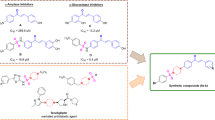
Abstract
Here in this work we successfully designed and synthesized a library of some novel pyrrolidine-triazole-aurone hybrids i.e. (Z)-2-benzylidene-6-((1-(2-(pyrrolidin-1-yl)ethyl)-1H-1,2,3-triazol-4-yl)methoxy)benzofuran-3(2H)-one derivatives 5(a-k). The structures of all the synthesized hybrid aurones were confirmed based on their spectral (FT-IR, 1H NMR, 13C NMR) and HRMS data. In biological studies, the impact of synthesized compounds on digestive enzymes, amylase, lipase, and trypsin was analyzed. These compounds exhibited varying effects on these enzymes as both trypsin and amylase were activated, but a significant inhibition was achieved for the lipase enzyme. Upon examining the binding energies of the synthesized compounds with the enzymes, it was observed that the experimental findings partially aligned with the docking results. These in vitro and in silico results of these hybrid aurones against digestive enzymes, signify their potential as anti-inflammatory and anti-obesity agents.










Similar content being viewed by others
Data availability
Data supporting the findings of this manuscript (spectral and biological data) is either included in the manuscript or provided as supplementary data.
References
F. Liu, D. Lv, L. Wang, X. Feng, R. Zhang, W. Liu, W. Han, BMC Pediatr. 22, 1 (2022)
J.A. Bell, M. Kivimaki, M. Hamer, Obes. Rev. 15, 504 (2014)
A. Abdullah, A. Peeters, M. de Courten, J. Stoelwinder, Diabetes Res. Clin. Pract. 89, 309 (2010)
P.W. Wilson, R.B. D’Agostino, L. Sullivan, H. Parise, W.B. Kannel, Arch. Intern. Med. 162, 1867 (2002)
Y. Lu, K. Hajifathalian, M. Ezzati, M. Woodward, E.B. Rimm, G. Danaei, C. D’Este, Lancet 383, 970 (2014)
E.E. Calle, M.J. Thun, Oncogene 23, 6365 (2004)
H.L. Daneschvar, M.D. Aronson, G.W. Smetana, Am. J. Med. 129, 879 (2016)
K. Woodard, L. Louque, D.S. Hsia, Ther. Adv. Endocrinol. Metab. 11, 2042018820918789 (2020)
E. Pilitsi, O.M. Farr, S.A. Polyzos, N. Perakakis, E. Nolen-Doerr, A.-E. Papathanasiou, C.S. Mantzoros, Metabolism 92, 170 (2019)
T.-T. Liu, X.-T. Liu, Q.-X. Chen, Y. Shi, Biomed. Pharmacother. 128, 110314 (2020)
N.M. Aldossari, E.E. El Gabry, G.E. Gawish, Medicine 98, e15878 (2019)
D. Shah, K. Mital, Adv. Ther. 35, 31 (2018)
B. Seligman, Angiology 6, 208 (1955)
T.N. Shamsi, R. Parveen, S. Afreen, M. Azam, P. Sen, Y. Sharma, Q.M.R. Haque, T. Fatma, N. Manzoor, S. Fatima, J. Diet. Suppl. 15, 939 (2018)
J. Jampilek, Molecules 24, 3839 (2019)
B. Saroha, G. Kumar, M. Kumari, R. Kaur, N. Raghav, P.K. Sharma, N. Kumar, S. Kumar, Int. J. Biol. Macromol. 222, 2270 (2022)
W. Yu, Z.-W. Zhai, D.E. Wedge, S.O. Duke, H.-K. Wu, J.-Q. Weng, C.-X. Tan, Y.-G. Zhang, X.-H. Liu, Res. Chem. Intermed. 45, 5989 (2019)
S. Narwal, S. Kumar, P.K. Verma, Res. Chem. Intermed. 47, 1625 (2021)
A. Gomtsyan, Chem. Heterocycl. Compd. 48, 7 (2012)
P. Pinto, C.M. Machado, J. Moreira, J.D.P. Almeida, P.M. Silva, A.C. Henriques, J.X. Soares, J.A. Salvador, C. Afonso, M. Pinto, Eur. J. Med. Chem. 184, 111752 (2019)
K. Wen, X. Fang, J. Yang, Y. Yao, K.S. Nandakumar, M.L. Salem, K. Cheng, Curr. Med. Chem. 28, 1042 (2021)
N.J. Lawrence, D. Rennison, A.T. McGown, J.A. Hadfield, Bioorg. Med. Chem. Lett. 13, 3759 (2003)
J. Wang, N. Wang, X. Yao, S. Kitanaka, Asian J. Tradit. Med. 2, 23 (2007)
M. Roussaki, S. Costa Lima, A.-M. Kypreou, P. Kefalas, A.C. da Silva, A. Detsi, Int. J. Med. Chem. 2012, 1 (2012)
B.P. Bandgar, S.A. Patil, B.L. Korbad, S.C. Biradar, S.N. Nile, C.N. Khobragade, Eur. J. Med. Chem. 45, 3223 (2010)
H.H. Jardosh, M.P. Patel, Arab. J. Chem. 10, S3781 (2017)
M. Zhang, X.H. Xu, Y. Cui, L.G. Xie, C.H. Kong, Pest Manag. Sci. 68, 1512 (2012)
R. Haudecoeur, A. Ahmed-Belkacem, W. Yi, A. Fortuné, R. Brillet, C. Belle, E. Nicolle, C. Pallier, J.M. Pawlotsky, A. Boumendjel, J. Med. Chem. 54, 5395 (2011)
