Abstract
In this study, a zirconium elemental organic framework (UiO-66-NH2) was prepared by a green synthesis method and showed a good adsorption performance for removing 2,4-dichlorophenoxyacetic acid (2,4-D) from water. UiO-66-NH2 was analyzed by a variety of characterization methods and the adsorption properties of 2,4-D on UiO-66-NH2 were investigated by static adsorption experiments. The results showed that the adsorption of 2,4-D had a wide pH range (2–10) and good salt tolerance with the adsorption equilibrium time about 2 h. The maximum adsorption capacity from Langmuir was up to 652 mg g−1 at 303 K. The isotherms can be described by Langmuir model and the adsorption kinetics was consistent with pseudo-second-order kinetic model and Elovich model. The regeneration efficiency was still 95% after 5 cycles with 0.01 mol L−1 NaOH as desorption solution. The feasibility of practical application of UiO-66-NH2 was explored by simulating actual wastewater at different pH. UiO-66-NH2 is promising to remove 2,4-D from water.
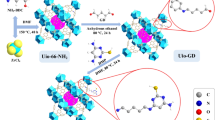
Graphical abstract










Similar content being viewed by others
Data availability
The dataset generated and analyzed during this study could be obtained from the corresponding author on reasonable request.
References
Afshin S, Rashtbari Y, Vosough M, Dargahi A, Fazlzadeh M, Behzad A, Yousefi M (2021) Application of Box-Behnken design for optimizing parameters of hexavalent chromium removal from aqueous solutions using Fe3O4 loaded on activated carbon prepared from alga: Kinetics and equilibrium study. J Water Process Eng 42:102113. https://doi.org/10.1016/j.jwpe.2021.102113
Aryee AA, Mpatani FM, Kani AN, Dovi E, Han RP, Li ZH, Qu LB (2021) A review on functionalized adsorbents based on peanut husk for the sequestration of pollutants in wastewater: modification methods and adsorption study. J Clean Prod 310:127502. https://doi.org/10.1016/j.jclepro.2021.127502
Carboneras B, Villasenor J, Fernandez-Morales FJ (2017) Modelling aerobic biodegradation of atrazine and 2,4-dichlorophenoxy acetic acid by mixed-cultures. Bioresour Technol 243:1044–1050. https://doi.org/10.1016/j.biortech.2017.07.089
Carles L, Martin-Laurent F, Devers M, Spor A, Rouard N, Beguet J, Besse-Hoggan P, Batisson I (2021) Potential of preventive bioremediation to reduce environmental contamination by pesticides in an agricultural context: a case study with the herbicide 2,4-D. J Hazard Mater 416:125740. https://doi.org/10.1016/j.jhazmat.2021.125740
Chen P, He XH, Pang MB, Dong XT, Zhao S, Zhang W (2020) Iodine capture using Zr-based metal–organic frameworks (Zr-MOFs): adsorption performance and mechanism. ACS Appl Mater Inter 12:20429–20439. https://doi.org/10.1021/acsami.0c02129
Chinalia FA, Killham KS (2006) 2,4-Dichlorophenoxyacetic acid (2,4-D) biodegradation in river sediments of Northeast-Scotland and its effect on the microbial communities (PLFA and DGGE). Chemosphere 64:1675–1683. https://doi.org/10.1016/j.chemosphere.2006.01.022
Dovi E, Kani AN, Aryee AA, Ma J, Li JJ, Li ZH, Qu LB, Han RP (2021) Decontamination of bisphenol A and Congo red dye from solution by using CTAB functionalised walnut shell. Environ Sci Pollut Res 28:28732–28749. https://doi.org/10.1007/s11356-021-12550-4
