Abstract
Saccharomyces cerevisiae is being used for long as a rich source of proteins, sugars, nucleotides, vitamins and minerals. Autolyzed and hydrolyzed yeast biomass has found numerous applications in the health food industry as well as livestock feeds. Here, we have compared three lysis methods for production of yeast lysates using autolysis, plasmolysis (ethyl acetate 1.5%), and enzymatic hydrolysis (Alcalase 0.2%). The efficiency of each process was compared according to soluble solid and protein contents, cell lysis monitoring, and release of intracellular materials, cell viability and microscopic analysis. Results showed that plasmolysis by ethyl acetate was found to be more efficient compared to autolysis, with a higher recovery of yeast extract (YE) content. In comparison, the content of released solids and proteins were higher during the enzymatic hydrolysis using Alcalase compared to autolysis and plasmolysis treatments. The highest decrease in optical density of 600 nm was monitored for the hydrolyzed cells. Besides, we defined “Degree of Leakage (DL)” as a new index of the lysis process, referring to the percentage of total released proteins from the cells and it was estimated to about 65.8%, which represents an appropriate indicator of the cell lysis. The biochemical and biophysical properties of the hydrolyzed yeast product as well as its biological activity (free radical scavenging activity and bacterial binding capacity) suggest that Alcalase could be used to accelerate the lysis of yeast cells and release the valuable intracellular components used for foodstuffs, feed and fermentation media applications.
Graphic abstract
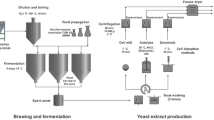
Production of baker’s yeast lysates using autolysis, plasmolysis, and enzymatic hydrolysis methods.






Similar content being viewed by others
References
Alexandre H, Guilloux-Benatier M (2006) Yeast autolysis in sparkling wine—a review. Aust J Grape Wine Res 12:119–127. https://doi.org/10.1111/j.1755-0238.2006.tb00051.x
AOAC (1990) Official methods of analysis, 16th edn. Association of Official Analytical chemists, Washington
AOAC (2000) Official method of analysis, 17th edn. Association of Official Anal ytical Chemists, Inc., Maryland
Babayan TL, Bezrukov MG, Latov VK, Belikov VM, Belavtseva EM, Titova EF (1981) Induced autolysis of Saccharomyces cerevisiae: morphological effects, rheological effects, and dynamics of accumulation of extracellular hydrolysis products. Current Microbiol 5:163–168. https://doi.org/10.1007/BF01578522
Banyay M, Sarkar M, Gräslund A (2003) A library of IR bands of nucleic acids in solution. Biophys Chem 104:477–488. https://doi.org/10.1016/s0301-4622(03)00035-8
Bashir KM, Choi JS (2017) Clinical and physiological perspectives of β-Glucans: the past, present, and future. Int J Mol Sci 18:1906. https://doi.org/10.3390/ijms18091906
Bayarjargal M, Munkhbat E, Ariunsaikhan T, Odonchimeg M, Uurzaikh T, Gan-Erdene T, Regdel D (2014) Utilization of spent brewer’s yeast Saccharomyces cerevisiae for the production of yeast enzymatic hydrolysate. Mong J Chem 12:88–91. https://doi.org/10.5564/mjc.v12i0.179
Behalova B, Beran K (1986) Autolysis of disintegrated cells of the yeast Saccharomyces cerevisiae. Acta Biotechnol 6:147–152. https://doi.org/10.1002/abio.370060212
Bekatorou A, Psarianos C, Koutinas AA (2006) Production of food grade yeasts. Food Technol Biotechnol 44:407–415
Breddam K, Beenfeldt T (1991) Acceleration of yeast autolysis by chemical methods for production of intracellular enzymes. Appl Microbiol Biotechnol. https://doi.org/10.1007/BF00172720