R. Sheng, Y. Xu, C. Hu, J. Zhang, X. Lin, J. Li, B. Yang, Q. He, Y. Hu, Eur. J. Med. Chem. 44, 7 (2009)
M.P. Carrasco, A.S. Newton, L. Gonçalves, A. Góis, M. Machado, J. Gut, F. Nogueira, T. Hänscheid, R.C. Guedes, D.J.V.A. dos Santos, P.J. Rosenthal, R. Moreira, Eur. J. Med. Chem. 80, 523 (2014)
F. Souard, S. Okombi, C. Beney, S. Chevalley, A. Valentin, A. Boumendjel, Bioorg. Med. Chem. 18, 5724 (2010)
M. Protopopov, V. Vdovin, S. Starosyla, I. Borysenko, A. Prykhod’ko, S. Lukashov, Y. Bilokin, V. Bdzhola, S. Yarmoluk, Bioorg. Chem. 102, 104062 (2020)
M. Morimoto, H. Fukumoto, T. Nozoe, A. Hagiwara, K. Komai, J. Agric. Food Chem. 55, 700 (2007)
B. Saroha, G. Kumar, R. Kumar, M. Kumari, S. Kumar, Chem. Biol. Drug Des. 100, 843 (2022)
J. Mi, J. Wu, C. Zhou, J. Pharmacol. Sci. 23, 84 (2008)
M. Akter, K. Rupa, P. Anbarasan, Chem. Rev. 122, 13108 (2022)
M. Marinescu, Antibiotics 12, 1220 (2023)
W. Ouellette, S. Jones, J. Zubieta, CrystEngComm 13, 4457 (2011)
C.H. Zhou, Y. Wang, Curr. Med. Chem. 19, 239 (2012)
R. Jalaja, S.G. Leela, P.K. Valmiki, C.T.F. Salfeena, K.T. Ashitha, V.R.D. Krishna Rao, M.S. Nair, R.K. Gopalan, S.B. Somappa, ACS Med. Chem. Lett. 9, 662 (2018)
D.R. Buckle, C.J. Rockell, H. Smith, B.A. Spicer, J. Med. Chem. 29, 2262 (1986)
M. Asif, Chem. Int. 1, 174 (2015)
S.H. Sumrra, W. Zafar, M. Imran, Z.H. Chohan, J. Coord. Chem. 75, 293 (2022)
G.C. Tron, T. Pirali, R.A. Billington, P.L. Canonico, G. Sorba, A.A. Genazzani, Med. Res. Rev. 28, 278 (2008)
G. Kumar, B. Saroha, R. Kumar, M. Kumari, S. Dalal, S. Kumar, J. Heterocycl. Chem. 59, 297 (2022)
B. Saroha, G. Kumar, S. Kumar, M. Kumari, M. Rani, N. Raghav, P.K. Sahoo, S. Ghosh, S. Mahata, V.D. Nasare, Eur. J. Med. Chem. Rep. 5, 100056 (2022)
B. Saroha, G. Kumar, S. Kumar, M. Kumari, M. Rani, N. Raghav, P.K. Sahoo, S. Ghosh, S. Mahata, V.D. Nasare, Curr. Res. Green Sustain. Chem. 5, 100295 (2022)
B. Saroha, G. Kumar, P. Arya, N. Raghav, S. Kumar, Bioorg. Chem. 140, 106805 (2023)
G. Kumar, E. Lathwal, B. Saroha, S. Kumar, S. Kumar, N.S. Chauhan, T. Kumar, ChemistrySelect 5, 3539 (2020)
S. Kumar, Green Chem. Lett. Rev. 7, 95 (2014)
F. Antoni, D. Wifling, G. Bernhardt, Eur. J. Med. Chem. 210, 112958 (2021)
E. Kashani-Amin, P. Yaghmaei, B. Larijani, A. Ebrahim-Habibi, Obes. Res. Clin. Pract. 7, e487 (2013)
P. Arya, N. Raghav, J. Mol. Struct. 1228, 129774 (2021)
B. Saroha, G. Kumar, E. Lathwal, S. Kumar, M. Kumari, N. Mor, N. Raghav, S. Kumar, Chem. Data Collect. 31, 100630 (2021)
Acknowledgements
Authors are thankful to Kurukshetra University Kurukshetra for lab facility and other support, DST-New Delhi for NMR support through FIST Programme 2017 (SR/FST/CS-1).
Funding
Author Suresh Kumar acknowledges Kurukshetra University Kurukshetra for the financial assistance in the form of seed money grant (DPA-I/32/22/MRP/2358-2500).
Author information
Authors and Affiliations
Contributions
SK & BS—designed the research work; SK, AB & PB—performed the research work; GK—analysed the spectral data and performed molecular docking studies; MR—did biological screening; BS—did computational study (ADME) and analysed the results; SK & EL—wrote the manuscript; NR—supervision of biological studies; RK—proofread the manuscript; SK*—overall supervision of the project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in publishing this article.
Ethical approval
No animal/human studies were carried out in the present work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumar, S., Lathwal, E., Saroha, B. et al. Design, synthesis, molecular docking and biological studies of some novel pyrrolidine-triazole-aurone hybrids against digestive enzymes. Res Chem Intermed 50, 1249–1271 (2024). https://doi.org/10.1007/s11164-023-05221-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05221-1