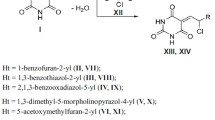
The effect of 5-aryl(hetaryl)methylidene-2,4,6-pyrimidine-2,4,6(1H,3H,5H)-triones and 5-(2-chloropropylidene)-2,4,6-pyrimidine-2,4,6(1H,3H,5H)-triones on the viability of laboratory strain M. tuberculosis H37Rv was studied. The minimum inhibitory and bactericidal concentrations and the acute toxicity in mice have been determined. Compounds XIII, XIV, XVII, and XIX had the greatest antituberculotic activity for M. tuberculosis H37Rv, indicating they were promising for further studies aimed at creating effective antituberculotic drugs.
Similar content being viewed by others
References
Toward Zero Leprosy. Global Leprosy (Hansen’s Disease) Strategy 2021 – 2030, World Health Organization, Apr. 15, 2021: https://apps.who.int/iris/handle/10665/342171; [English version: https://www.who.int/publications/i/item/9789290228509].
L. A. Vinnik, Tuberculosis and Leprosy: Two Fates of the Past? [in Russian], Astrakhan. Gos. Med. Akad., Astrakhan (1997).
A. A. Tsibizova, A. L. Yasenyavskaya, A. A. Ozerov, et al, Sib. Nauchn. Med. Zh., 41(6), 56 – 60 (2021); doi: https://doi.org/10.18699/SSMJ20210606.
O. N. Chupakhin, V. N. Charushin, and V. L. Rusinov, Vestn. Ross. Akad. Nauk, 86(6), 546 – 552 (2016).
M. A. Samatrueva, A. A. Tsibizova, A. L. Yasenyavskaya, et al., Astrakh. Med. Zh., 10(1), 12 – 29 (2015).
V. A. Makarov, Author’s Abstract of a Doctoral Dissertation in Pharmaceutical Sciences, Moscow (2003).
A. V. Khrapova, L. V. Saroyants, M. Yu. Yushin, et al., Khim.- farm. Zh., 55(10), 48 – 54 (2021); Pharm. Chem. J., 55(10), 1108 – 1114 (2022).
L. A. Aleksandrova, E. R. Shmalenyuk, S. N. Kochetkov, et al., Acta Nat., 2(1), 115 – 118 (2010).
E. R. Shmalenyuk, L. N. Chernousova, I. L. Karpenko, et al., Bioorg. Med. Chem., 20, 4874 – 4884 (2013).
E. Matyugina, A. Khandazhinskaya, L. Chernousova, et al., Bioorg. Med. Chem., 20, 6680 – 6686 (2012).
L. A. Alexandrova, V. O. Chekhov, E. R. Shmalenyuk, et al., Bioorg. Med. Chem., 23, 7131 – 7137 (2015).
A. L. Khandazhinskaya, L. A. Alexandrova, E. S. Matyugina, et al., Molecules, 23, 3069 (2018).
M. Yu. Yushin, A. G. Tyrkov, L. V. Saroyants, et al., Khim.-farm. Zh., 54(2), 32 – 35 (2020); Pharm. Chem. J., 54(2), 134 – 137 (2020); doi: https://doi.org/10.30906/0023-1134-2020-54-2-32-35.
M. Yu. Yushin, A. K. Ayupova, A. G. Tyrov, et al., Khim.-farm. Zh., 55(10), 14 – 17 (2021); Pharm. Chem. J., 55(10), 1019 – 1024 (2022); doi: https://doi.org/10.30906/0023-1134-2021-55-10-14-17.
S. A. Luzhnova, A. G. Tyrkov, N. M. Gabitova, et al., Khim.- farm. Zh., 52(6), 18 – 21 (2018); Pharm. Chem. J., 52(6), 506 – 509 (2018); doi: https://doi.org/10.1007/s11094-018-1849-7.
V. V. Menshikov (ed.), Methods of Clinical Laboratory Investigations [in Russian], Vol. 3, Labora, Moscow (2009), pp. 358 – 366.
A. N. Mironov (ed.), Handbook for Preclinical Drug Studies [in Russian], Grif i K, Moscow (2012).
I. V. Berezovskaya, Khim.-farm. Zh., 7(3), 32 – 34 (2003).
Acknowledgments
The work was performed in the framework of State Task No. 056-00113-21-00 Development of Diagnostic and Treatment Methods for Leprosy Infection Based on Personalized Medicine Principles.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 57, No. 2, pp. 28 – 31, February, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yushin, M.Y., Tyrkov, A.G., Lutsenko, A.V. et al. Antituberculotic Activity of 5-Aryl(Hetaryl)-Methylidene- 2,4,6-Pyrimidine-2,4,6(1H,3H,5H)-Triones and 5-(2-Chloropropylidene)-2,4,6-Pyrimidine-2,4,6(1H,3H,5H)-Triones. Pharm Chem J 57, 214–217 (2023). https://doi.org/10.1007/s11094-023-02885-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-023-02885-2