Abstract
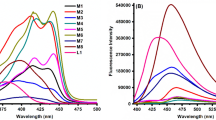
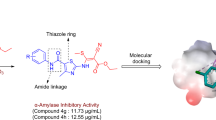
In search of 1,2,4-triazole-based antidiabetic agents, new mixed-ligand chelates of the type, [M(L)(H2O)3] (where M = Co(II), Ni(II), Cu(II) and Zn(II)), were synthesized using 2-(((3-mercapto-5-(pyridin-3-yl)-4H-1,2,4-triazol-4-yl)imino)methyl)-4-nitrophenol (H2L1) ligand and the corresponding metal acetates in 1:1 molar ratio. FTIR, NMR, mass, UV–Vis, elemental analyses, conductivity measurements, ESR, XRD, TGA, SEM, and EDAX studies were used to determine the nature of bonding, coordination characteristics and the stability of compounds. Spectral and physico-analytical studies suggest that the ligand functioned in a binegative tridentate manner with phenolic-O, azomethine-N and thiol-S as the coordinating sites and an octahedral geometry was proposed for the metal complexes. The quantum computational calculations of the synthesized compounds were executed in the ground state using DFT/B3LYP level with 6-311++G as basis set. In vitro alpha-amylase and alpha-glucosidase inhibitory assay revealed moderate to good results for the synthesized compounds and especially the complex 3 against alpha-amylase and complex 4 against alpha-glucosidase were found to be effective inhibitors. Additionally, the compounds were explored for molecular docking studies against the human pancreatic alpha-amylase (PDB id: 1BSI) and alpha-glucosidase (PDB id: 5ZCC). The molecular docking calculations of complex 3 into 1BSI revealed a binding energy of − 8.6 kcal/mol, whereas that of complex 4 against 5ZCC resulted in a binding energy of − 7.4 kcal/mol respectively, both of which exceeds that of the standard Acarbose.
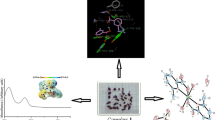
Graphical Abstract
New Schiff base ligand and its transition metal chelates were synthesized. Furthermore, the synthesized compounds were examined for in vitro alpha-amylase and alpha-glucosidase inhibitory activity.










Similar content being viewed by others
Data Availability
No data was used for the research described in the article.
References
M. Dioguardi, S. Cantore, S. Scacco, C. Quarta, D. Sovereto, F. Spirito, M. Alovisi, G. Troiana, R. Aiuto, D. Garcovich, V. Crincoli, L. Laino, M. Covelli, A. Malcangi, L.L. Muzio, A. Ballini, M. Di Cosola, J. Pers. Med. 12, 235 (2022)
L. Deswal, V. Verma, D. Kumar, Y. Deswal, A. Kumar, R. Kumar, M. Parshad, M. Bhatia, Chem. Pap. 76, 7607 (2022)
H. Sun, P. Saeedi, S. Karuranga, M. Pinkepank, K. Ogurtsova, B.B. Duncan, C. Stein, A. Chan, C. Basit, J.C. Mbanya, M.E. Pavkov, A. Ramchandran, S.H. Wild, S. James, W.H. Herman, P. Zhang, C. Boomer, S. Kuo, E. Boyko, D.J. Magliano, Diabetes Res. Clin. Pract. 183, 109119 (2022)
