Abstract
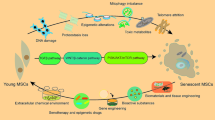
Bone marrow mesenchymal stem cells (BM-MSCs) have a momentous function in the composition of the bone marrow microenvironment because of their many valuable properties and abilities, such as immunomodulation and hematopoiesis. The features and actions of MSCs are influenced by senescence, which may be affected by various factors such as nutritional/micronutrients status, e.g., vitamin D. This study aimed to examine the effects of a high-calorie diet (HCD) with/without vitamin D on BM-MSCs senescence. In the first phase, 48 middle-aged rats were fed a normal chow diet (NCD, n = 24) and an HCD (n = 24) for 26 weeks. Afterward, the rats in each group were randomly divided into three equal subgroups. Immediately, eight-rat from each diet group were sacrificed to assess the HCD effects on the first phase measurements. In the second phase, the remaining 4 groups of rats were fed either NCD or HCD with (6 IU/g) or without vitamin D (standard intake: 1 IU/g); in other words, in this phase, the animals were fed (a) NCD, (b) NCD plus vitamin D, (c) HCD, and (d) HCD plus vitamin D for 4 months. BM-MSCs were isolated and evaluated for P16INK4a, P38 MAPK, and Bmi-1 gene expression, reactive oxygen species (ROS) levels, SA-β-gal activity, and cell cycle profile at the end of both phases. After 26 weeks (first phase), the ROS level, SA-β-gal-positive cells, and cells in the G1 phase were significantly higher in HCD-fed rats than in NCD-fed ones (P < 0.05). HCD prescription did not significantly affect cells in the S and G2 phases (p > 0.05). Compared with the NCD-fed animals, P16INK4a and P38 MAPK gene expression were up-regulated in the HCD-fed animals; also, Bmi-1 gene expression was down-regulated (P < 0.05). BM-MSCs from vitamin D-treated rats (second phase) exhibited reduced mRNA levels of P16INK4a and P38 MAPK genes and increased Bmi-1 mRNA levels (all P < 0.05). Vitamin D prescription also declined the percentage of SA-β-gal-positive cells, ROS levels, and the cells in the G1 phase and increased the cells in the S phase in both NCD and HCD-fed animals (P < 0.05). The reduction of the cells in the G2 phase in rats fed with an NCD plus vitamin D was statistically non-significant (P = 0.128) and significant in HCD plus vitamin D rats (P = 0.002). HCD accelerates BM-MSCs senescence, and vitamin D reduces BM-MSCs senescence biomarkers.





Similar content being viewed by others
References
Acar MB, Ayaz-Güner S, Di Bernardo G, Murat A, Peluso G, Özcan S, Galderisi U (2020) Obesity induced by high-fat diet is associated with critical changes in biological and molecular functions of mesenchymal stromal cells present in visceral adipose tissue. Aging 12:24894
Al-Azab M, Safi M, Idiiatullina E, Al-Shaebi F, Mohamed YZ (2022) Aging of mesenchymal stem cell: machinery, markers, and strategies of fighting. Cell Mol Biol Lett 27:1–40
Alessio N, Acar MB, Demirsoy IH, Squillaro T, Siniscalco D, Bernardo GD, Peluso G, Özcan S, Galderisi U (2020a) Obesity is associated with senescence of mesenchymal stromal cells derived from bone marrow, subcutaneous and visceral fat of young mice. Aging (Albany NY). 12(13):12609–12621. https://doi.org/10.18632/aging.103606
Alessio N, Acar MB, Demirsoy IH, Squillaro T, Siniscalco D, Gianfranco U, Peluso G, Galderisi U (2020b) Obesity is associated with senescence of mesenchymal stromal cells derived from bone marrow, subcutaneous and visceral fat of young mice. Aging 12:12609
