Abstract
Purpose
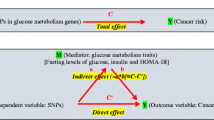
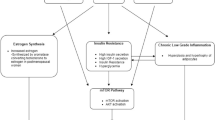
Genetic variants and traits in metabolic signaling pathways may interact with obesity, physical activity, and exogenous estrogen (E), influencing postmenopausal breast cancer risk, but these inter-related pathways are incompletely understood.
Methods
We used 75 single-nucleotide polymorphisms (SNPs) in genes related to insulin-like growth factor-I (IGF-I)/insulin resistance (IR) traits and signaling pathways, and data from 1003 postmenopausal women in Women’s Health Initiative Observation ancillary studies. Stratifying via obesity and lifestyle modifiers, we assessed the role of IGF-I/IR traits (fasting IGF-I, IGF-binding protein 3, insulin, glucose, and homeostatic model assessment-insulin resistance) in breast cancer risk as a mediator or influencing factor.
Results
Seven SNPs in IGF-I and INS genes were associated with breast cancer risk. These associations differed between non-obese/active and obese/inactive women and between exogenous E non-users and users. The mediation effects of IGF-I/IR traits on the relationship between these SNPs and cancer differed between strata, but only roughly 35% of the cancer risk due to the SNPs was mediated by traits. Similarly, carriers of 20 SNPs in PIK3R1, AKT1/2, and MAPK1 genes (signaling pathways–genetic variants) had different associations with breast cancer between strata, and the proportion of the SNP–cancer relationship explained by traits varied 45–50% between the strata.
Conclusions
Our findings suggest that IGF-I/IR genetic variants interact with obesity and lifestyle factors, altering cancer risk partially through pathways other than IGF-I/IR traits. Unraveling gene–phenotype–lifestyle interactions will provide data on potential genetic targets in clinical trials for cancer prevention and intervention strategies to reduce breast cancer risk.


Similar content being viewed by others
References
American Cancer Society. Breast cancer facts & figures 2015–2016: American Cancer Society, Inc. 2015. http://www.cancer.org/acs/groups/content/@research/documents/document/acspc-046381.pdf
Rose DP et al (2012) The cellular and molecular mechanisms by which insulin influences breast cancer risk and progression. Endocr Relat Cancer 19(6):R225–R241
Weichhaus M et al (2012) A novel role for insulin resistance in the connection between obesity and postmenopausal breast cancer. Int J Oncol 41(2):745–752
De Marco P et al (2014) GPER1 is regulated by insulin in cancer cells and cancer-associated fibroblasts. Endocr Relat Cancer 21(5):739–753
Muendlein A et al (2013) Association of a common genetic variant of the IGF-1 gene with event-free survival in patients with HER2-positive breast cancer. J Cancer Res Clin Oncol 139(3):491–498
Agurs-Collins T et al (2000) Insulin-like growth factor-1 and breast cancer risk in postmenopausal African-American women. Cancer Detect Prev 24(3):199–206
Li BD et al (2001) Free insulin-like growth factor-I and breast cancer risk. Int J Cancer 91(5):736–739
Ferroni P et al (2016) Pretreatment insulin levels as a prognostic factor for breast cancer progression. Oncol 21:1041–1049
Kabat GC et al (2009) Repeated measures of serum glucose and insulin in relation to postmenopausal breast cancer. Int J Cancer 125(11):2704–2710
Gunter MJ et al (2009) Insulin, insulin-like growth factor-I, and risk of breast cancer in postmenopausal women. J Natl Cancer Inst 101(1):48–60
Sieri S et al (2012) Prospective study on the role of glucose metabolism in breast cancer occurrence. Int J Cancer 130(4):921–929
Parekh N et al (2015) Insulin receptor variants and obesity-related cancers in the Framingham heart study. Cancer Causes Control 26(8):1189–1195
Argiles JM et al (2001) Insulin and cancer (review). Int J Oncol 18(4):683–687
Arcidiacono B et al (2012) Insulin resistance and cancer risk: an overview of the pathogenetic mechanisms. Exp Diabet Res 2012:789174
Al-Ajmi K et al (2012) Insulin-like growth factor 1 gene polymorphism and breast cancer risk among Arab Omani women: a case-control study. Breast Cancer 6:103–112
DeLellis K et al (2003) IGF1 genotype, mean plasma level and breast cancer risk in the Hawaii/Los Angeles multiethnic cohort. Br J Cancer 88(2):277–282
Al-Zahrani A et al (2006) IGF1 and IGFBP3 tagging polymorphisms are associated with circulating levels of IGF1, IGFBP3 and risk of breast cancer. Hum Mol Genet 15(1):1–10
