Abstract
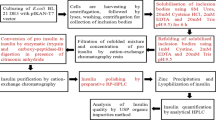
To reduce the immunogenicity of β-lactoglobulin (BLG), we prepared single amino acid substituted recombinant BLG mutants (BLG/P126A, BLG/V128D and BLG/D129A) in the methylotrophic yeast Pichia Pastris by fusion of the cDNA to the sequence coding for the α-factor signal peptide from Saccharomyces cerevisiae. Isoelectric points of single amino acid substituted BLGs were lower than that of native BLG. CD spectra indicated that the secondary structure of BLG had maintained native structure in single amino acid substituted BLGs. Fluorescence studies indicated that the conformation around Trp had not changed in single amino acid substituted BLGs. Anti-BLG antibody response was evaluated after immunization to C57BL/6 mice. Antibody response was reduced after immunization with BLG/P126A, BLG/V128D and BLG/D129A. And novel immunogenicity was not observed in the experiments. T cell proliferative response was evaluated in C57BL/6 mice, and it was clarified that BLG mutants also showed low response. Methods employed in this study was considered to be very effective to reduce immunogenicity of BLG.








Similar content being viewed by others
Data availability
The data underlying this article are available in the article and also from the corresponding author upon request.
Abbreviations
- BLG:
-
β-Lactoglobulin
- wt:
-
Wild-type
- SDS–PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- PBS:
-
Phosphate-buffered saline
- mAb:
-
Monoclonal antibody
References
Armstrong JM, McKenzie HA, Sawyer WH (1967) On the fractionation of beta-lactoglobulin and alpha-lactalbumin. Biochim Biophys Acta 147:60–72. https://doi.org/10.1016/0005-2795(67)90090-6
Brownlow S, Cabral JH, Cooper R, Flower DR, Yewdall SJ, Polikarpov I, North AC, Sawyer L (1997) Bovine β-lactoglobulin at 1.8 Å resolution–still an enigmatic lipocalin. Structure 5:481–495. https://doi.org/10.1016/s0969-2126(97)00205-0
Chen FM, Lee JH, Yang YH, Lin YT, Wang LC, Yu HH, Chiang BL (2014) Analysis of α-lactalbumin-, β-lactoglobulin-, and casein-specific IgE among children with atopic diseases in a tertiary medical center in northern Taiwan. J Microbiol Immunol Infect 47:130–136. https://doi.org/10.1016/j.jmii.2012.08.009
Cogan U, Kopelman M, Mokady S, Shintzky M (1976) Binding affinities of retinol and related compounds to retinol binding proteins. Eur J Biochem 65:71–78. https://doi.org/10.1111/j.1432-1033.1976.tb10390.x
Flower DR (1996) The lipocalin protein family: structure and function. Biochem J 318:1–14. https://doi.org/10.1042/bj3180001
Futterman S, Heller J (1972) The enhancement of fluorescence and decreased susceptibility to enzyme oxidation of retinol complexed with bovine serum albumin, β-lactoglobulin, and the retinol-binding protein of human plasma. J Biol Chem 247:5168–5172. https://doi.org/10.1016/S0021-9258(19)44953-3
Hattori M, Ametani A, Katakura Y, Shimizu M, Kaminogawa S (1993) Unfolding/refolding studies on bovine β-lactoglobulin with monoclonal antibodies as probes: does a renatured protein completely refold? J Biol Chem 268:22414–22419. https://doi.org/10.1016/S0021-9258(18)41545-1
Hattori M, Nagasawa K, Ametani A, Kaminogawa S, Takahash K (1994) Functional changes in β-lactoglobulin by conjugation with carboxymethyl dextran. J Agric Food Chem 42:2120–2125. https://doi.org/10.1021/jf00046a009
Iikura Y, Imai Y, Imai T, Akasawa A, Fujita K, Hoshiyama K, Nakura H, Kohno Y, Koike K, Okudaira H, Iwasaki E (1999) Frequency of immediate-type food allergy in children in Japan. Int Arch Allergy Immunol 118:251–252. https://doi.org/10.1159/000024089
Kaminogawa S (1996) Food allergy, oral tolerance and immunization—their molecular and cellular mechanism. Biosci Biotechnol Biochem 60:1749–1756. https://doi.org/10.1271/bbb.60.1749
Kramlova M, Printstoupil T, Kraml J (1986) First experience with the use of the Pharmacia PhastSystem for the characterization of hemoglobulins by isoelectric focusing. J Chromatogr 367:443–445. https://doi.org/10.1016/S0021-9673(00)94868-4
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
McKenzie HA (1971) β-Lactoglobulins chemistry and molecular biology. In: Mckenzie HA (ed) Milk proteins. Academic Press, New York, pp 257–330
Mulvihill DM, Kinsella JE (1987) Gelation characteristics of whey proteins and β-lactoglobulin. Food Technol 41:102–111
Papiz MZ, Sawyer L, Eliopoulos EE, North AC, Findlay JB, Sivaprasadarao R, Jones TA, Newcomer ME, Kraulis PJ (1986) The structure of β-lactoglobulin and its similarity to plasma retinol-binding protein. Nature 324:383–385. https://doi.org/10.1038/324383a0
Shimizu M, Saito M, Yamauchi K (1985) Emulsifying and structural properties of β-lactoglobulin at different pHs. Agric Biol Chem 49:189–194. https://doi.org/10.1271/bbb1961.49.189
Toda M, Totsuka M, Furukawa S, Yokota K, Yoshioka T, Ametani A, Kaminogawa S (2000) Down-regulation of antigen-specific antibody production by TCR antagonist peptides in vivo. Eur J Immunol 30:403–414. https://doi.org/10.1002/1521-4141(200002)30:2%3c403::AID-IMMU403%3e3.0.CO;2-8
Totsuka M, Katakura Y, Shimizu M, Kumagai I, Miura K, Kaminogawa S (1990) Expression and secretion of bovine β-lactoglobulin in Saccharomyces cerevisiae. Agric Biol Chem 54:3111–3116. https://doi.org/10.1271/bbb1961.54.3111
Totsuka M, Furukawa S, Sato E, Ametani A, Kaminogawa S (1997) Antigen-specific inhibition of CD4+ T-cell responses to beta-lactoglobulin by its single amino acid-substituted mutant form through T-cell receptor antagonism. Cytotechnology 25:115–126. https://doi.org/10.1023/A:1007934832359
Virtanen T, Zeiler T, Rsutinainen J, Mäntyjärvi R (1999) Allergy to lipocalins: a consequence of misguided T-cell recognition of self and nonself? Immunol Today 20:398–400. https://doi.org/10.1016/s0167-5699(99)01515-7
Waniska RD, Kinsella JE (1988) Foaming and emulsifying properties of glycosylated β-lactoglobulin. Food Hydrocoll 2:439–449. https://doi.org/10.1016/S0268-005X(88)80044-4
Acknowledgements
This work was supported in part by JSPS KAKENHI Grant Number JP17580107.
Author information
Authors and Affiliations
Contributions
TY, MT: Conceptualization, Funding acquisition, Writing manuscript, KY: Conceptualization, Data curation, Writing manuscript, CK, AS, FY, NT, RK: Data curation, Writing manuscript, MH: Conceptualization, Writing manuscript, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, T., Kume, C., Sachi, A. et al. Reduced immunogenicity of β-lactoglobulin by single amino acid substitution. Cytotechnology 74, 623–634 (2022). https://doi.org/10.1007/s10616-022-00549-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-022-00549-9