9196-9
Ghader S, Mansouri SS, Shadravan V, Farsi A (2010) Representation of material balance for fractional crystallization of reciprocal salt pair systems: KNO3 production case study. J Appl Sci 10:2989–2997
Grabas K, Pawełczyk A, Stręk W et al (2018) Study on the properties of waste apatite phosphogypsum as a raw material of prospective applications. Waste and Biomass Valorization 10:1–13. https://doi.org/10.1007/s12649-018-0316-8
Guerrero JL, Pérez-Moreno SM, Gutiérrez-Álvarez I et al (2020) Behaviour of heavy metals and natural radionuclides in the mixing of phosphogypsum leachates with seawater *. Environ Pollut J 268:115843. https://doi.org/10.1016/j.envpol.2020.115843
Guo XF, Li D, Liu JL et al (2020) Separation of sodium and potassium using adsorption – elution/crystallization scheme from bittern. Chem Eng Res Des 161:72–81. https://doi.org/10.1016/j.cherd.2020.06.028
Hakkar M, Ezzahra Arhouni F, Mahrou A et al (2021) Enhancing rare earth element transfer from phosphate rock to phosphoric acid using an inexpensive fly ash additive. Miner Eng 172:107166. https://doi.org/10.1016/j.mineng.2021.107166
Hammas I, Horchani-Naifer K, Férid M (2013) Solubility study and valorization of phosphogypsum salt solution. Int J Miner Process 123:87–93. https://doi.org/10.1016/j.minpro.2013.05.008
Haneklaus N, Barbossa S, Basallote MD et al (2022) Closing the upcoming EU gypsum gap with phosphogypsum. Resour Conserv Recycl 182:106328. https://doi.org/10.1016/j.resconrec.2022.106328
Horgan DE, Crowley LM, Stokes SP, Lawrence SE, Moynihan HA (2015) Impurity exclusion and retention during crystallisation and recrystallisation—the phenacetin by ethylation of paracetamol process. Advanced Topics in Crystallization. https://doi.org/10.5772/59715
Idboufrade A, Bouargane B, Ennasraoui B et al (2021) Phosphogypsum two-step ammonia-carbonation resulting in ammonium sulfate and calcium carbonate synthesis: effect of the molar ratio OH−/Ca2+ on the conversion process. Waste and Biomass Valorization. https://doi.org/10.1007/s12649-021-01600-0
Išek JI, Kaluđerović LM, Vuković NS et al (2020) Refinement of waste phosphogypsum from Prahovo, Serbia: characterization and assessment of application in civil engineering. Clay Miner 55:63–70. https://doi.org/10.1180/clm.2020.11
Islam GMS, Chowdhury FH, Raihan MT et al (2017) Effect of phosphogypsum on the properties of Portland Cement. Procedia Eng 171:744–751. https://doi.org/10.1016/j.proeng.2017.01.440
Janecke E, Anorg Z (1906) uber eine neue Darstellungsform der wasserigen Losungen zweier und dreier gleichioniger Salze, reziproker Salzpaare und der v a n ’ t Hoffschen Untersuchwngen iiber ozeanische Salzablagerungen. Chem 51:132–157
Jiang G, Wang H, Chen Q et al (2016) Preparation of alpha-calcium sulfate hemihydrate from FGD gypsum in chloride-free Ca(NO3)2 solution under mild conditions. Fuel 174:235–241. https://doi.org/10.1016/j.fuel.2016.01.073
Jurišová J, Fellner P, Danielik V et al (2013) Preparation of potassium nitrate from potassium chloride and magnesium nitrate in a laboratory scale using industrial raw materials. Acta Chim Slovaca 6:15–19. https://doi.org/10.2478/acs-2013-0003
Khlissa F, M’nif A, Rokbani R (2004) Application of the conductimetry to the study of the transformation of KC1 and Na2SO4 into K2SO4 between 5 and 30 °C. Chem Eng Process Process Intensif 43:929–934. https://doi.org/10.1016/j.cep.2003.08.002
Lewis AE, Nathoo J, Thomsen K et al (2010) Design of a eutectic freeze crystallization process for multicomponent waste water stream. Chem Eng Res Des 88:1290–1296. https://doi.org/10.1016/j.cherd.2010.01.023
Liu Y, Guo Y, Wang S et al (2015) Predictive phase equilibria for aqueous quaternary system containing sodium, potassium, chloride and sulfate ions from 273.15 to 473.15 K. Asian J Chem 27:591–596
Liu Y, Zhang Q, Chen Q et al (2019) Utilisation of water-washing pre-treated phosphogypsum for cemented paste backfill. Minerals 9:1–20. https://doi.org/10.3390/min9030175
Lorenz H, Le Minh T, Kaemmerer H et al (2013) Exploitation of shifts of eutectic compositions in crystallization-based enantioseparation. Chem Eng Res Des 91:1890–1902. https://doi.org/10.1016/j.cherd.2013.08.013