Slattery ML et al (2007) Genetic variation in IGF1, IGFBP3, IRS1, IRS2 and risk of breast cancer in women living in Southwestern United States. Breast Cancer Res Treat 104(2):197–209
Wang Q et al (2014) Genetic and dietary determinants of insulin-like growth factor (IGF)-1 and IGF binding protein (BP)-3 levels among Chinese women. PLoS One 9(10):e108934
Cleveland RJ et al (2006) IGF1 CA repeat polymorphisms, lifestyle factors and breast cancer risk in the Long Island Breast Cancer Study Project. Carcinogenesis 27(4):758–765
Quan H et al (2014) IGF1(CA)19 and IGFBP-3-202A/C gene polymorphism and cancer risk: a meta-analysis. Cell Biochem Biophys 69(1):169–178
Haiman CA et al (2013) Genome-wide testing of putative functional exonic variants in relationship with breast and prostate cancer risk in a multiethnic population. PLoS Genet 9(3):e1003419
Dillon LM et al (2015) P-REX1 creates a positive feedback loop to activate growth factor receptor, PI3K/AKT and MEK/ERK signaling in breast cancer. Oncogene 34(30):3968–3976
Wang Y et al (2007) Nested case-control study of energy regulation candidate gene single nucleotide polymorphisms and breast cancer. Anticancer Res 27(1B):589–593
Zhang H et al (2013) Association between insulin receptor substrate 1 Gly972Arg polymorphism and cancer risk. Tumour Biol 34(5):2929–2936
Slattery ML et al (2015) MAPK genes interact with diet and lifestyle factors to alter risk of breast cancer: the Breast Cancer Health Disparities Study. Nutr Cancer 67(2):292–304
Yarden RI et al (2010) Single-nucleotide polymorphisms in the p53 pathway genes modify cancer risk in BRCA1 and BRCA2 carriers of Jewish-Ashkenazi descent. Mol Carcinog 49(6):545–555
Iyengar NM et al. (2013) Obesity and inflammation: new insights into breast cancer development and progression. American Society of Clinical Oncology educational book/ASCO American Society of Clinical Oncology Meeting. pp. 46-51
Wasserman L et al (2004) Correlates of obesity in postmenopausal women with breast cancer: comparison of genetic, demographic, disease-related, life history and dietary factors. Int J Obes Relat Metab Disord 28(1):49–56
Werner H (2012) Tumor suppressors govern insulin-like growth factor signaling pathways: implications in metabolism and cancer. Oncogene 31(22):2703–2714
Booth FW et al (2012) Lack of exercise is a major cause of chronic diseases. Compr Physiol 2(2):1143–1211
Yaren A et al (2012) Insulin-like growth factor I (Igf-1) gene polymorphism in patients with non-metastatic breast cancer. Gene 503(2):244–247
Pendyala S et al (2011) Diet-induced weight loss reduces colorectal inflammation: implications for colorectal carcinogenesis. Am J Clin Nutr 93(2):234–242
Creighton CJ et al (2012) A gene transcription signature of obesity in breast cancer. Breast Cancer Res Treat 132(3):993–1000
Franks SE et al (2012) Transgenic IGF-IR overexpression induces mammary tumors with basal-like characteristics, whereas IGF-IR-independent mammary tumors express a claudin-low gene signature. Oncogene 31(27):3298–3309
Clemmons DR (2007) Modifying IGF1 activity: an approach to treat endocrine disorders, atherosclerosis and cancer. Nat Rev Drug Discov 6(10):821–833
Abbas A et al (2008) Role of IGF-1 in glucose regulation and cardiovascular disease. Expert Rev Cardiovasc Ther 6(8):1135–1149
Anderson GL et al (2004) Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women’s Health Initiative randomized controlled trial. JAMA 291(14):1701–1712
Morimoto LM et al (2005) Insulin-like growth factor polymorphisms and colorectal cancer risk. Cancer Epidemiol Biomark Prev 14(5):1204–1211
Rudolph A et al (2013) Colorectal cancer risk associated with hormone use varies by expression of estrogen receptor-beta. Cancer Res 73(11):3306–3315
Campagnoli C et al (2005) Progestins and progesterone in hormone replacement therapy and the risk of breast cancer. J Steroid Biochem Mol Biol 96(2):95–108
Gunter MJ et al (2008) Insulin, insulin-like growth factor-I, endogenous estradiol, and risk of colorectal cancer in postmenopausal women. Cancer Res 68(1):329–337
Rossouw JE et al (2002) Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA 288(3):321–333
The Women’s Health Initiative Study Group (1998) Design of the Women’s Health Initiative clinical trial and observational study. The Women’s Health Initiative Study Group. Control Clin Trials 19(1):61–109