Fajardo-Díaz JL, López-Urías F, Muñoz-Sandoval E (2020) Chloride functionalized carbon nanotube sponge: high charge capacity and high magnetic saturation. Carbon 164:324–336. https://doi.org/10.1016/j.carbon.2020.04.016
Fang X, Wu SB, Wu YH, Yang W, Li YL, He JY, Hong PD, Nie MX, Xie C, Wu ZJ, Zhang KS, Kong LT, Liu JH (2020) High-efficiency adsorption of norfloxacin using octahedral UIO-66-NH2 nanomaterials: dynamics, thermodynamics, and mechanisms. Appl Surf Sci 518:146226. https://doi.org/10.1016/j.apsusc.2020.146226
Franco D, Silva LFO, Martinello KdB, Diel JC, Georgin J, Netto MS, Pereira HA, Lima EC, Dotto GL (2021) Transforming agricultural waste into adsorbent: application of Fagopyrum esculentum wheat husks treated with H2SO4 to adsorption of the 2,4-D herbicide. J Environ Chem Eng 9:106872. https://doi.org/10.1016/j.jece.2021.106872
Fu LK, Wang SX, Lin G, Zhang LB, Liu QM, Zhou HH, Kang CX, Wan SY, Li HW, Wen S (2019) Post-modification of UiO-66-NH2 by resorcyl aldehyde for selective removal of Pb(II) in aqueous media. J Clean Prod 229:470–479. https://doi.org/10.1016/j.jclepro.2019.05.043
Giri RR, Ozaki H, Ota S, Taniguchi S, Takanami R (2010) Influence of inorganic solids on photocatalytic oxidation of 2,4-dichlorophenoxyacetic acid with UV and TiO2 fiber in aqueous solution. Desalination 255:9–14. https://doi.org/10.1016/j.desal.2010.01.025
Hameed BH, Salman JM, Ahmad AL (2009) Adsorption isotherm and kinetic modeling of 2,4-D pesticide on activated carbon derived from date stones. J Hazard Mater 163:121–126. https://doi.org/10.1016/j.jhazmat.2008.06.069
Hernández-Moreno EJ, Martínez de la Cruz A, Hinojosa-Reyes L, Guzmán-Mar J, Gracia-Pinilla MA, Hernández-Ramírez A (2021) Synthesis, characterization, and visible light–induced photocatalytic evaluation of WO3/NaNbO3 composites for the degradation of 2,4-D herbicide. Mater Today Chem 19:100406. https://doi.org/10.1016/j.mtchem.2020.100406
Huang LJ, He M, Chen BB, Hu B (2018) Magnetic Zr-MOFs nanocomposites for rapid removal of heavy metal ions and dyes from water. Chemosphere 199:435–444. https://doi.org/10.1016/j.chemosphere.2018.02.019
Ighalo JO, Adeniyi AG, Adelodun AA (2021) Recent advances on the adsorption of herbicides and pesticides from polluted waters: performance evaluation via physical attributes. J Ind Eng Chem 93:117–137. https://doi.org/10.1016/j.jiec.2020.10.011
Jung BK, Hasan Z, Jhung SH (2013) Adsorptive removal of 2,4-dichlorophenoxyacetic acid (2,4-D) from water with a metal–organic framework. Chem Eng J 234:99–105. https://doi.org/10.1016/j.cej.2013.08.110
Khan MM, Khan A, Bhatti HN, Zahid M, Alissa SA, El-Badry YA, Hussein EE, Iqbal M (2021) Composite of polypyrrole with sugarcane bagasse cellulosic biomass and adsorption efficiency for 2,4-dicholrophonxy acetic acid in column mode. J Mater Res Technol 15:2016–2025. https://doi.org/10.1016/j.jmrt.2021.09.028
Lazarotto JS, Martinello KdB, Georgin J, Franco DSP, Netto MS, Piccilli DGA, Silva LFO, Lima EC, Dotto GL (2021) Preparation of activated carbon from the residues of the mushroom (Agaricus bisporus) production chain for the adsorption of the 2,4-dichlorophenoxyacetic herbicide. J Environ Chem Eng 9:106843. https://doi.org/10.1016/j.jece.2021.106843
