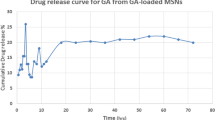
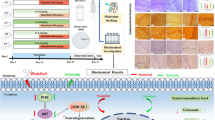
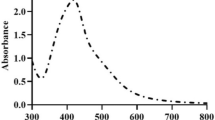
Abstract
Background
Methylphenidate (MPH) abuse has been criticized for its role in neurodegeneration. Also, a high risk of seizure was reported in the first month of MPH treatment. Topiramate, a broad-spectrum Antiepileptic Drug (AED), has been used as a neuroprotective agent in both aforementioned complications. Nanotechnology is introduced to increase desirable neurological treatment with minimum side effects. We aimed to investigate the potential neuroprotective activity of topiramate loaded on nanoparticles.
Methods and results
MTT assay was performed to evaluate the cellular cytotoxicity of Mesoporous Silica Nanoparticles (MSN). Male rats were randomly divided into eight groups. Rats received an intraperitoneal (i.p) MPH (10 mg/kg) injection and a daily oral dose of topiramate (TPM, 30 mg/kg), MSN with Zn core (10 and 30 mg/kg), and MSN with Cu core (10 and 30 mg/kg) for three weeks. On day 21, a seizure was induced by a single injection of pentylenetetrazole (PTZ) to evaluate the protective effects of TPM-loaded nanoparticles on seizure latency and duration following MPH-induced neurotoxicity. Moreover, the hippocampal content of tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), malondialdehyde (MDA), and the anti-oxidant enzymes (SOD, GPx, and GR) activities were assessed. Also, BAX and Bcl-2 as two main apoptotic markers were evaluated.
Results
MPH neurotoxicity was observed as a raised duration and reduced latency in PTZ-induced seizure. However, TPM-loaded MSN with Zn species (NE) treatment reduced the duration and improved the latency time. Also, NE and, somewhat, TPM-loaded MSN with Cu species (NM) administration reduced inflammatory cytokines, MDA, and Bax levels and increased activities in the rat hippocampus.
Conclusion
TPM-loaded nanoparticles could be used as neuroprotective agents against MPH-induced neurodegeneration by improving seizure parameters and reducing inflammatory, oxidant, and apoptotic factors.







Similar content being viewed by others
Data availability
Any datasets used can be accessed if required.
References
Payne DE et al (2021) Identifying seizure risk factors: a comparison of sleep, weather, and temporal features using a bayesian forecast. Epilepsia 62(2):371–382
Sheibani M et al (2023) Orexin receptor antagonists in the pathophysiology and treatment of sleep disorders and epilepsy. Neuropeptides 99:102335
Christensen J et al (2023) Estimates of epilepsy prevalence, psychiatric co-morbidity and cost seizure: European. J Epilepsy 107:162–171
Organization WH (2023) Intersectoral Global Action Plan on Epilepsy and Other Neurological Disorders 2022–2031
Meenu M et al (2019) Evaluation of sodium valproate loaded nanoparticles in acute and chronic pentylenetetrazole induced seizure models. Epilepsy Res 158:106219
Perucca E, Meador KJ (2005) Adverse effects of antiepileptic drugs. Acta Neurol Scand 112(s181):30–35
Kumar S, Kaur G (2014) Second generation anti-epileptic drugs adversely affect reproductive functions in young non-epileptic female rats. Eur Neuropsychopharmacol 24(10):1709–1718
Guerrini R, Parmeggiani L (2006) Topiramate and its clinical applications in epilepsy. Expert Opin Pharmacother 7(6):811–823
Łotowska J et al (2019) Effects of topiramate on the ultrastructure of synaptic endings in the hippocampal CA1 and CA3 sectors in the rat experimental model of febrile seizures: the first electron microscopy report. Folia Neuropathol 57(3):267–276
Mula M et al (2003) Topiramate and psychiatric adverse events in patients with epilepsy. Epilepsia 44(5):659–663
Motaghinejad M et al (2017) Topiramate via NMDA, AMPA/kainate, GABA(A) and Alpha2 receptors and by modulation of CREB/BDNF and Akt/GSK3 signaling pathway exerts neuroprotective effects against methylphenidate-induced neurotoxicity in rats. J Neural Transm (Vienna) 124(11):1369–1387
