Abstract
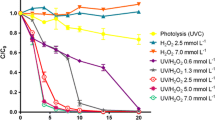
Sulfaquinoxaline (SQX) is an antimicrobial of the sulfonamide class, frequently detected at low levels in drinking and surface water as organic micropollutant. The main goal of the present study is the evaluation of SQX reactivity during chlorination and UV irradiations which are two processes mainly used in water treatment plants. The SQX transformation by chlorination and UV lights (254 nm) was investigated in purified water at common conditions used for water disinfection (pH = 7.2, temperature = 25 °C, [chlorine] = 3 mg L−1). The result shows a slow degradation of SQX during photolysis compared with chlorination process. Kinetic studies that fitted a fluence-based first-order kinetic model were used to determine the kinetic constants of SQX degradation; they were equal to 0.7 × 10−4 and 0.7 × 10−2 s−1corresponding to the half time lives of 162 and 1.64 min during photolysis and chlorination, respectively. In the second step, seven by-products were generated during a chlorination and photo-transformation of SQX and identified using liquid chromatography with electrospray ionization and tandem mass spectrometry (MS-MS). SO2 extrusion and direct decomposition were the common degradation pathway during photolysis and chlorination. Hydroxylation and isomerization were observed during photodegradation only while electrophilic substitution was observed during chlorination process.






Similar content being viewed by others
References
Abdallah H, Arnaudguilhem C, Jaber F, Lobinski R (2014) Multiresidue analysis of 22 sulfonamides and their metabolites in animal tissues using quick, easy, cheap, effective, rugged, and safe extraction and high resolution mass spectrometry (hybrid linear iontrap-Orbitrap). J Chromatogr A 1355:61–72. https://doi.org/10.1016/j.chroma.2014.05.078
Acero JL, Javier Benitez FJ, Real FJ, Roldan G (2010) Kinetics of aqueous chlorination of some pharmaceuticals and their elimination from water matrices. Water Res 44(14):4158–4170. https://doi.org/10.1016/j.watres.2010.05.012
Adams C, Wang Y, Loftin K, Meyer M (2002) Removal of antibiotics from surface and distilled water in conventional water treatment processes. J Environ Eng 128(3):253–260. https://doi.org/10.1061/(ASCE)0733-9372(2002)128:3(253)
Avisar D, Lester Y, Mamane H (2010) pH induced polychromatic UV treatment for the removal of a mixture of SMX, OTC and CIP from water. J Hazard Mater 175(1-3):1068–1074. https://doi.org/10.1016/j.jhazmat.2009.10.122
Baran W, Adamek E, Ziemiańska J, Sobczak A (2011) Effects of the presence of sulfonamides in the environment and their influence on human health. J Hazard Mater 196:1–15. https://doi.org/10.1016/j.jhazmat.2011.08.082
Boxall ABA, Kolpin DW, Halling-Sorensen B, Tolls J (2003) Are veterinary medicines causing environmental risks? Environ Sci Technol 37:286–294
Campbell WC (2008) History of the discovery of sulfaquinoxaline as a coccidiostat. J Parasitol 94(4):934–945. https://doi.org/10.1645/GE-1413.1
Chamberlain E, Adams C (2006) Oxidation of sulfonamides, macrolides, and carbadox with free chlorine and monochloramine. Water Res 40(13):2517–2526. https://doi.org/10.1016/j.watres.2006.04.039
Cui C, Jin L, Jiang L, Han Q, Lin K, Lu S, Zhang D, Cao G (2016) Removal of trace level amounts of twelve sulfonamides from drinking water by UV-activated peroxymonosulfate. Sci Total Environ 572:244–251. https://doi.org/10.1016/j.scitotenv.2016.07.183
De Liguoro M, Di Leva V, Gallina G, Faccio E, Pinto G, Pollio A (2010) Evaluation of the aquatic toxicity of two veterinary sulfonamides using five test organisms. Chemosphere 81(6):788–793. https://doi.org/10.1016/j.chemosphere.2010.07.003
Dodd MC, Huang CH (2004) Transformation of the antibacterial agent sulfamethoxazole in reactions with chlorine: kinetics, mechanisms, and pathways. Environ Sci Technol 38(21):5607–5615. https://doi.org/10.1021/es035225z
Doretto KM, Peruchi LM, Rath S (2014) Sorption and desorption of sulfadimethoxine, sulfaquinoxaline and sulfamethazine antimicrobials in Brazilian soils. Sci Total Environ 476–477:406–414. https://doi.org/10.1016/j.scitotenv.2014.01.024
Eisenberg G (1943) Colorimetric determination of hydrogen peroxide. Ind Eng Chem Anal Ed 15(5):327–328. https://doi.org/10.1021/i560117a011