Bzducha-Wróbel A, Kieliszek M, Błażejak S (2013) Chemical composition of the cell wall of probiotic and brewer’s yeast in response to cultivation medium with glycerol as a carbon source. Eur Food Res Technol 237:489–499. https://doi.org/10.1007/s00217-013-2016-8
Bzducha-Wróbel A, Błażejak S, Kawarska A, Stasiak-Różańska L, Gientka I, Majewska E (2014) Evaluation of the efficiency of different disruption methods on yeast cell wall preparation for β-glucan isolation. Molecules 19:20941–20961. https://doi.org/10.3390/molecules191220941
Chae HJ, Joo H, In MJ (2001) Utilization of brewer’s yeast cells for the production of food-grade yeast extract. Part 1: effects of different enzymatic treatments on solid and protein recovery and flavor characteristics. Bioresour Technol 76:253–258. https://doi.org/10.1016/S0960-8524(00)00102-4
Chang ST, Wu JH, Wang SY, Kang PL, Yang NS, Shyur LF (2001) Antioxidant activity of extracts from Acacia confusa bark and heartwood. J Agric Food Chem 49:3420–3424. https://doi.org/10.1021/jf0100907
Conway J, Gaudreau H, Champagne CP (2001) The effect of the addition of proteases and glucanases during yeast autolysis on the production and properties of yeast extracts. Can J Microbiol 47:18–24. https://doi.org/10.1139/w00-118
Dolińska B, Zieliński M, Dobrzański Z, Chojnacka K, Opaliński S, Ryszka F (2012) Influence of incubation conditions on hydrolysis efficiency and iodine enrichment in baker’s yeast. Biol Trace Elem Res 147:354–358. https://doi.org/10.1007/s12011-011-9318-0
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Ganner A, Stoiber C, Uhlik J, Dohnal I, Schatzmayr G (2013) Quantitative evaluation of E. coli F4 and Salmonella typhimurium binding capacity of yeast derivatives. AMB Express 3:62. https://doi.org/10.1186/2191-0855-3-62
Geciova J, Bury D, Jelen P (2002) Methods for disruption of microbial cells for potential use in the dairy industry—a review. Int Dairy J 12:541–553. https://doi.org/10.1016/S0958-6946(02)00038-9
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum proteins by means of the biuret reaction. J Biol Chem 177:751–766
Halász A, Lásztity R (2017) Use of Yeast Biomass in Food Production, 1st edn. Routledge, Abingdon
Hassan HMM (2011) Antioxidant and immunostimulating activities of yeast (Saccharomyces cerevisiae) autolysates. World Appl Sci J 15:1110–1119
Hatoum R, Labrie S, Fliss I (2012) Antimicrobial and probiotic properties of yeasts: from fundamental to novel applications. Front Microbiol 3:421. https://doi.org/10.3389/fmicb.2012.00421
Hromádková Z, Ebringerová A, Sasinková V, Šandula J, Hřı́balová V, Omelková J (2003) Influence of the drying method on the physical properties and immunomodulatory activity of the particulate (1→3)-β-d-glucan from Saccharomyces cerevisiae. Carbohyd Polym 51:9–15. https://doi.org/10.1016/S0144-8617(02)00110-8
In MJ, Kim DC, Chae HJ (2005) Downstream process for the production of yeast extract using brewer’s yeast cells. Biotechnol Bioprocess Eng 10:85–90. https://doi.org/10.1007/BF02931188
Iten W, Matile P (1970) Role of chitinase and other lysosomal enzymes of Coprinus lagopus in the autolysis of fruiting bodies. J Gen Microbiol 61:301–309. https://doi.org/10.1099/00221287-61-3-301
Jouany JP, Yiannikouris A, Bertin G (2005) The chemical bonds between mycotoxins and cell wall components of Saccharomyces cerevisiae have been identified. Arch Zootech 8:26–50