Andrade C (2020) Methylphenidate and the risk of new-onset seizures. J Clin Psychiatry 81(4):11893
Man KKC et al (2020) Association between methylphenidate treatment and risk of seizure: a population-based, self-controlled case-series study. Lancet Child Adolesc Health 4(6):435–443
Andrade C (2020) Methylphenidate and the risk of new-onset seizures. J Clin Psychiatry. https://doi.org/10.4088/JCP.20f13586
Al Omairi NE et al (2022) Selenium nanoparticles with prodigiosin rescue hippocampal damage associated with epileptic seizures induced by pentylenetetrazole in rats. Biology 11(3):354
Nanoparticles as Drug Delivery Systems (2023) A Review of the Implication of nanoparticles’ physicochemical properties on responses in biological systems. Polymers 15(7):1596
Gelperina S et al (2005) The potential advantages of nanoparticle drug delivery systems in chemotherapy of tuberculosis. Am J Respir Crit Care Med 172(12):1487–1490
Gupta J et al (2019) Nanoparticle formulations in the diagnosis and therapy of Alzheimer’s disease. Int J Biol Macromol 130:515–526
Huo X et al (2019) A novel synthesis of selenium nanoparticles encapsulated PLGA nanospheres with curcumin molecules for the inhibition of amyloid β aggregation in Alzheimer’s disease. J Photochem Photobiol B 190:98–102
Barua S, Mitragotri S (2014) Challenges associated with penetration of nanoparticles across cell and tissue barriers: a review of current status and future prospects. Nano Today 9(2):223–243
Bennewitz MF, Saltzman WM (2009) Nanotechnology for delivery of drugs to the brain for epilepsy. Neurotherapeutics 6(2):323–336
Shringarpure M et al (2021) Management of epileptic disorders using nanotechnology-based strategies for nose-to-brain drug delivery. Expert Opin Drug Deliv 18(2):169–185
Bonilla L et al (2022) Biodegradable nanoparticles for the treatment of Epilepsy: from current advances to future challenges. Epilepsia Open 7(Suppl 1):S121-s132
Praveen A et al (2019) Lamotrigine encapsulated intra-nasal nanoliposome formulation for epilepsy treatment: formulation design, characterization and nasal toxicity study. Colloids Surf B Biointerfaces 174:553–562
Ali O et al (2022) Nanotechnological advances in the treatment of epilepsy. CNS Neurol Disord Drug Targets 21(10):994–1003
Raman S, Kannan R, Vimal J (2016) Mono disperse polymer nanoparticles, functionalized nanoparticles and controlled formation method. United States patent application US 15/030,755. 2016 Aug 25
Nagare S, Senna M (2006) Drug nano-particle, method and apparatus for preparing pharmaceutical preparation using the particle. United States patent application US 10/560,030. 2006 Nov 9
Javed M, Pottoo F, Alam M (2016) Metallic nanoparticle alone and/or in combination as novel agent for the treatment of uncontrolled electric conductance related disorders and/or seizure, epilepsy & convulsions. Patent Acquir Oct 10:40
Nezamoleslami S et al (2020) Glatiramer acetate attenuates renal ischemia reperfusion injury in rat model. Exp Mol Pathol 112:104329
Motaghinejad M, Motevalian M, Shabab B (2016) Effects of chronic treatment with methylphenidate on oxidative stress and inflammation in hippocampus of adult rats. Neurosci Lett 619:106–113
Jamshidi D, Sazegar MR (2020) Antibacterial activity of a novel biocomposite chitosan/graphite based on zinc-grafted mesoporous silica nanoparticles. Int J Nanomed 15:871–883
Sadasivan S et al (2012) Methylphenidate exposure induces dopamine neuron loss and activation of microglia in the basal ganglia of mice. PLoS ONE 7(3):e33693
Meurs A et al (2008) Seizure activity and changes in hippocampal extracellular glutamate, GABA, dopamine and serotonin. Epilepsy Res 78(1):50–59
Mumtaz F et al (2020) Involvement of nNOS, and α1, α2, β1, and β2 subunits of Soluble Guanylyl cyclase genes expression in anticonvulsant effect of sumatriptan on pentylenetetrazole-induced seizure in mice. Iran J Pharm Res 19(4):181–192
Verghese C, Abdijadid S (2021) Methylphenidate, in StatPearls [Internet]. StatPearls Publishing.