Arif M, Khan MK, Riaz S, Nazir A, Maan AA, Amin U, Saeed F, Afzaal M (2022) Role of fruits in aging and age-related disorders. Exp Gerontol. https://doi.org/10.1016/j.exger.2022.111763
Beane OS, Vera C, Fonseca LL, Cooper G, Koren, Eric MD (2014) Impact of aging on the regenerative properties of bone marrow-, muscle-, and adipose-derived mesenchymal stem/stromal cells. PLoS ONE 9:e115963
Bi J, Li Q, Yang Z, Cai L, Lv T, Yang X, Liu L, Yan L, Wang Q, Fu X, Xiao R (2021) CXCL2 impairs functions of bone marrow mesenchymal stem cells and can serve as a serum marker in high-fat diet-fed rats. Front Cell Dev Biol. https://doi.org/10.3389/fcell.2021.687942
Borodkina AV, Shatrova AN, Nikolsky NN, Burova EB (2016) The role of p38 MAP-kinase in stress-induced senescence of human endometrium-derived mesenchymal stem cells. Cell and Tissue Biology 10:365–371
Borojević A, Jauković A, Kukolj T, Mojsilović S, Obradović H, Trivanović D, Živanović M, Zečević Ž, Simić M, Gobeljić B (2022) Vitamin D3 stimulates proliferation capacity, expression of pluripotency markers, and osteogenesis of human bone marrow mesenchymal stromal/stem cells, partly through sirt1 signaling. Biomolecules 12:323
Chen L, Yang R, Qiao W, Zhang W, Chen J, Mao L, Goltzman D, Miao D (2019) 1,25-Dihydroxyvitamin D exerts an antiaging role by activation of Nrf2‐antioxidant signaling and inactivation of p16/p53‐senescence signaling. Aging Cell 18:e12951
Chen L, Holder R, Porter C, and Zubair Shah (2021) Vitamin D3 attenuates doxorubicin-induced senescence of human aortic endothelial cells by upregulation of IL-10 via the pAMPKα/Sirt1/Foxo3a signaling pathway. PLoS ONE 16:e0252816
Chen H, Liu O, Chen S, Yueying Zhou (2022) Aging and mesenchymal stem cells: therapeutic opportunities and challenges in the older group. Gerontology 68:339–52
Chen J, Zhang J, Li J, Qin R, Lu N, Goltzman D, Miao D, Yang R (2023) 1, 25-Dihydroxyvitamin D Deficiency accelerates aging-related osteoarthritis via downregulation of Sirt1 in mice. Int J Biol Sci 19:610–624
Conley SM, LaTonya J, Hickson TA, Kellogg T, McKenzie, Julie K, Heimbach T, Taner H, Tang KL, Jordan IM, Saadiq, John RW (2020) Human obesity induces dysfunction and early senescence in adipose tissue-derived mesenchymal stromal/stem cells. Front Cell Dev Biology 8:197
Cortez M, Carmo LS, Rogero MM, Borelli P, Ricardo Ambrósio F (2013) A high-fat diet increases IL-1, IL-6, and TNF-α production by increasing NF-κB and attenuating PPAR-γ expression in bone marrow mesenchymal stem cells. Inflammation 36:379–86
da Silva S, Vargas M, Renovato-Martins C, Ribeiro‐Pereira M, Citelli, Barja‐Fidalgo C (2016) Obesity modifies bone marrow microenvironment and directs bone marrow mesenchymal cells to adipogenesis. Obesity 24:2522–2532
Dorronsoro A, Santiago FE, Grassi D, Zhang T, Lai RC, McGowan SJ, Angelini L, Lavasani M, Carbo L, Lu A (2021) Mesenchymal stem cell-derived extracellular vesicles reduce senescence and extend health span in mouse models of aging. Aging Cell 20:13337
Faraonio R (2022) Oxidative stress and cell senescence process. Antioxidants. 11(9):1718. https://doi.org/10.3390/antiox11091718
Festing MFW (2006) Design and statistical methods in studies using animal models of development. ILAR J 47:5–14
Freund A, Patil CK, Campisi J (2011) p38MAPK is a novel DNA damage response-independent regulator of the senescence‐associated secretory phenotype. EMBO J 30:1536–1548
