Abstract
Purpose
To explore the correlation between shear-wave elastography (SWE) parameters and pathological profiles of invasive breast cancer.
Methods
A total of 197 invasive breast cancers undergoing preoperative SWE and primary surgical treatment were included. Maximum elastic modulus (Emax), mean elastic modulus (Emean), and elastic modulus standard deviation (Esd) were calculated by SWE. Pathological profile was gold standard according to postoperative pathology. The relationship between SWE parameters and pathological factors were analyzed using univariate and multivariate analysis.
Results
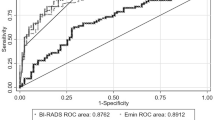
In univariate analysis, large cancers showed significantly higher Emax, Emean and Esd (all P < 0.001). Emax and Esd in the group of histological grade III were higher than those in the group of grade I (both P < 0.05). Invasive lobular carcinomas (ILC) showed higher Emean than invasive ductal carcinoma (IDC) (P < 0.001). Lymphovascular invasion (LVI) group showed higher Emax values than negative group (P < 0.05). Emax, Emean and Esd of the Ki-67 positive group presented higher values than negative group (all P < 0.05). Androgen receptor (AR) positive lesions had lower Esd than AR negative lesions (P < 0.05). In multivariate analysis, invasive size independently influenced Emax (P < 0.001). Invasive size and pathological type both independently influenced Emean (both P < 0.001). Invasive size and AR status were both independently influenced Esd (both P < 0.05).
Conclusion
SWE parameters correlated with pathological profiles of invasive breast cancer.In particular, AR positive group showed significantly low Esd than negative group.


Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
Abbreviations
- SWE:
-
Shear-wave elastography
- Emax:
-
Maximum elastic modulus
- Emean:
-
Mean elastic modulus
- Esd:
-
Elastic modulus standard deviation
- ILC:
-
Invasive lobular carcinomas
- IDC:
-
Invasive ductal carcinoma
- LVI:
-
Lymphovascular invasion
- AR:
-
Androgen receptor
- TNBC:
-
Triple negative breast cancer
- FOV:
-
Field of view
- ROI:
-
Regions of interest
- TILs:
-
Tumor infiltrating lymphocytes
- FISH:
-
Fluorescence in situ hybridization
- ECM:
-
Extracellular matrix
- AFM:
-
Atomic force microscopy
- CSC:
-
Cancer stem-like cells
References
Berg WA, Cosgrove DO, Dore CJ, Schafer FK, Svensson WE, Hooley RJ et al (2012) Shear-wave elastography improves the specificity of breast US: the BE1 multinational study of 939 masses. Radiology 262:435–449
Lee SH, Chang JM, Kim WH, Bae MS, Seo M, Koo HR et al (2014) Added value of shear-wave elastography for evaluation of breast masses detected with screening US imaging. Radiology 273:61–69
Chang JM, Park IA, Lee SH, Kim WH, Bae MS, Koo HR et al (2013) Stiffness of tumours measured by shear-wave elastography correlated with subtypes of breast cancer. Eur Radiol 23:2450–2458
Youk JH, Gweon HM, Son EJ, Kim JA, Jeong J (2013) Shear-wave elastography of invasive breast cancer: correlation between quantitative mean elasticity value and immunohistochemical profile. Breast Cancer Res Treat 138:119–126
Liu C, Zhou J, Chang C, Zhi W (2021) Feasibility of shear wave elastography imaging for evaluating the biological behavior of breast cancer. Front Oncol 11:820102
Ganau S, Andreu FJ, Escribano F, Martin A, Tortajada L, Villajos M et al (2015) Shear-wave elastography and immunohistochemical profiles in invasive breast cancer: evaluation of maximum and mean elasticity values. Eur J Radiol 84:617–622
Vasiliou SK, Diamandis EP (2019) Androgen receptor: A promising therapeutic target in breast cancer. Crit Rev Clin Lab Sci 56:200–223