Kiviranta AM et al (2013) Topiramate as an add-on antiepileptic drug in treating refractory canine idiopathic epilepsy. J Small Anim Pract 54(10):512–520
Mula M, Cavanna AE, Monaco F (2006) Psychopharmacology of topiramate: from epilepsy to bipolar disorder. Neuropsychiatr Dis Treat 2(4):475
Zhang J, Zhan P, Tian H (2021) Recent updates in the polysaccharides-based Nano-biocarriers for drugs delivery and its application in diseases treatment: a review. Int J Biol Macromol 182:115–128
Kiaie SH et al (2023) Nano-immunotherapy: overcoming delivery challenge of immune checkpoint therapy. J Nanobiotechnol 21(1):339
Wohlfart S et al (2011) Kinetics of transport of doxorubicin bound to nanoparticles across the blood–brain barrier. J Control Release 154(1):103–107
Almeida JMFd et al (2021) pH-responsive release system of topiramate transported on silica nanoparticles by melting method. Drug Dev Ind Pharm 47(1):126–145
Foschiera LN, Schmitz F, Wyse AT (2022) Evidence of methylphenidate effect on mitochondria, redox homeostasis, and inflammatory aspects: insights from animal studies. Prog Neuropsychopharmacol Biol Psychiatry 116:110518
Alvarez-Arellano L et al (2020) Antioxidants as a potential target against inflammation and oxidative stress in attention-deficit/hyperactivity disorder. Antioxidants 9(2):176
Andreazza AC et al (2007) DNA damage in rats after treatment with methylphenidate. Prog Neuropsychopharmacol Biol Psychiatry 31(6):1282–1288
Gomes KM et al (2008) Antioxidant enzyme activities following acute or chronic methylphenidate treatment in young rats. Neurochem Res 33(6):1024–1027
Raoofi A et al (2020) Toxicology of long-term and high-dose administration of methylphenidate on the kidney tissue–a histopathology and molecular study. Toxicol Mech Methods 30(8):611–619
Silva I et al (2022) Anti-inflammatory effect of topiramate in a chronic model of TNBS-induced colitis. Int J Mol Sci. https://doi.org/10.3390/ijms23169127
Armagan A et al (2008) Topiramate and vitamin e modulate antioxidant enzyme activities, nitric oxide and lipid peroxidation levels in pentylenetetrazol-induced nephrotoxicity in rats. Basic Clin Pharmacol Toxicol 103(2):166–170
Cárdenas-Rodríguez N et al (2013) Antioxidant activity of topiramate: an antiepileptic agent. Neurol Sci 34(5):741–747
Patel RJ, Parikh RH (2020) Intranasal delivery of topiramate nanoemulsion: pharmacodynamic, pharmacokinetic and brain uptake studies. Int J Pharm 585:119486
Acknowledgements
The authors would like to thank Dr Khalili and Dr Amri for their assistance.
Funding
This work has received financial support from the research deputy of Iran University of Medical Sciences, Tehran, Iran (Grant No: 19568).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by E.P., M.S., M.S. and M.M.: Experimental steps were performed by E.P., M.S., A.M., and S.M.: The first draft of the manuscript was written by M.K. and M.S. Critical revisions were made by M.S. and M.M. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict interest
The authors declare no conflicts of interest.
Ethical approval
The protocol of this study was approved by the Ethics Committee of Iran University of Medical Sciences (Ethics code: IR.IUMS.REC.1400.544).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pari, E., Sheibani, M., Sazegar, M.R. et al. Comparison of neuroprotective effects of a topiramate-loaded biocomposite based on mesoporous silica nanoparticles with pure topiramate against methylphenidate-induced neurodegeneration. Mol Biol Rep 51, 65 (2024). https://doi.org/10.1007/s11033-023-09011-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11033-023-09011-1