Gao G, Li W, Fan C, Liang R, Wei C, Chen X, Yang Y, Zhong Y, Shao Y, Kong Y (2021) Mesenchymal stem cells: ideal seeds for treating diseases. Hum Cell 34:1585–600
Gerdes EO, Wissler Y, Zhu BM, Weigand U, Tripathi TC, Burns T, Tchkonia, James LK (2020) Cellular senescence in aging and age-related diseases: implications for neurodegenerative diseases. Int Rev Neurobiol 155:203–234
He S, Sharpless NE (2017) Senescence in health and disease. Cell 169(6):1000–1011. https://doi.org/10.1016/j.cell.2017.05.015
Jeong S-G, Cho G-W (2015) Endogenous ROS levels are increased in replicative senescence in human bone marrow mesenchymal stromal cells. Biochem Biophys Res Commun 460:971–76
Kim S, Rin K, Jiang M, Ogrodnik X, Chen X-Y, Zhu H, Lohmeier L, Ahmed H, Tang T, Tchkonia, LaTonya JH (2019) Increased renal cellular senescence in murine high-fat diet: effect of the senolytic drug quercetin. Translational Res 213:112–123
Klotz B, Mentrup B, Regensburger M, Zeck S, Schneidereit J, Schupp N, Linden C, Merz C, Ebert R, Jakob F (2012) 1, 25-dihydroxyvitamin D3 treatment delays cellular aging in human mesenchymal stem cells while maintaining their multipotent capacity. PLoS One 7:e29959
Li J, Karim MA, Che H, Geng Q, Dengshun, Miao (2020) Deletion of p16 prevents estrogen deficiency-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence. Am J Translational Res 12:672
Li W, Che Xu, Chen X, Zhou M, Luo X, Tao, Liu (2021a) Study of calcitriol anti-aging effects on human natural killer cells in vitro. Bioengineered 12:6844–6854
Li Y, Lu L, Xie Y, Chen X, Tian L, Liang Y, Li H, Zhang J, Liu Y, and Xijie Yu (2021b) Interleukin-6 knockout inhibits senescence of bone mesenchymal stem cells in high-fat diet-induced bone loss. Front Endocrinol 11:622950
Li X, Wang X, Zhang C, Wang J, Wang S, Lei Hu (2022) Dysfunction of metabolic activity of bone marrow mesenchymal stem cells in aged mice. Cell Prolif 55:e13191
Liu J, Gao J, Liang Z, Gao C, Niu Q, Wu F, Zhang L (2022) Mesenchymal stem cells and their microenvironment. Stem Cell Res Ther 13:429
Luque-Campos N, Bustamante-Barrientos FA, Pradenas C, García C, Araya MJ, Bohaud C, Contreras-López R, Elizondo-Vega R, Djouad F, Luz-Crawford P (2021) The Macrophage response is driven by mesenchymal stem cell-mediated metabolic reprogramming. Front Immunol 12:624746
Marampon F, Gravina GL, Festuccia C, Popov VM, Colapietro EA, Sanita P, Musio D, De Felice F, Lenzi A, Jannini EA (2016) Vitamin D protects endothelial cells from irradiation-induced senescence and apoptosis by modulating MAPK/SirT1 axis. J Endocrinol Investig 39:411–422
Minamino T, Orimo M, Shimizu I, Kunieda T, Yokoyama M, Ito T, Akira NA, Nabetani YO, Matsubara H (2009) A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nat Med 15:1082–87
Molofsky AV, Pardal R, Iwashita T, Park I-K, Clarke MF, Sean JM (2003) Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425:962–67
Neri S, Rosa Maria B (2020) Molecular mechanisms contributing to mesenchymal stromal cell aging. Biomolecules 10:340
Omidifar N, Mousavi SM, Hashemi SA, Gholami A, Shokripour M, Sohrabi Zahra (2021) ‘Trends in natural nutrients for oxidative stresscell senescence. Oxid Med Cell Longev. 2021:7501424. https://doi.org/10.1155/2021/7501424
Pini M, Czibik G, Sawaki D, Mezdari Z, Braud L, Delmont T, Mercedes R, Martel C, Buron N, Marcelin G (2021) Adipose tissue senescence is mediated by increased ATP content after a short-term high-fat diet exposure. Aging Cell 20:e13421