Marrouche A, Bouargane B, Mabrouk A et al (2019) Solubility in the ternary system MgCl2-FeCl2-H2O at 288 K by conductance method. Mediterr J Chem 8:10. https://doi.org/10.13171/mjc811902523am
Martín JE, Bolívar JP, Respaldiza MA et al (1995) Environmental impact of fertilizer industries evaluated by PIXE. Nucl Inst Methods Phys Res B 103:477–481. https://doi.org/10.1016/0168-583X(95)00649-4
Masmoudi-Soussi A, Hammas-Nasri I, Horchani-Naifer K, Férid M (2019) Study of rare earths leaching after hydrothermal conversion of phosphogypsum. Chem Africa 2:415–422. https://doi.org/10.1007/s42250-019-00048-z
Mehahad MS, Bounar A (2019) The extractive industries and society phosphate mining, corporate social responsibility and community development in the Gantour Basin, Morocco. Extr Ind Soc 7:170–180. https://doi.org/10.1016/j.exis.2019.11.016
Monat L, Chaudhury S, Nir O (2020) Enhancing the sustainability of phosphogypsum recycling by integrating electrodialysis with bipolar membranes. ACS Sustain Chem Eng 8:2490–2497. https://doi.org/10.1021/acssuschemeng.9b07038
Nicolaisen H, Rasmussen P, Sørensen JM (1993) Correlation and prediction of mineral solubilities in the reciprocal salt system (Na+, K+)(Cl-, SO42-)-H2O at 0–100 °C. Chem Eng Sci 48:3149–3158. https://doi.org/10.1016/0009-2509(93)80201-Z
Paz-Gómez DC, Vilarinho IS, Pérez-Moreno SM, Carvalheiras J, Guerrero JL, Novais RM, Seabra MP, Ríos G, Bolívar JP, Labrincha JA (2021) Immobilization of hazardous wastes on one-part blast furnace slag-based geopolymers. Sustainability 13(23):13455. https://doi.org/10.3390/su132313455
Rentería-Villalobos M, Vioque I, Mantero J, Manjón G (2010) Radiological, chemical and morphological characterizations of phosphate rock and phosphogypsum from phosphoric acid factories in SW Spain. J Hazard Mater 181:193–203. https://doi.org/10.1016/j.jhazmat.2010.04.116
Romero-Hermida MI, Borrero-López AM, Alejandre FJ, et al (2019) Phosphogypsum waste lime as a promising substitute of commercial limes: a rheological approach. Cem Concr Compos 95https://doi.org/10.1016/j.cemconcomp.2018.11.007
Rutherford PM, Dudas MJ, Arocena JM (1996) Heterogeneous distribution of radionuclides, barium and strontium in phosphogypsum by-product. Sci Total Environ 180:201–209. https://doi.org/10.1016/0048-9697(95)04939-8
Salas BV, Wiener MS, Martinez JRS (2017) Phosphoric acid industry: problems and solutions. Phosphoric Acid Ind - Probl Solut. https://doi.org/10.5772/intechopen.70031
Seader JD, Henley EJ, Rope DK (2010) Separation process principles: chemical and biochemical operations (3rd ed.). In: John Wiley & Sons, Inc., p 792. https://imtk.ui.ac.id/wp-content/uploads/2014/02/Separation-Process-Principles-Third-Edition.pdf
Shuyskiy AI, Prokopov AY, Torlina EA (2020) Phosphogypsum processing method. IOP Conf Ser Mater Sci Eng 753https://doi.org/10.1088/1757-899X/753/2/022013
Silva LFO, Oliveira MLS, Crissien TJ, et al (2021) A review on the environmental impact of phosphogypsum and potential health impacts through the release of nanoparticles. Chemosphere 131513https://doi.org/10.1016/j.chemosphere.2021.131513
Soliev L (2020) Invariant equilibria in multinary systems. Russ J Inorg Chem 65:212–216. https://doi.org/10.1134/S0036023620020187
Szajerski P (2020) Distribution of uranium and thorium chains radionuclides in different fractions of phosphogypsum grains. Environ Sci Pollut Res 27:15856–15868. https://doi.org/10.1007/s11356-020-08090-y
Taboada ME, Graber TA, Cisternas LA et al (2007) Process design for drowning-out crystallization of lithium hydroxide monohydrate. Chem Eng Res Des 85:1325–1330. https://doi.org/10.1205/cherd06251
Taha Y, Elghali A, Hakkou R, Benzaazoua M (2021) Towards zero solid waste in the sedimentary phosphate industry: challenges and opportunities. Minerals 11:1–20. https://doi.org/10.3390/min11111250
Takano K, Gani R, Ishikawa T, Kolar P (2000) Computer aided design and analysis of separation processes with electrolyte systems. Comput Chem Eng 24:645–651. https://doi.org/10.1016/S0098-1354(00)00383-5
Tam SK, Chan HC, Ng KM (2011) Design of protein crystallization processes guided by phase diagrams. Ind Eng Chem Res 50:8163–8175