Yeh S, Hu YC, Wang PH, Xie C, Xu Q, Tsai MY et al (2003) Abnormal mammary gland development and growth retardation in female mice and MCF7 breast cancer cells lacking androgen receptor. J Exp Med 198:1899–1908
Simanainen U, Gao YR, Walters KA, Watson G, Desai R, Jimenez M et al (2012) Androgen resistance in female mice increases susceptibility to DMBA-induced mammary tumors. Horm Cancer 3:113–124
Missmer SA, Eliassen AH, Barbieri RL, Hankinson SE (2004) Endogenous estrogen, androgen, and progesterone concentrations and breast cancer risk among postmenopausal women. J Natl Cancer Inst 96:1856–1865
Gucalp A, Tolaney S, Isakoff SJ, Ingle JN, Liu MC, Carey LA et al (2013) Phase II trial of bicalutamide in patients with androgen receptor-positive, estrogen receptor-negative metastatic Breast cancer. Clin Cancer Res 19:5505–5512
Traina TA, Miller K, Yardley DA, Eakle J, Schwartzberg LS, O’Shaughnessy J et al (2018) Enzalutamide for the treatment of androgen receptor-expressing triple-negative breast cancer. J Clin Oncol 36:884–890
Bae MS, Park SY, Song SE, Kim WH, Lee SH, Han W et al (2015) Heterogeneity of triple-negative breast cancer: mammographic, US, and MR imaging features according to androgen receptor expression. Eur Radiol 25:419–427
Huang Y, Wei L, Hu Y, Shao N, Lin Y, He S et al (2021) Multi-parametric MRI-based radiomics models for predicting molecular subtype and androgen receptor expression in breast cancer. Front Oncol 11:706733
Zhou J, Zhan W, Chang C, Zhang X, Jia Y, Dong Y et al (2014) Breast lesions: evaluation with shear wave elastography, with special emphasis on the “stiff rim” sign. Radiology 272:63–72
Dieci MV, Mathieu MC, Guarneri V, Conte P, Delaloge S, Andre F et al (2015) Prognostic and predictive value of tumor-infiltrating lymphocytes in two phase III randomized adjuvant breast cancer trials. Ann Oncol 26:1698–1704
Evans A, Whelehan P, Thomson K, McLean D, Brauer K, Purdie C et al (2012) Invasive breast cancer: relationship between shear-wave elastographic findings and histologic prognostic factors. Radiology 263:673–677
Au FW, Ghai S, Lu FI, Moshonov H, Crystal P (2015) Quantitative shear wave elastography: correlation with prognostic histologic features and immunohistochemical biomarkers of breast cancer. Acad Radiol 22:269–277
Evans A, Rauchhaus P, Whelehan P, Thomson K, Purdie CA, Jordan LB et al (2014) Does shear wave ultrasound independently predict axillary lymph node metastasis in women with invasive breast cancer? Breast Cancer Res Treat 143:153–157
Denis M, Gregory A, Bayat M, Fazzio RT, Whaley DH, Ghosh K et al (2016) Correlating tumor stiffness with immunohistochemical subtypes of breast cancers: prognostic value of comb-push ultrasound shear elastography for differentiating luminal subtypes. PLoS ONE 11:e0165003
Paramagul CP, Helvie MA, Adler DD (1995) Invasive lobular carcinoma: sonographic appearance and role of sonography in improving diagnostic sensitivity. Radiology 195:231–234
Hu XQ, Peng L, Wintermark M, Lipson JA, Zhang YR, Gao Y (2021) Shear wave elastography of invasive ductal carcinoma: correlations between shear wave velocity and histological prognostic factors. Curr Med Sci 41:173–179
Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A et al (2005) Tensional homeostasis and the malignant phenotype. Cancer Cell 8:241–254
Hoyt K, Castaneda B, Zhang M, Nigwekar P, di Sant’agnese PA, Joseph JV et al (2008) Tissue elasticity properties as biomarkers for prostate cancer. Cancer Biomark 4:213–225
Baker EL, Lu J, Yu D, Bonnecaze RT, Zaman MH (2010) Cancer cell stiffness: integrated roles of three-dimensional matrix stiffness and transforming potential. Biophys J 99:2048–2057
Lopez JI, Mouw JK, Weaver VM (2008) Biomechanical regulation of cell orientation and fate. Oncogene 27:6981–6993