L. Deswal, V. Verma, D. Kumar, C.P. Kaushik, A. Kumar, Y. Deswal, S. Punia, Arch. Pharm. 353, 2000090 (2020)
W.S. Azar, R. Njeim, A.H. Fares, N.S. Azar, S.T. Azar, M. El Sayed, A.A. Eid, Rev. Endocr. Metab. Disord. 21, 451 (2020)
A. Kumari, C. Singh, J. Drug Deliv. Ther. 9, 1123 (2019)
Y. Liang, M. Li, Y. Yang, L. Qiao, H. Xu, B. Guo, ACS Nano 16, 3194 (2022)
A. Ito, I. Horie, M. Miwa, A. Sako, T. Niri, Y. Nakashima, R. Shigeno, A. Haraguchi, S. Natsuda, S. Akazawa, A. Kamada, A. Kawakami, N. Abriu, J. Diabetes Investig. 12, 1367 (2021)
Y. Deswal, S. Asija, A. Dubey, L. Deswal, D. Kumar, D.K. Jindal, J. Devi, J. Mol. Struct. 1253, 132266 (2022)
T.C. Farias, H.Y. Kawaguti, M.G.B. Koblitz, Biocat. Agric Biotech. 35, 102054 (2021)
L. Deswal, V. Verma, J.S. Kirar, D. Kumar, Y. Deswal, A. Kumar, M. Bhatia, Res. Chem. Intermed. 49, 1059 (2023)
P. Agarwal, S. Asija, Y. Deswal, N. Kumar, J. Indian Chem. Soc. 99, 100556 (2022)
A.S. Dabhi, N.R. Bhatt, M.J. Shah, J. Clin. Diagnostic Res. 7, 3023 (2013)
T. Fujisawa, H. Ikegami, K. Inoue, Y. Kawabata, T. Ogihara, Metabolism 54, 387 (2005)
O.M. Ogunyemi, A.G. Gyebi, J.O. Adebayo, J.A. Oguntola, C.O. Olaiya, S.N. Appl, Sci. 2, 1 (2020)
G. Lopes, M. Barbosa, P.B. Andrade, P. Valentao, J. Appl. Phycol. 31, 3143 (2019)
Y. Deswal, S. Asija, D. Kumar, D.K. Jindal, G. Chandan, V. Panwar, S. Saroya, N. Kumar, Res. Chem. Intermed. 48, 703 (2022)
W. Zafar, S.H. Sumrra, Z.H. Chohan, Eur. J. Med. Chem. 222, 113602 (2021)
E. Stingaci, M. Zveaghinteva, S. Pogrebnoi, L. Lupascu, V. Valica, L. Uncu, A. Smetanscaia, M. Drumea, A. Petrou, A. Ciric, J. Glamoclija, Bioorg. Med. Chem. Lett. 30, 127368 (2020)
H.A. El-Sherief, B.G. Youssif, S.N.A. Bukhari, A.H. Abdelazeem, M. Abdel-Aziz, H.M. Abdel-Rahman, Eur. J. Med. Chem. 156, 774 (2018)
T.T. Calam, Int. J. Environ. Anal. Chem. 99, 1298 (2019)
B. Kapron, J.J. Luszczki, A. Płazińska, A. Siwek, T. Karcz, A. Grybos, G. Nowak, A. Makuch, K. Walczak, E. Langner, K. Szalast, Eur. J. Pharm. Sci. 129, 42 (2019)
D. Dixit, P.K. Verma, R.K. Marwaha, J. Iran. Chem. Soc. 18, 2535 (2021)
M.A. Malik, O.A. Dar, P. Gull, M.Y. Wani, A.A. Hashmi, MedChemComm 9, 409 (2018)
N. Kumar, S. Asija, Y. Deswal, S. Saroya, A. Kumar, Res. Chem. Intermed. 48, 5133 (2022)
N. Kumar, S. Asija, Y. Deswal, S. Saroya, A. Kumar, J. Devi, Phosphorus Sulfur Silicon Relat. Elem. 197, 952 (2022)
S. Saroya, S. Asija, N. Kumar, Y. Deswal, J. Indian Chem. Soc. 99, 100379 (2022)
S. Sharma, S. Gupta, A.K. Narula, Indian J. Chem. Sect. A 33A, 1119 (1994)
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian 16 Revision B.01 (Gaussian Inc, Wallingford CT, 2010)
B. Miehlich, A. Savin, H. Stoll, H. Preuss, Chem. Phys. Lett. 157, 200 (1989)