El Najjar NH, Deborde M, Journel R, Vel Leitner NK (2013) Aqueous chlorination of levofloxacin: kinetic and mechanistic study, transformation product identification and toxicity. Water Res 47(1):121–129. https://doi.org/10.1016/j.watres.2012.09.035
Gaffney Vde J, Cardoso W, Benoliel MJ, Almeida CM (2016) Chlorination and oxidation of sulfonamides by free chlorine: identification and behaviour of reaction products by UPLC-MS/MS. J Environ Manag 166:466–477. https://doi.org/10.1016/j.jenvman.2015.10.048
Garcia-Galan MJ, Silvia Diaz-cruz M, Damia Barcelo (2008) Identification and determination of metabolites and degradation products of sulfonamide antibiotics. TrAC 27:1008–1022
Hamilton RJ (1998) Blackie academic and professional. Thomson Science, New York
Hoff RB, Barreto F, Melo J, Jank L, Ruaro Peralba MC, Pizzolato TM (2012) Characterization and estimation of sulfaquinoxaline metabolites in animal tissues using liquid chromatography coupled to tandem mass spectrometry. Anal Method 4(9):2822–2830. https://doi.org/10.1039/c2ay25197c
Hoff B, Meneghini L, Pizzolato T, Peralba M, Diaz-Cruz M, Barcelo D (2014) Structural elucidation of sulfaquinoxaline metabolism products and their occurrence in biological samples using high-resolution orbitrap mass spectrometry. Anal Chem 86(11):5579–5586. https://doi.org/10.1021/ac501132r
Klavarioti M, Mantzavinos D, Kassinos D (2009) Removal of residual pharmaceuticals from aqueous systems by advanced oxidation processes. Environ Internat 35(2):402–417. https://doi.org/10.1016/j.envint.2008.07.009
Le Fur C, Legeret B, de Sainte CP, Wong-Wah-Chung P, Sarakha M (2013) Liquid chromatography/electrospray ionization quadrupole time-of-flight mass spectrometry for the analysis of sulfaquinoxaline byproducts formed in water upon solar light irradiation. Rapid Commun Mass Spectrom 27(6):722–730. https://doi.org/10.1002/rcm.6507
Lian J, Qiang Z, Li M, Bolton JR, Qu J (2015) UV photolysis kinetics of sulfonamides in aqueous solution based on optimized fluence quantification. Water Res 75:43–50. https://doi.org/10.1016/j.watres.2015.02.026
Liao Q-N, Ji F, Li J-C, Zhan X, Hu Z-H (2016) Decomposition and mineralization of sulfaquinoxaline sodium during UV/H2O2 oxidation processes. Chem Eng J 284:494–502. https://doi.org/10.1016/j.cej.2015.08.150
Li L, Wei D, Wei G, Du Y (2013) Transformation of cefazolin during chlorination process: products, mechanism and genotoxicity assessment. J Hazard Mater 262:48–54. https://doi.org/10.1016/j.jhazmat.2013.08.029
Nassar R, Trivella A, Mokh S, Al Iskandarani M, Budzinski H, Mazellier P (2017) Photodegradation of sulfamethazine, sulfamethoxypiridazine, amitriptyline, and clomipramine drugs in aqueous media. J Photochem Photobiol A 336:176–1820. https://doi.org/10.1016/j.jphotochem.2016.12.008
Nicole I, De Laat J, Dore M, Duguet JP, Bonnel C (1990) Use of UV radiation in water treatment measurement of photonic flux hydrogen peroxide actinometry. Water Res 24(2):157–168. https://doi.org/10.1016/0043-1354(90)90098-Q
Shemer H, Sharpless CM, Linden KG (2005) Photodegradation of 3,5,6-trichloro-2-pyridinol in aqueous solution. Water Air Soil Pollut 168(1–4):145–155. https://doi.org/10.1007/s11270-005-0960-z
Urbano V, Maniero M, Perez-Moya M, Guimaraes J (2016) Influence of pH and ozone dose on sulfaquinoxaline ozonation. J Environ. Manage 195:224–231
Yassine M, Rifai A, Doumyati S, Trivella A, Mazellier P, Budzinski H, Al Iskandarani M (2017a) Oxidation of danofloxacin by free chlorine—kinetic study, structural identification of by-products by LC–MS/MS and potential toxicity of by-products using in silico test. Environ Sci Pollut Res 24(9):7982–7993. https://doi.org/10.1007/s11356-017-8409-7
Yassine MH, Rifai A, Hoteit M, Mazellier P, Al Iskandarani, M. (2017b) Study of the degradation process of ofloxacin with free chlorine by using ESI-LCMSMS: kinetic study, byproducts formation pathways and fragmentation mechanisms. Chemosphere 189:46–54
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Nassar, R., Mokh, S., Rifai, A. et al. Transformation of sulfaquinoxaline by chlorine and UV light in water: kinetics and by-product identification. Environ Sci Pollut Res 25, 34863–34872 (2018). https://doi.org/10.1007/s11356-017-0814-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0814-4