Li SM, Feng F, Chen S, Zhang XL, Liang YX, Shan SS (2020) Preparation of UiO-66-NH2 and UiO-66-NH2/sponge for adsorption of 2,4-dichlorophenoxyacetic acid in water. Ecotox Environ Safe 194:110440. https://doi.org/10.1016/j.ecoenv.2020.110440
Lin DW, Liu X, Huang RL, Qi W, Su RX, He ZM (2019) One-pot synthesis of mercapto functionalized Zr-MOFs for the enhanced removal of Hg2+ ions from water. Chem Commun 55:6775–6778. https://doi.org/10.1039/c9cc03481a
Lin KA, Liu YT, Chen SY (2016) Adsorption of fluoride to UiO-66-NH2 in water: stability, kinetic, isotherm and thermodynamic studies. J Colloid Interface Sci 461:79–87. https://doi.org/10.1016/j.jcis.2015.08.061
Liu BY, Guo ND, Wang Z, Wang Y, Hao X, Yang ZL, Yang Q (2022) Adsorption of 2,4-Dichlorophenoxyacetic acid over Fe–Zr-based metal-organic frameworks: synthesis, characterization, kinetics, and mechanism studies. J Environ Chem Eng 22:107472. https://doi.org/10.1016/j.jece.2022.107472
Liu MY, Zhang XT, Li ZH, Qu LB, Han RP (2020) Fabrication of zirconium (IV)-loaded chitosan/Fe3O4/graphene oxide for efficient removal of alizarin red from aqueous solution. Carbohydr Polym 248:116792. https://doi.org/10.1016/j.carbpol.2020.116792
Liu W, Yang Q, Yang ZL, Wang WJ (2016) Adsorption of 2,4-D on magnetic graphene and mechanism study. Colloid Surface A 509:367–375. https://doi.org/10.1016/j.colsurfa.2016.09.039
Liu XY, Kirlikovali KO, Chen ZJ, Ma KK, Idrees KB, Cao R, Zhang X, Islamoglu T, Liu YL, Farha OK (2021a) Small molecules, big effects: tuning adsorption and catalytic properties of metal–organic frameworks. Chem Mater 33:1444–1454. https://doi.org/10.1021/acs.chemmater.0c04675
Liu YY, Sun YQ, Wan ZH, Jing FQ, Li ZX, Chen JW, Tsang DCW (2021b) Tailored design of food waste hydrochar for efficient adsorption and catalytic degradation of refractory organic contaminant. J Clean Prod 310:127482. https://doi.org/10.1016/j.jclepro.2021.127482
Luo HP, Zhou X, Chen QY, Zhou J (2021) Removal of 2,4-dichlorophenoxyacetic acid by the boron-nitrogen co-doped carbon nanotubes: insights into peroxymonosulfate adsorption and activation. Sep Purif Technol 259:118196. https://doi.org/10.1016/j.seppur.2020.118196
Mandal S, Sarkar B, Igalavithana AD, Ok YS, Yang X, Lombi E, Bolan N (2017) Mechanistic insights of 2,4-D sorption onto biochar: influence of feedstock materials and biochar properties. Bioresour Technol 246:160–167. https://doi.org/10.1016/j.biortech.2017.07.073
Mazloomi S, Yousefi M, Nourmoradi H, Shams M (2019) Evaluation of phosphate removal from aqueous solution using metal organic framework; isotherm, kinetic and thermodynamic study. J Environ Health Sci 17:209–218. https://doi.org/10.1007/s40201-019-00341-6
Moghaddam MH, Nabizadeh R, Dehghani MH, Akbarpour B, Azari A, Yousefi M (2019) Performance investigation of zeolitic imidazolate framework-8 (ZIF-8) in the removal of trichloroethylene from aqueous solutions. Microchem J 150:104185. https://doi.org/10.1016/j.microc.2019.104185