C. Lee, W. Yang, R.G. Parr, Phys. Rev. B 37, 785 (1988)
S. Khokhar, Y. Feng, M.R. Campitelli, R.J. Quinn, J.N. Hooper, M.G. Ekins, R.A. Davis, J. Nat. Prod. 76, 2100 (2013)
E.H. Rydberg, G. Sidhu, H.C. Vo, J. Hewitt, H.C. Côte, Y. Wang, S. Numao, R.T. MacGillivray, C.M. Overall, G.D. Brayer, S.G. Withers, Protein Sci. 8, 635 (1999)
W. Auiewiriyanukul, W. Saburi, K. Kato, M. Yao, H. Mori, FEBS Lett. 592, 2268 (2018)
P.W. Rose, A. Prlic, C. Bi, W.F. Bluhm, C.H. Christie, S. Dutta, R.K. Green, D.S. Goodsell, J.D. Westbrook, J. Woo, J. Young, C. Zardecki, H.M. Berman, P.E. Bourne, S.K. Burley, Nucleic Acids Res. 43, 345 (2015)
G.M. Sastry, M. Adzhigirey, T. Day, R. Annabhimoju, W. Sherman, J. Comput. Aided Mol. Des. 27, 221 (2013)
A. Dubey, A. Marabotti, P.W. Ramteke, A. Facchiano, Future Med. Chem. 8, 841 (2016)
A. Dubey, A. Marabotti, P.W. Ramteke, A. Facchiano, Biochem. Biophys. Res. Commun. 473, 449 (2016)
S. Bharadwaj, A. Dubey, N.K. Kamboj, A.K. Sahoo, S.G. Kang, U. Yadava, Sci. Rep. 11, 10169 (2021)
A. Dubey, S. Dotolo, P.W. Ramteke, A. Facchiano, A. Marabotti, Biomolecules 9, 5 (2019)
B. Kumar, J. Devi, A. Manuja, Res. Chem. Intermed. 49, 2277 (2023)
Z. Xiao, R. Storms, A. Tsang, Anal. Biochem. 351, 146 (2006)
J.B. Xiao, P. Hogger, Curr. Med. Chem. 22, 23 (2015)
M.A. Neelakantan, S.S. Marriappan, J. Dharmaraja, T. Jeyakumar, K. Muthuku-maran, Spectrochim. Acta-A: Mol. Biomol Spectrosc. 71, 628 (2008)
V.P. Radha, S. Chitra, S. Jonekirubavathi, I.M. Chung, S.H. Kim, M. Prabakaran, J. Coord. Chem. 73, 1009 (2020)
S. Saroya, S. Asija, Y. Deswal, N. Kumar, D. Kumar, D.K. Jindal, P. Puri, S. Kumar, Res. Chem. Intermed. 11, 4671 (2022)
V.V. Dhayabaran, T.D. Prakash, R. Renganathan, E. Friehs, D.W. Bahnemann, J. Fluoresc. 27, 135 (2017)
A.A. Olanrewaju, F.S. Fabiyi, C.U. Ibeji, E.G. Kolawole, R. Gupta, J. Mol. Struct. 1211, 128057 (2020)
B. Kumari, K. Singh, A. Sharma, Chem. Data Coll. 38, 100833 (2022)
B.U. Gauthama, B. Narayana, B.K. Sarojini, N.K. Suresh, Y. Sangappa, A.K. Kudva, G. Satyanarayana, S.V. Raghu, Microchem. J. 166, 106233 (2021)
B.J. Okoli, J.S. Modise, Antioxidants 7, 113 (2018)
M. Azam, S.M. Wabaidur, M. Alam, Z. Khan, I.O. Alanazi, S.I. Al-Resayes, I.S. Moon, Transit. Met. Chem. 46, 65 (2021)
A.K. Singh, O.P. Pandey, S.K. Sengupta, Spectrochim. Acta-A: Mol. Biomol. 113, 393 (2013)
O.A. El-Gammal, A.A. El-Bindary, F.S. Mohamed, G.N. Rezk, M.A. El-Bindary, J. Mol. Liq. 346, 117850 (2022)
S. Saroya, S. Asija, Y. Deswal, N. Kumar, A. Kumar, Res. Chem. Intermed. 48, 2949 (2022)
M. Chaurasia, D. Tomar, S. Chandra, J. Mol. Struct. 1179, 431 (2021)
W.J. Geary, Coord. Chem. Rev. 7, 81 (1971)
W.H. Mahmoud, R.G. Deghadi, G.G. Mohamed, J. Therm. Anal. Calorim. 127, 2149 (2017)
