Abstract
Background
REarranged during Transfection (RET) gene fusion is one of the common oncogenic variants detectable in non-small cell lung cancer (NSCLC). The feature of most oncogenic RET gene fusion cases is that RET tyrosine kinase domain is retained in fusions and the partner gene includes a coiled-coil or LIS1 homology domain. However, only a few studies reported about the exceptional form of RET fusion in NSCLC so far.
Methods
Targeted next-generation sequencing (NGS) and fluorescence in situ hybridization (FISH) were performed on resected cancerous tissue.
Results
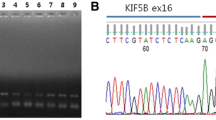
A rare form of RET fusion was identified in a 45 year-old Chinese female patient, in which exon 1–4 of LDLR fused with exon 12–21 of RET. The result was validated by FISH.
Conclusions
This novel form of RET fusion in NSCLC is reported for the first time worldwide, offering a new treatment option for the patient with the possibility of using RET-selective inhibitors.

Similar content being viewed by others
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request
Abbreviations
- NSCLC:
-
nonsmall cell lung cancer.
- FFPE:
-
formalin fixed paraffin embedded.
- MKIs:
-
multikinase inhibitors.
- FISH:
-
fluorescent in-situ hybridization.
- IHC:
-
immunohistochemistry.
- TKIs:
-
tyrosine kinase inhibitors.
References
Markham A (2020) Selpercatinib: first approval. Drugs 80(11):1119–1124
Ou SHI, Zhu VW (2020) Catalog of 5′ fusion partners in RET + NSCLC circa 2020. JTO Clin Res Rep 1(2):100037
Ferrara R, Auger N, Auclin E et al (2018) Clinical and translational implications of RET rearrangements in non–small cell lung cancer. J Thorac Oncol 13(1):27–45
Acknowledgements
The authors thank the patient and her families for agreeing to use her data for research purposes, and specifically, for publication of this report.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Guanglin Shi drafted the Original manuscript, Yuyan Luo collected clinical data, Zhaonan Yu did bioinformatics analysis, Yue Wang drew the diagram and Bojin Zhu did supervision, writing- reviewing and editing .
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The research program was approved by the Research Ethics Committee of The Prople’s Hosptial of Hai’An. I confirm the relevant ethics statement has been included in the manuscript, where applicable.
Consent for publication
The patient signed an informed consent form.
Conflict of interest
The authors declare that they have no competing interests.
Disclosure of potential conflicts of interest
The authors declare that they have no potential conflicts of interest.
Research involving Human Participants and/or Animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The patient signed an informed consent form.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shi, G., Luo, Y., Yu, Z. et al. Identifification of a novel LDLR-RET Fusion in Lung Adenocarcinoma. Invest New Drugs 40, 858–860 (2022). https://doi.org/10.1007/s10637-022-01246-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-022-01246-2