Mpatani FM, Aryee AA, Han RP, Kani AN, Li ZH, Qu LB (2021) Green fabrication of a novel cetylpyridinium-bagasse adsorbent for sequestration of micropollutant 2,4-D herbicide in aqueous system and its antibacterial properties against S. aureus and E. coli. J Environ Chem Eng 9:106714. https://doi.org/10.1016/j.jece.2021.106714
Mpatani FM, Aryee AA, Kani AN, Han RP, Li ZH, Dovi E, Qu LB (2021b) A review of treatment techniques applied for selective removal of emerging pollutant-trimethoprim from aqueous systems. J Clean Prod 308:127359. https://doi.org/10.1016/j.jclepro.2021.127359
Orduz AE, Acebal C, Zanini G (2021) Activated carbon from peanut shells: 2,4-D desorption kinetics study for application as a green material for analytical purposes. J Environ Chem Eng 9:104601. https://doi.org/10.1016/j.jece.2020.104601
Rezaei R, Mohseni M (2017) Impact of pH on the kinetics of photocatalytic oxidation of 2,4-dichlorophenoxy acetic acid in a fluidized bed photocatalytic reactor. Appl Catal B-Environ 205:302–309. https://doi.org/10.1016/j.apcatb.2016.12.038
Rodriguez JL, Valenzuela MA, Poznyak T, Lartundo L, Chairez I (2013) Reactivity of NiO for 2,4-D degradation with ozone: XPS studies. J Hazard Mater 262:472–481. https://doi.org/10.1016/j.jhazmat.2013.08.041
Salomón YLdO, Georgin J, Franco DSP, Netto MS, Piccilli DGA, Foletto EL, Oliveira LFS, Dotto GL (2021) High-performance removal of 2,4-dichlorophenoxyacetic acid herbicide in water using activated carbon derived from Queen palm fruit endocarp (Syagrus romanzoffiana). J Environ Chem Eng 9:104911. https://doi.org/10.1016/j.jece.2020.104911
Shi XY, Zhang XD, Bi FK, Zheng ZH, Sheng LJ, Xu JC, Wang Z, Yang YQ (2020) Effective toluene adsorption over defective UiO-66-NH2: an experimental and computational exploration. J Mol Liq 316:113812. https://doi.org/10.1016/j.molliq.2020.113812
Tan KL, Foo KY (2021) Preparation of MIL-100 via a novel water-based heatless synthesis technique for the effective remediation of phenoxyacetic acid-based pesticide. J Environ Chem Eng 9:104923. https://doi.org/10.1016/j.jece.2020.104923
Wang C, Xiong C, He YL, Yang C, Li XT, Zheng JZ, Wang SX (2021a) Facile preparation of magnetic Zr-MOF for adsorption of Pb(II) and Cr(VI) from water: adsorption characteristics and mechanisms. Chem Eng J 415:128923. https://doi.org/10.1016/j.cej.2021.128923
Wang YF, Zhao W, Qi ZY, Zhang L, Zhang YN, Huang HO, Peng YZ (2020) Designing ZIF-8/hydroxylated MWCNT nanocomposites for phosphate adsorption from water: capability and mechanism. Chem Eng J 394:124992. https://doi.org/10.1016/j.cej.2020.124992
Wu GG, Ma JP, Li S, Wang SS, Jiang B, Luo SY, Li JH, Wang XY, Guan YF, Chen LX (2020) Cationic metal-organic frameworks as an efficient adsorbent for the removal of 2,4-dichlorophenoxyacetic acid from aqueous solutions. Environ Res 186:109542. https://doi.org/10.1016/j.envres.2020.109542
Wu HY, Gong L, Zhang X, He FR, Li ZL (2021) Bifunctional porous polyethyleneimine-grafted lignin microspheres for efficient adsorption of 2,4-dichlorophenoxyacetic acid over a wide pH range and controlled release. Chem Eng J 411:128539. https://doi.org/10.1016/j.cej.2021.128539
Xia J, Gao YX, Yu G (2021) Tetracycline removal from aqueous solution using zirconium-based metal-organic frameworks (Zr-MOFs) with different pore size and topology: adsorption isotherm, kinetic and mechanism studies. J Colloid Interf Sci 590:495–505. https://doi.org/10.1016/j.jcis.2021.01.046