B.J. Hathaway, A.A.G. Tomilinson, Coord. Chem. Rev. 5, 1 (1970)
M.A. Diab, G.G. Mohamed, W.H. Mahmoud, A.Z. ElSonbati, S.M. Morgan, S.Y. Abbas, Appl. Organomet. Chem. 33, 4945 (2019)
W.H. El-Shwiniy, A.G. Ibrahim, S.A. Sadeek, W.A. Zordok, Appl. Organomet. Chem. 35, 6174 (2021)
M.S. Refat, I.M. El-Deen, H.K. Ibrahim, S. El-Ghool, Spectrochim. Acta-A: Mol. Biomol. 65, 1208 (2006)
C.G. Zhan, J.A. Nichols, D.A. Dixon, J. Phys. Chem. A 107, 4184 (2003)
T.L. Yusuf, S.D. Oladipo, S. Zamisa, H.M. Kumalo, I.A. Lawal, M.M. Lawal, N. Mabuba, ACS Omega 6, 13704 (2021)
P.K. Vishwakarma, J.M. Mir, R.C. Maurya, J. Chem. Sci. 128, 511 (2016)
M.M. Lawal, T. Govender, G.E. Maguire, H.G. Kruger, B. Honarparvar, Int. J. Quantum Chem. 118, 25497 (2018)
M.M. Lawal, T. Govender, G.E. Maguire, B. Honarparvar, H.G. Kruger, J. Mol. Model. 22, 235 (2016)
S.N. Shukla, P. Gaur, M.L. Raidas, B. Chaurasia, J. Mol. Struct. 1202, 127362 (2020)
J.S. Kirar, S. Khare, N. Tiwari, ChemistrySelect 6, 11557 (2021)
Y. Deswal, S. Asija, A. Tufail, A. Dubey, L. Deswal, N. Kumar, S. Saroya, J.S. Kirar, N.M. Gupta, Appl. Organomet. Chem. 37, 7050 (2023)
S. Philip, E.G. Jayasree, K. Mohanan, Res. Chem. Intermed. 46, 75 (2020)
M. Sohrabi, M.R. Binaeizadeh, A. Iraji, B. Larijani, M. Saeedi, M. Mahdavi, RSC Adv. 12, 12011 (2022)
Acknowledgements
One of the authors (Yogesh Deswal) is highly thankful to Dr. APJ Abdul Kalam Central Instrumentation Laboratory (CIL), GJUS&T, Hisar for providing assistance in characterization of compounds.
Funding
Council of Scientific & Industrial Research (CSIR), New Delhi, India (Ref. No.: 9/752(0087)/2018-EMR-I).
Author information
Authors and Affiliations
Contributions
YD: Writing-original draft, Conceptualization, Methodology, Investigation, Validation, Writing-review & editing, Visualization, Funding acquisition, Data curation. SA: Supervision, Formal analysis, Validation. AT: DFT and molecular docking calculations. AD: DFT and molecular docking calculations. LD: Writing-review & editing, Data curation, Validation. NK: Data curation. JS Kirar: Investigation. NM Gupta: Formal analysis. PB: Investigation.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Ethical Approval
Not applicable.
Consent for Publication
Not applicable.
Consent to Participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deswal, Y., Asija, S., Tufail, A. et al. Metal Complexes of 1,2,4-Triazole Based Ligand: Synthesis, Structural Elucidation, DFT Calculations, Alpha-Amylase and Alpha-Glucosidase Inhibitory Activity Along with Molecular Docking Studies. J Inorg Organomet Polym 34, 144–160 (2024). https://doi.org/10.1007/s10904-023-02808-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-023-02808-4