Tayibi H, Choura M, López FA et al (2009) Environmental impact and management of phosphogypsum. J Environ Manage 90:2377–2386. https://doi.org/10.1016/j.jenvman.2009.03.007
Urwin SJ, Levilain G, Marziano I et al (2020) A structured approach to cope with impurities during industrial crystallization development a structured approach to cope with impurities during industrial crystallization development. Org Process Res Dev. https://doi.org/10.1021/acs.oprd.0c00166
Wu S, Yao X, Ren C et al (2020) Recycling phosphogypsum as a sole calcium oxide source in calcium sulfoaluminate cement and its environmental effects. J Environ Manage 271:110986. https://doi.org/10.1016/j.jenvman.2020.110986
Wu F, Ren Y, Qu G et al (2022) Utilization path of bulk industrial solid waste: a review on the multi-directional resource utilization path of phosphogypsum. J Environ Manage 313:114957
Xia X, Zhang L, Li Z et al (2022) Recovery of CaO from CaSO4 via CO reduction decomposition under different atmospheres. J Environ Manage 301:113855
Xie C, Zhang T, Wang X et al (2018) Fluid phase equilibria solid-liquid phase equilibria in aqueous solutions of four common fertilizers at 303.2 K and atmospheric pressure. Fluid Phase Equilib 474:131–140. https://doi.org/10.1016/j.fluid.2018.07.016
Yang L, Zhang Y, Yan Y (2016) Utilization of original phosphogypsum as raw material for the preparation of self-leveling mortar. J Clean Prod 127:204–213. https://doi.org/10.1016/j.jclepro.2016.04.054
Yang Y, Zhang T, Wang X, Tang S (2018) Fluid phase equilibria an investigation on the solid e liquid phase equilibrium of the quaternary system KH2PO4 - H3PO4 - CH2OHCH2OH - H2O. Fluid Phase Equilib 464:12–21. https://doi.org/10.1016/j.fluid.2018.02.014
Zheng S, Ning P, Ma L et al (2011) Reductive decomposition of phosphogypsum with high-sulfur-concentration coal to SO2 in an inert atmosphere. Chem Eng Res Des 89:2736–2741. https://doi.org/10.1016/j.cherd.2011.03.016
Zhou H, Zhang J, Zhang H et al (2012) Salt-forming regions of Na+, Mg2+//Cl−, SO42−−H2O system at 373.15 K in the nonequilibrium state of isothermal boiling evaporation. J Chem Eng Data 57:1192–1202
Zuo Y, Chen Q, Li C et al (2019) Removal of fluorine from wet-process phosphoric acid using a solvent extraction technique with tributyl phosphate and silicon oil. ACS Omega 4:11593–11601. https://doi.org/10.1021/acsomega.9b01383
Funding
The authors would like to acknowledge the support provided by the University of Ibn Zohr, Faculty of Sciences Agadir, through the research project APUiz/2018.
Author information
Authors and Affiliations
Contributions
Brahim Bouargane: investigation, methodology, conceptualization, visualization, and writing original draft. Khaoula Laaboubi: investigation, experimental methodology, and visualization. Moreno Silvia Pérez: characterization and analysis. Bahcine Bakiz: writing—review and editing. Ali Atbir: visualization, review and editing, supervision, project administration, and resource acquisition. Juan Pedro Bolívar Raya: supervision, project administration, review and editing, and resource acquisition.
Corresponding author
Ethics declarations
Ethics approval
This is not applicable.
Consent to participate
The authors consent to participate in the publication of this manuscript at the Environmental Science and Pollution Research journal.
Consent for publication
This is not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• The transformation of the Moroccan PG waste into K2SO4 was done.
• The quaternary phase diagram Na+, K+//Cl-, SO42--H2O at 25 °C was used to determine the operating conditions of the PG conversion to K2SO4.
• The main advantage of this process over the conventional process is that it does not require the use of commercial Na2SO4 or synthetic gypsum but only PG waste.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Laaboubi, K., Bouargane, B., Moreno, S.P. et al. Continuous and simultaneous conversion of phosphogypsum waste to sodium sulfate and potassium sulfate using quaternary phase diagram. Environ Sci Pollut Res 30, 37344–37356 (2023). https://doi.org/10.1007/s11356-022-24799-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24799-4