National Cancer Institute. SEER program: comparative staging guide for cancer June 1993
Matthews DR et al (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7):412–419
MacKinnon DP et al (2007) Mediation analysis. Annu Rev Psychol 58:593–614
Mackinnon DP et al (1995) A simulation study of mediated effect measures. Multivar Behav Res 30(1):41
Ingelsson E et al (2010) Detailed physiologic characterization reveals diverse mechanisms for novel genetic loci regulating glucose and insulin metabolism in humans. Diabetes 59(5):1266–1275
Dupuis J et al (2010) New genetic loci implicated in fasting glucose homeostasis and their impact on type 2 diabetes risk. Nat Genet 42(2):105–116
Pechlivanis S et al (2007) Polymorphisms in the insulin like growth factor 1 and IGF binding protein 3 genes and risk of colorectal cancer. Cancer Detect Prev 31(5):408–416
Henningson M et al (2011) IGF1 htSNPs in relation to IGF-1 levels in young women from high-risk breast cancer families: implications for early-onset breast cancer. Fam Cancer 10(2):173–185
Tamimi RM et al (2007) Common genetic variation in IGF1, IGFBP-1, and IGFBP-3 in relation to mammographic density: a cross-sectional study. Breast cancer Res 9(1):R18
Bergman D et al (2013) Insulin-like growth factor 2 in development and disease: a mini-review. Gerontology 59(3):240–249
Boyault S et al (2012) Mutational characterization of individual breast tumors: tP53 and PI3 K pathway genes are frequently and distinctively mutated in different subtypes. Breast Cancer Res Treat 132(1):29–39
Li J et al (2015) Epidermal growth factor receptor and AKT1 gene copy numbers by multi-gene fluorescence in situ hybridization impact on prognosis in breast cancer. Cancer Sci 106(5):642–649
Wheler JJ et al (2015) Multiple gene aberrations and breast cancer: lessons from super-responders. BMC Cancer 15:442
Hardt O et al (2012) Highly sensitive profiling of CD44+/CD24− breast cancer stem cells by combining global mRNA amplification and next generation sequencing: evidence for a hyperactive PI3K pathway. Cancer Lett 325(2):165–174
Andrade VP et al (2015) Gene expression profiling of lobular carcinoma in situ reveals candidate precursor genes for invasion. Mol Oncol 9(4):772–782
Acknowledgements
We thank Xu Jianfeng and Zheng Siqun Lilly (NorthShore University HealthSystem, Program for Personalized Cancer Care) for assistance with performing the genetic part. Part of the data for this project were provided by The WHI program which is funded by the National Heart, Lung, and Blood Institute, National Institutes of Health, and U.S. Department of Health and Human Services through contracts HHSN268201100046C, HHSN268201100001C, HHSN268201100002C, HHSN268201100003C, HHSN268201100004C, and HHSN271201100004C. Program Office: National Heart, Lung, and Blood Institute, Bethesda, MD: Jacques Rossouw, Shari Ludlam, Dale Burwen, Joan McGowan, Leslie Ford, and Nancy Geller. Clinical Coordinating Center: Fred Hutchinson Cancer Research Center, Seattle, WA: Garnet Anderson, Ross Prentice, Andrea LaCroix, and Charles Kooperberg. Investigators and Academic Centers: Brigham and Women’s Hospital, Harvard Medical School, Boston, MA: JoAnn E. Manson; MedStar Health Research Institute/Howard University, Washington, DC: Barbara V. Howard; Stanford Prevention Research Center, Stanford, CA: Marcia L. Stefanick; The Ohio State University, Columbus, OH: Rebecca Jackson; University of Arizona, Tucson/Phoenix, AZ: Cynthia A. Thomson; University at Buffalo, Buffalo, NY: Jean Wactawski-Wende; University of Florida, Gainesville/Jacksonville, FL: Marian Limacher; University of Iowa, Iowa City/Davenport, IA: Robert Wallace; University of Pittsburgh, Pittsburgh, PA: Lewis Kuller; Wake Forest University School of Medicine, Winston-Salem, NC: Sally Shumaker. Women’s Health Initiative Memory Study: Wake Forest University School of Medicine, Winston-Salem, NC: Sally Shumaker.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jung, S.Y., Ho, G., Rohan, T. et al. Interaction of insulin-like growth factor-I and insulin resistance-related genetic variants with lifestyle factors on postmenopausal breast cancer risk. Breast Cancer Res Treat 164, 475–495 (2017). https://doi.org/10.1007/s10549-017-4272-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-017-4272-y