Yousefi M, Gholami M, Oskoei V, Mohammadi AA, Baziar M, Esrafili A (2021) Comparison of LSSVM and RSM in simulating the removal of ciprofloxacin from aqueous solutions using magnetization of functionalized multi-walled carbon nanotubes: Process optimization using GA and RSM techniques. J Environ Chem Eng 9:105677. https://doi.org/10.1016/j.jece.2021.105677
Zahid M, Zhang D, Xu X, Pan M, UlHaq MH, Reda AT, Xu W (2021) Barbituric and thiobarbituric acid-based UiO-66-NH2 adsorbents for iodine gas capture: characterization, efficiency and mechanisms. J Hazard Mater 416:125835. https://doi.org/10.1016/j.jhazmat.2021.125835
Zeng HJ, Yu ZX, Shao LY, Li XH, Zhu M, Liu YC, Feng XF, Zhu XM (2021) A novel strategy for enhancing the performance of membranes for dyes separation: embedding PAA@UiO-66-NH2 between graphene oxide sheets. Chem Eng J 403:126281. https://doi.org/10.1016/j.cej.2020.126281
Zhang MM, Sun Q, Wang YJ, Shan WJ, Lou ZN, Xiong Y (2021a) Synthesis of porous UiO-66-NH2-based mixed matrix membranes with high stability, flux and separation selectivity for Ga(III). Chem Eng J 421:129748. https://doi.org/10.1016/j.cej.2021.129748
Zhang XT, Liu MY, Han RP (2021b) Adsorption of phosphate on UiO-66-NH2 prepared by a green synthesis method. J Environ Chem Eng 9:106672. https://doi.org/10.1016/j.jece.2021.106672
Zhang XT, Zhang SS, Ou Yang GF, Han RP (2021c) Removal of Cr(VI) from solution using UiO-66-NH2 prepared in a green way. Korean J Chem Eng 39:1839–1849. https://doi.org/10.1007/s11814-021-1015-x
Zhao F, Yang WX, Han Y, Luo XL, Tang WZ, Yue TL, Li ZH (2021) A straightforward strategy to synthesize supramolecular amorphous zirconium metal-organic gel for efficient Pb(II) removal. Chem Eng J 407:126744. https://doi.org/10.1016/j.cej.2020.126744
Zhu XX, Li JF, Xie B, Feng DQ, Li YM (2020) Accelerating effects of biochar for pyrite-catalyzed Fenton-like oxidation of herbicide 2,4-D. Chem Eng J 391:123605. https://doi.org/10.1016/j.cej.2019.123605
Zhuang ST, Cheng R, Wang JL (2019) Adsorption of diclofenac from aqueous solution using UiO-66-type metal-organic frameworks. Chem Eng J 359:354–362. https://doi.org/10.1016/j.cej.2018.11.150
Zuanazzi NR, Ghisi NC, Oliveira EC (2020) Analysis of global trends and gaps for studies about 2,4-D herbicide toxicity: a scientometric review. Chemosphere 241:125016. https://doi.org/10.1016/j.chemosphere.2019.125016
Funding
This work was supported in part by the Henan Province Basis and Advancing Technology Research Project in China (142300410224).
Author information
Authors and Affiliations
Contributions
Xiaoting Zhang: conceptualization; methodology; formal analysis; investigation; visualization, software; writing original draft.
Runping Han: conceptualization; resources; project administration; writing—review and editing; visualization; supervision; funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Han, R. Adsorption of 2,4-dichlorophenoxyacetic acid by UiO-66-NH2 obtained in a green way. Environ Sci Pollut Res 29, 90738–90751 (2022). https://doi.org/10.1007/s11356-022-22127-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22127-4