Jung EY, Lee HS, Choi JW, Ra KS, Kim MR, Suh HJ (2011) Glucose tolerance and antioxidant activity of spent Brewer’s yeast hydrolysate with a high content of cyclo-his-pro (CHP). J Food Sci 76:C272–C278. https://doi.org/10.1111/j.1750-3841.2010.01997.x
Kollár R, Šturdík E, Farkaš V (1991) Induction and acceleration of yeast lysis by addition of fresh yeast autolysate. Biotechnol Lett 13:543–546. https://doi.org/10.1007/BF01033406
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lee HJ, Son HS, Park C, Suh HJ (2015) Preparation of yeast hydrolysate enriched in cyclo-his-pro (CHP) by enzymatic hydrolysis and evaluation of its functionality. Prev Nutr Food Sci 20:284–291. https://doi.org/10.3746/pnf.2015.20.4.284
Liesche J, Marek M, Günther-Pomorski T (2015) Cell wall staining with Trypan blue enables quantitative analysis of morphological changes in yeast cells. Front Microbiol 6:107. https://doi.org/10.3389/fmicb.2015.00107
Lü JM, Lin PH, Yao Q, Chen C (2010) Chemical and molecular mechanisms of antioxidants: experimental approaches and model systems. J Cell Mol Med 14:840–860. https://doi.org/10.1111/j.1582-4934.2009.00897.x
Martínez JM, Cebrián G, Álvarez I, Raso J (2016) Release of Mannoproteins during Saccharomyces cerevisiae autolysis induced by pulsed electric field. Front Microbiol 7:1435. https://doi.org/10.3389/fmicb.2016.01435
Martı́nez-Rodrı́guez AJ, Polo MC, Carrascosa AV (2001) Structural and ultrastructural changes in yeast cells during autolysis in a model wine system and in sparkling wines. Int J Food Microbiol 71:45–51. https://doi.org/10.1016/S0168-1605(01)00554-2
Milic TV, Rakin M, Siler-Marinkovic S (2007) Utilization of baker’s yeast (Saccharomyces cerevisiae) for the production of yeast extract: effects of different enzymatic treatments on solid, protein and carbohydrate recovery. J Serb Chem Soc 72:451–457. https://doi.org/10.2298/JSC0705451V
Nagodawithana T (1992) Yeast-derived flavors and flavor enhancers and their probable mode of action. Food Tech (USA) 46:138–144
Novák M, Synytsya A, Gedeon O, Slepička P, Procházka V, Synytsya A, Čopíková J (2012) Yeast β(1–3), (1–6)-d-glucan films: preparation and characterization of some structural and physical properties. Carbohyd Polym 87:2496–2504. https://doi.org/10.1016/j.carbpol.2011.11.031
Orban E, Quaglia GB, Casini I, Moresi M (1994) Effect of temperature and yeast concentration on the autolysis of Kluyverommyces fragilis grown on lactose-based media. J Food Eng 21:245–261. https://doi.org/10.1016/0260-8774(94)90190-2
Ou K, Liu Y, Zhang L, Yang X, Huang Z, Nout MJR, Liang J (2010) Effect of neutrase, alcalase, and papain hydrolysis of whey protein concentrates on iron uptake by Caco-2 cells. J Agric Food Chem 58:4894–4900. https://doi.org/10.1021/jf100055y
Pérez-Sotelo LS, Talavera-Rojas M, Monroy-Salazar HG, Lagunas-Bernabé S, Cuarón-Ibargüengoytia JA, Jimenez RMO, Vázquez-Chagoyán JC (2005) In vitro evaluation of the binding capacity of Saccharomyces cerevisiae Sc47 to adhere to the wall of Salmonella spp. Rev Latinoam Microbiol 47:70–75
Piotrowska M, Masek A (2015) Saccharomyces cerevisiae cell wall components as tools for ochratoxin A decontamination. Toxins 7:1151–1162. https://doi.org/10.3390/toxins7041151
Schiavone M, Sieczkowski N, Castex M, Dague E, Marie François J (2015) Effects of the strain background and autolysis process on the composition and biophysical properties of the cell wall from two different industrial yeasts. FEMS Yeast Res. https://doi.org/10.1093/femsyr/fou012