Qiao W, Yu S, Sun H, Chen L, Wang R, Wu X, Goltzman D, Miao D (2020) 1, 25-Dihydroxyvitamin D insufficiency accelerates age-related bone loss by increasing oxidative stress and cell senescence. Am J Translational Res 12:507
Samsonraj RM, Raghunath M, Nurcombe V, Hui JH, Andre J, van Wijnen, Simon MC (2017) Concise review: multifaceted characterization of human mesenchymal stem cells for use in regenerative medicine. Stem cells translational medicine 6:2173–2185
Shakeri H, Lemmens K, Andreas B, Gevaert, Guido RY, De Meyer, Vincent FMS (2018) Cellular senescence links aging and diabetes in cardiovascular disease. Am J Physiol Heart Circ Physiol 315:H448–H62
Sun H, Qiao W, Cui M, Yang C, Wang R, Goltzman D, Jin J, and Dengshun Miao (2020) The polycomb protein Bmi1 plays a crucial role in the Prevention of 1, 25 (OH) 2D Deficiency-Induced Bone loss. J Bone Miner Res 35:583–595
Teresa Rubio-Tomás, Rueda-Robles Ascensión, Plaza-Díaz Julio, Álvarez-Mercado Ana I (2022) Nutrition and cellular senescence in obesity-related disorders. J Nutr Biochem 99:108861
Woods K, and Borhane Guezguez (2021) Dynamic changes of the bone marrow niche: mesenchymal stromal cells and their progeny during aging and leukemia. Front Cell Dev Biology 9:714716
Xiao Y, Zhu Q, Liu X, Jiang M, Hao H, Zhu H, Peter J, Cowan X, He Q, Liu, Zhou S (2020) High-fat diet selectively decreases bone marrow lin–/CD117 + cell population in aging mice through increased ROS production. J Tissue Eng Regen Med 14:884–892
Xie C, Jin J, Lv X, Tao J, Wang R, and Dengshun Miao (2015) Anti-aging effect of transplanted amniotic membrane mesenchymal stem cells in a premature aging model of Bmi-1 deficiency. Sci Rep 5:13975
Yang R, Chen J, Zhang J, Qin R, Wang R, Qiu Y, Mao Z, Goltzman D, Miao D (2020) 1, 25-Dihydroxyvitamin D protects against age‐related osteoporosis by a novel VDR‐Ezh2‐p16 signal axis. Aging Cell 19:e13095
Yin M, Zhang Y, Haibo Yu, and, Li X (2021) Role of hyperglycemia in the senescence of mesenchymal stem cells. Front Cell Dev Biology 9:665412
Zhang H-W, Ding J, Jin Jian‐Liang, Guo J, Liu Jing‐Ning, Karaplis A, Goltzman D, Miao D (2010) Defects in mesenchymal stem cell self‐renewal and cell fate determination lead to an osteopenic phenotype in Bmi‐1 null mice. J Bone Miner Res 25:640–652
Zhang X, Xu GB, Zhou D, Yuan-Xiang P (2018) High-fat diet modifies expression of hepatic cellular senescence gene p16 (INK4a) through chromatin modifications in adult male rats. Genes & nutrition 13:1–12
Zhang Y, Liao S, Fu Q, Hamrick M (2021) Mesenchymal stem cell senescence and rejuvenation. Front Cell Develop Biol. 9:772476. https://doi.org/10.3389/fcell.2021.772476
Funding
The Iran University of Medical Sciences funded the study.
Author information
Authors and Affiliations
Contributions
FA, NA, SHM and MRJ were contributed in the research idea and final drafting of the study , FA and MH, FS, and MV were participated in experimenting. FA and FV took part in the writing.
Corresponding authors
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Agh, F., Mousavi, S.H., Aryaeian, N. et al. Senescence of bone marrow mesenchymal stem cells in Wistar male rats receiving normal chow/high-calorie diets with/without vitamin D. Biogerontology 24, 801–812 (2023). https://doi.org/10.1007/s10522-023-10048-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-023-10048-9