Mouw JK, Yui Y, Damiano L, Bainer RO, Lakins JN, Acerbi I et al (2014) Tissue mechanics modulate microRNA-dependent PTEN expression to regulate malignant progression. Nat Med 20:360–367
Levental KR, Yu H, Kass L, Lakins JN, Egeblad M, Erler JT et al (2009) Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139:891–906
Ferraioli G, Tinelli C, Dal Bello B, Zicchetti M, Filice G, Filice C et al (2012) Accuracy of real-time shear wave elastography for assessing liver fibrosis in chronic hepatitis C: a pilot study. Hepatology 56:2125–2133
Juge L, Doan BT, Seguin J, Albuquerque M, Larrat B, Mignet N et al (2012) Colon tumor growth and antivascular treatment in mice: complementary assessment with MR elastography and diffusion-weighted MR imaging. Radiology 264:436–444
Chamming’s F, Latorre-Ossa H, Le Frere-Belda MA, Fitoussi V, Quibel T, Assayag F et al (2013) Shear wave elastography of tumour growth in a human breast cancer model with pathological correlation. Eur Radiol 23:2079–2086
Boyd NF, Rommens JM, Vogt K, Lee V, Hopper JL, Yaffe MJ et al (2005) Mammographic breast density as an intermediate phenotype for breast cancer. Lancet Oncol 6:798–808
Li T, Sun L, Miller N, Nicklee T, Woo J, Hulse-Smith L et al (2005) The association of measured breast tissue characteristics with mammographic density and other risk factors for breast cancer. Cancer Epidemiol Biomarkers Prev 14:343–349
Axelson H, Fredlund E, Ovenberger M, Landberg G, Pahlman S (2005) Hypoxia-induced dedifferentiation of tumor cells–a mechanism behind heterogeneity and aggressiveness of solid tumors. Semin Cell Dev Biol 16:554–563
Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF (2003) Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA 100:3983–3988
Pang MF, Siedlik MJ, Han S, Stallings-Mann M, Radisky DC, Nelson CM (2016) Tissue stiffness and hypoxia modulate the integrin-linked kinase ILK to control breast cancer stem-like cells. Cancer Res 76:5277–5287
Leng X, Japaer R, Zhang H, Yeerlan M, Ma F, Ding J (2021) Relationship of shear wave elastography anisotropy with tumor stem cells and epithelial-mesenchymal transition in breast cancer. BMC Med Imaging 21:171
Al-Othman N, Hammad H, Ahram M (2018) Dihydrotestosterone regulates expression of CD44 via miR-328-3p in triple-negative breast cancer cells. Gene 675:128–135
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y et al (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121:2750–2767
Park S, Koo JS, Kim MS, Park HS, Lee JS, Lee JS et al (2011) Androgen receptor expression is significantly associated with better outcomes in estrogen receptor-positive breast cancers. Ann Oncol 22:1755–1762
Kim H, Lee J, Kang BJ, Kim SH (2021) What shear wave elastography parameter best differentiates breast cancer and predicts its histologic aggressiveness? Ultrasonography 40:265–273
Funding
This work was supported by a grant from the National Natural Science Foundation of China Grant No. 82172025. Author Hong Lu has received research support. And it was also funded by Tianjin Key Medical Discipline(Specialty) Construction Project(TJYXZDXK-012A).
Author information
Authors and Affiliations
Contributions
Conceptualization: HL, JL; Data curation: BS; Formal analysis: YL; Investigation: SL; Methodology: HL, JW; Project administration: YZ, HL; Resources: JL, BS; Supervision: YZ; Validation: YL; BS; Roles/Writing—original draft: JL, BS Writing—review and editing: JL, HL.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Sun, B., Li, Y. et al. Correlation analysis between shear-wave elastography and pathological profiles in breast cancer. Breast Cancer Res Treat 197, 269–276 (2023). https://doi.org/10.1007/s10549-022-06804-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-022-06804-z