Shehadul Islam M, Aryasomayajula A, Selvaganapathy P (2017) A review on macroscale and microscale cell lysis methods. Micromachines 8:83. https://doi.org/10.3390/mi8030083
Spring P, Wenk C, Dawson KA, Newman KE (2000) The effects of dietary mannaoligosaccharides on cecal parameters and the concentrations of enteric bacteria in the ceca of salmonella-challenged broiler chicks. Poult Sci 79:205–211. https://doi.org/10.1093/ps/79.2.205
Sugimoto H (1974) Synergistic effect of ethanol and sodium chloride on autolysis of baker’s yeast for preparing food-grade yeast extracts. J Food Sci 39:939–942. https://doi.org/10.1111/j.1365-2621.1974.tb07281.x
Tanguler H, Erten H (2008) Utilisation of spent brewer’s yeast for yeast extract production by autolysis: the effect of temperature. Food Bioprod Process 86:317–321. https://doi.org/10.1016/j.fbp.2007.10.015
Teng PY, Kim WK (2018) Review: roles of prebiotics in intestinal ecosystem of broilers. Front Vet Sci 5:245. https://doi.org/10.3389/fvets.2018.00245
Varelas V, Liouni M, Calokerinos AC, Nerantzis ET (2016) An evaluation study of different methods for the production of β-D-glucan from yeast biomass: an evaluation study of different methods for the production of β-D-glucan from yeast biomass. Drug Test Anal 8:46–55. https://doi.org/10.1002/dta.1833
Vaštag Ž, Popović L, Popović S, Krimer V, Peričin D (2011) Production of enzymatic hydrolysates with antioxidant and angiotensin-I converting enzyme inhibitory activity from pumpkin oil cake protein isolate. Food Chem 124:1316–1321. https://doi.org/10.1016/j.foodchem.2010.07.062
Verduyn C, Suksomcheep A, Suphantharika M (1999) Effect of high pressure homogenization and papain on the preparation of autolysed yeast extract. World J Microbiol Biotechnol 15:57–63. https://doi.org/10.1023/A:1008818511497
Vieira ÉD, Andrietta MGS, Andrietta SR (2013) Yeast biomass production: a new approach in glucose-limited feeding strategy. Braz J Microbiol 44:551–558. https://doi.org/10.1590/S1517-83822013000200035
Vosti DC, Joslyn MA (1954) Autolysis of baker’s yeast. Appl Microbiol 2:70–78
Wang Y, Yao S, Wu T (2003) Combination of induced autolysis and sodium hypochlorite oxidation for the production of Saccharomyces cerevisiae (1 3)-β-D-glucan. World J Microbiol Biotechnol 19:947–952. https://doi.org/10.1023/B:WIBI.0000007328.21451.78
Wang J, Li M, Zheng F, Niu C, Liu C, Li Q, Sun J (2018) Cell wall polysaccharides: before and after autolysis of brewer’s yeast. World J Microbiol Biotechnol 34:137. https://doi.org/10.1007/s11274-018-2508-6
Xu X, Qiao Y, Peng Q, Gao L, Shi B (2017) Inhibitory effects of YCW and MOS from Saccharomyces cerevisiae on Escherichia coli and Salmonella pullorum adhesion to Caco-2 cells. Front Biol 12:370–375. https://doi.org/10.1007/s11515-017-1464-0
Acknowledgements
This work was financially supported by Kimiazyme Company, Tehran, Iran. The technical support by Tarbiat Modares University is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The study was supported by Kimiazyme Company. The authors RHS and MN own shares of the company and ZT and RN are Kimiazyme employees. The Kimiazyme employees have financial interests. Kimiazyme is a producer of feed additives for the animal feed market.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Takalloo, Z., Nikkhah, M., Nemati, R. et al. Autolysis, plasmolysis and enzymatic hydrolysis of baker's yeast (Saccharomyces cerevisiae): a comparative study. World J Microbiol Biotechnol 36, 68 (2020). https://doi.org/10.1007/s11274-020-02840-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-020-02840-3